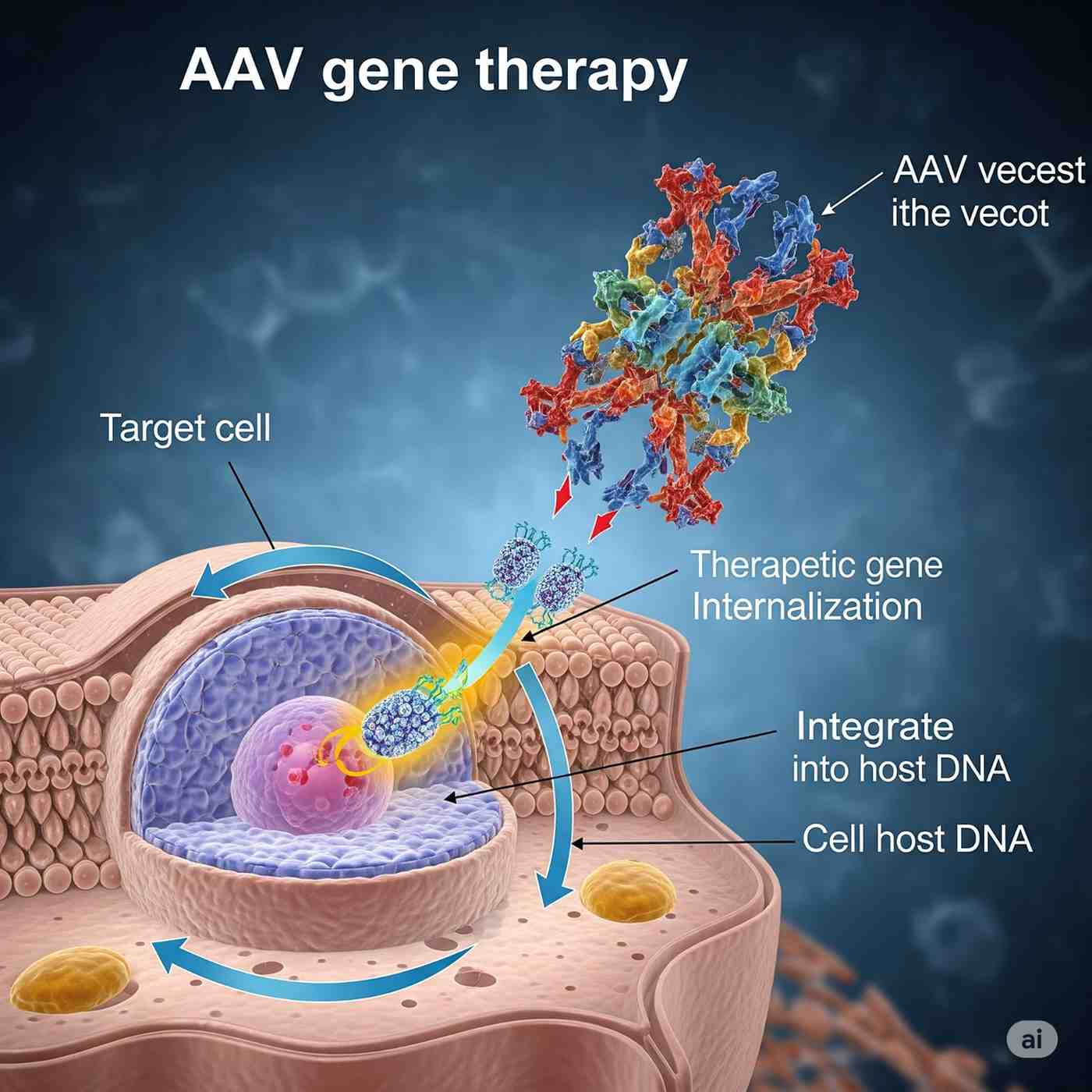
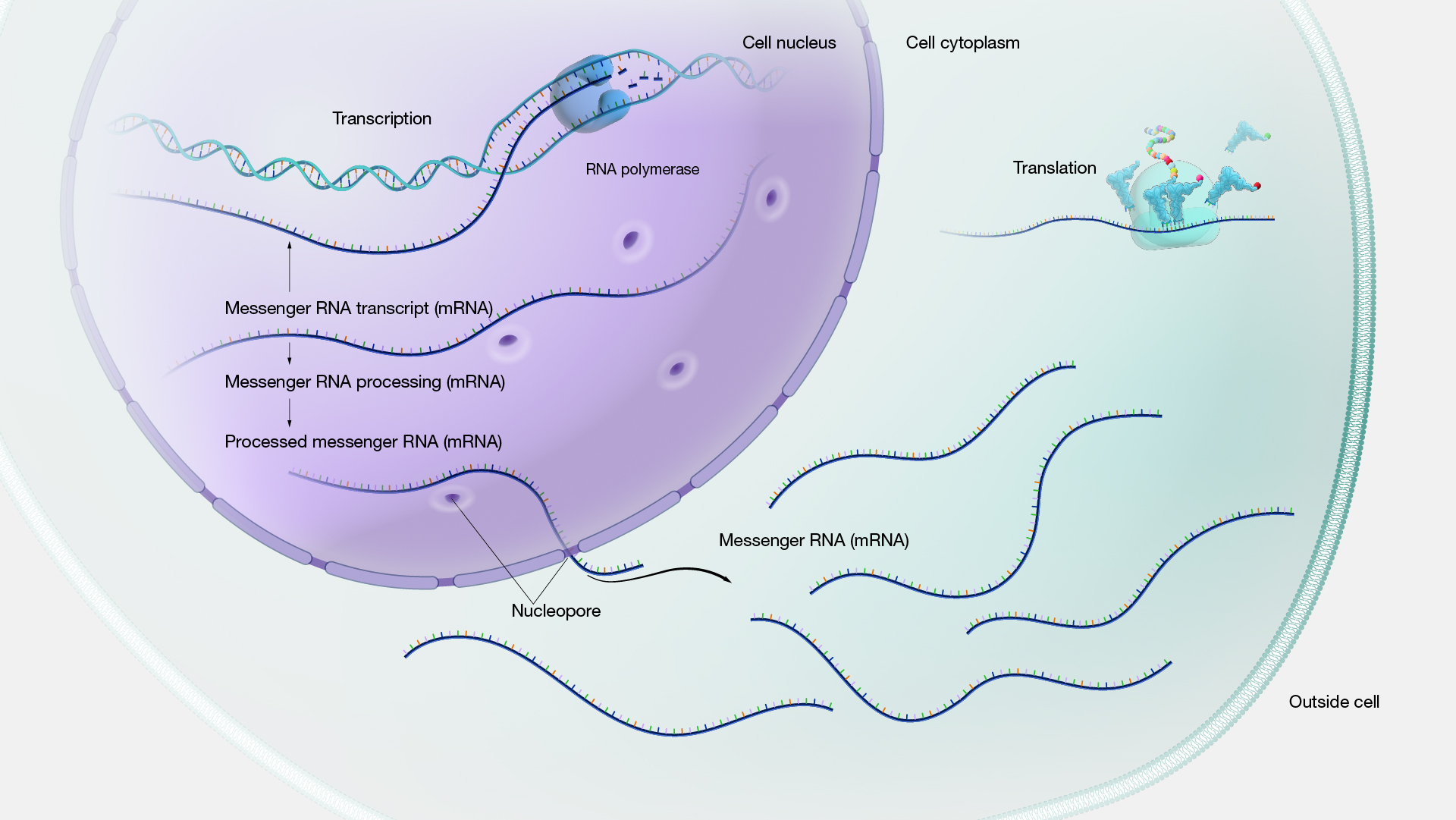
Germany stands at the forefront of Adeno-Associated Virus (AAV) gene therapy in Europe, driven by a robust biotech ecosystem, significant investments, and a commitment to addressing rare genetic disorders. This article provides a detailed analysis of Germany’s AAV gene therapy market, focusing on the years 2025 and 2026.
📊 Market Size and Growth Projections
-
2025 Market Value: Germany’s AAV gene therapy market is projected to reach approximately USD 1.2 billion.
-
2026 Forecast: The market is expected to grow at a Compound Annual Growth Rate (CAGR) of 16.5%, reaching around USD 1.4 billion by the end of 2026.
-
Global Context: Globally, the AAV gene therapy market is anticipated to expand from USD 1.2 billion in 2024 to USD 4.5 billion by 2033, reflecting a CAGR of 16.5%.
🧬 Therapeutic Applications in Germany
Germany’s AAV gene therapy applications are diverse, with significant focus areas including:
-
Oncology: Utilizing AAV vectors to deliver therapeutic genes directly to cancer cells, enhancing treatment specificity and efficacy.
-
Neurological Disorders: Addressing conditions like Alzheimer’s and Parkinson’s by delivering genes that can modify disease progression or restore lost functions.
-
Cardiovascular Diseases: Correcting genetic mutations contributing to heart diseases, potentially revolutionizing treatment approaches.
-
Genetic Disorders: Treating rare inherited conditions through AAV vectors that facilitate the delivery of corrective genes to affected tissues.
-
Infectious Diseases: Targeting underlying genetic mechanisms to improve therapeutic outcomes.
🏭 Key Industry Players
Germany hosts several leading biotech companies and research institutions contributing to the AAV gene therapy landscape:
-
BioNTech SE: Known for its mRNA technology, BioNTech is exploring AAV vectors for gene therapy applications.
-
Qiagen NV: Provides essential tools and technologies for gene therapy research and development.
-
Roche Holding AG: Through its subsidiary Spark Therapeutics, Roche is actively involved in AAV-based therapies.
-
Pfizer Inc.: Engaged in developing AAV gene therapies, including treatments for hemophilia.
-
Sarepta Therapeutics: Focuses on precision genetic medicine, including AAV-mediated therapies for neuromuscular diseases.
🔬 Research and Clinical Trials
Germany is a hub for clinical research in AAV gene therapy, with numerous ongoing trials:
-
Clinical Trials: Over 250 clinical trials worldwide have utilized AAV vectors, with Germany contributing significantly to this number.
-
Regulatory Approvals: The European Medicines Agency (EMA) approved fidanacogene elaparvovec (Beqvez) for hemophilia B in July 2024, marking a significant milestone for AAV therapies in Europe.
💰 Investment and Funding
Investment in Germany’s AAV gene therapy sector is robust:
-
Research Funding: Significant government and private sector funding support ongoing research and development.
-
Infrastructure Development: Investments are being made to enhance manufacturing capabilities and clinical trial infrastructure.
📈 Future Outlook
Germany’s AAV gene therapy market is poised for substantial growth in the coming years:
-
Innovation: Continued advancements in vector design and delivery methods are expected to improve therapy efficacy and safety.
-
Market Expansion: The market is anticipated to diversify, addressing a broader range of diseases and reaching more patients.
-
Global Leadership: Germany is set to maintain its position as a leader in the AAV gene therapy field, contributing to global advancements and setting industry standards.
Sources:
-
Wikipedia: Adeno-associated virus en.wikipedia.org
-
Wikipedia: Fidanacogene elaparvovec en.wikipedia.org
- Towards Healthcare