The global health landscape rarely witnesses a transformation powerful enough to alter how we understand, diagnose, and monitor one of the world’s most feared diseases. Yet liquid biopsy has steadily emerged from the fringes of experimental diagnostics to become one of the most promising, scientifically compelling, and clinically relevant innovations of our time. The world may still be debating its limits, but for those who have tracked this sector for more than a decade, liquid biopsy is no longer an experimental alternative but a visible turning point in oncology.
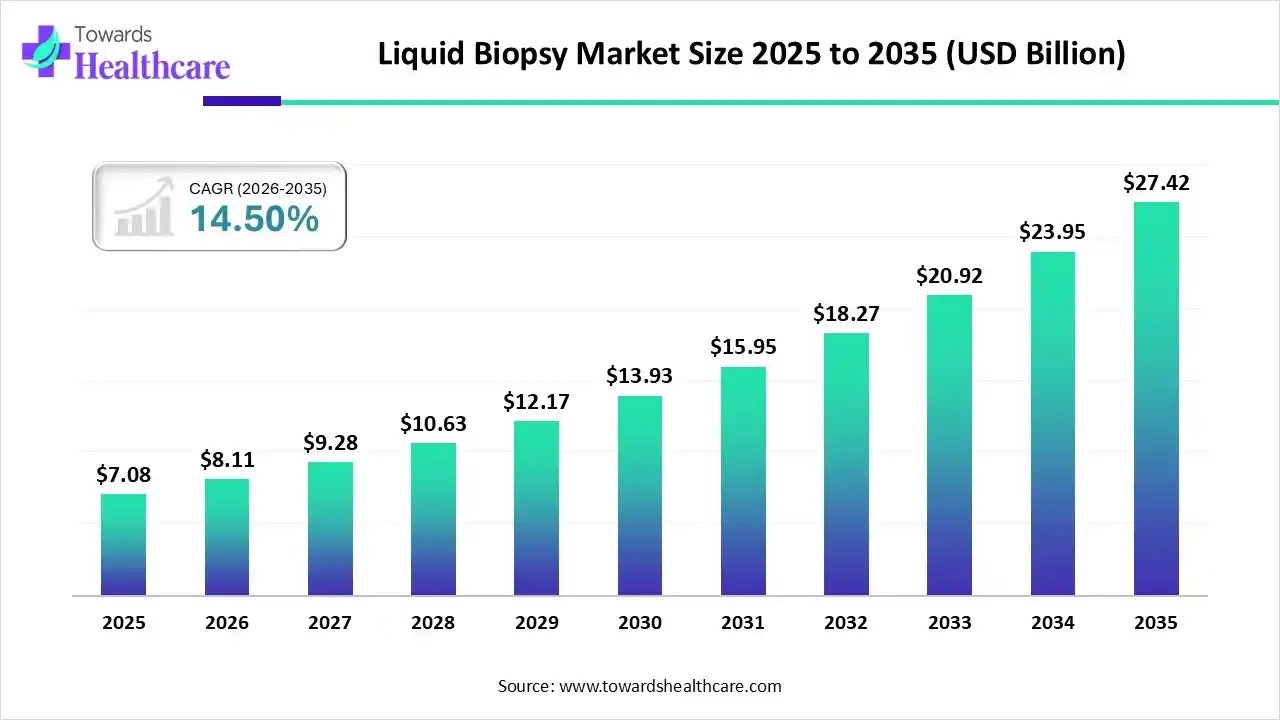
The rising liquid biopsy market size, from USD 7.08 billion in 2025 to a projected USD 27.42 billion by 2035, reflects more than commercial traction. It reflects a shift in clinical behavior, patient preference, and research direction. It represents the medical community’s growing willingness to replace invasive surgical biopsies with a molecular-level analysis of cancer through a simple blood draw. And it signifies a collective recognition that cancer cannot be fought with slow, invasive, and reactive methods in an era that demands speed, precision, and personalization.
Download Sample:
https://www.towardshealthcare.com/download-sample/5031
The Silent Revolution Behind the Blood Draw
Liquid biopsy has been discussed widely, but its fundamental value still feels under-appreciated outside scientific circles. At its core, it relies on capturing tiny fragments released by cancerous tumors, circulating tumor DNA, circulating tumor cells, and extracellular vesicles, floating in the bloodstream. These fragments hold the genetic, proteomic, and epigenetic signatures of tumors. They provide a real-time snapshot of disease progression and allow clinicians to understand what a tumor is doing without touching the tumor itself.
This is not only revolutionary; it is profoundly practical. Surgical biopsies come with pain, risk, recovery time, and sometimes even the impossibility of access when tumors lie too deep or in dangerously delicate regions. Tumors are also heterogeneous, which means that different parts of the same tumor can carry different genetic mutations. A tissue biopsy, no matter how carefully taken, samples only one part of the tumor’s total character. A liquid biopsy, on the other hand, captures what tumors shed across the body, giving doctors a panoramic molecular profile rather than a small cellular snapshot.
This paradigm shift has placed liquid biopsy squarely at the intersection of precision medicine, oncology innovation, and digital health transformation.
Why the World Is Turning Toward Less Invasive Diagnostics
There is an undeniable global tilt toward non-invasive diagnostic techniques, driven partly by rising cancer incidence and partly by shifting patient expectations. Patients today value comfort and rapid results, and healthcare systems value methods that reduce hospital stay, surgical procedures, and overall cost. As the world moves toward minimally invasive diagnostics in cardiology, neurology, and prenatal care, oncology clearly stands on the brink of the same cultural shift.
Liquid biopsy fits seamlessly into this trend. It demands minimal effort from patients while enabling doctors to repeat the test multiple times during a treatment cycle without causing physical strain. This creates a monitoring system that feels almost like a continuous feed of tumor intelligence, something unthinkable a decade ago.
The market’s rapid growth is a direct reflection of this shift. When clinicians adopt a technique not because it is new, but because it is easier, safer, and more insightful, the adoption curve becomes exponential. The current projections for the liquid biopsy market through 2035 reflect exactly that: a compound effect of medical necessity and modern expectations.
Where Precision Meets Possibility: The Technology Driving the Market
The backbone of liquid biopsy’s expansion lies in the technology that powers it. Over the years, next-generation sequencing (NGS) has become the dominant driver of multi-gene analysis. This method reads vast numbers of genetic fragments simultaneously, discovering mutations, rearrangements, and patterns hidden deep within tumor DNA. What once required tissue extraction, lab culturing, and weeks of waiting can now be accomplished within a few days, sometimes even hours.
The more established polymerase chain reaction (PCR) methods continue to evolve as well. Digital PCR models now push sensitivity limits, capturing even faint traces of tumor DNA with astonishing precision. This makes PCR-based single-gene analysis particularly valuable in cases where clinicians already suspect specific actionable mutations.
In recent years, the scientific community has also begun paying more attention to exosomes and microvesicles, small particles that tumors release into the bloodstream carrying proteins, genetic materials, and critical molecular information. Their structural integrity makes them especially valuable for early detection and treatment monitoring. They are rapidly becoming one of the fastest-growing biomarker categories in liquid biopsy research and commercialization.
These technological advancements are not isolated scientific achievements; they form the foundation of the market’s accelerating growth and the industry’s widening confidence.
A New Kind of Lens on Cancer: From Screening to Surveillance
Where liquid biopsy truly distinguishes itself is in its versatility. Unlike many diagnostic tools designed for a single purpose, liquid biopsy sits comfortably across multiple stages of the cancer care continuum.
In early detection, it offers the possibility of identifying cancers before symptoms appear, potentially enabling treatment at the stage where survival probability is highest. While early-stage sensitivity remains one of the field’s ongoing challenges, the combination of advanced sequencing and multi-analyte biomarker profiling is gradually improving the resolution of early screening technologies.
During treatment selection, liquid biopsy serves as a molecular compass, helping oncologists navigate the complexities of targeted therapies. As cancer treatments grow more personalized and mutation-specific, the demand for accurate genetic mapping grows in parallel. Liquid biopsy fills this gap efficiently by offering real-time tumor profiles.
In monitoring disease progression, it provides dynamic updates about how tumors respond to therapy. When tumors mutate and develop resistance, liquid biopsy detects these changes swiftly, often before symptoms return or imaging scans reveal progression. This creates a window of opportunity for clinicians to adjust therapeutic strategies proactively.
Its value extends to minimal residual disease (MRD) detection as well, which helps determine whether microscopic traces of cancer remain in the body after treatment. This is one of the field’s most promising applications, with the potential to prevent relapse through early intervention.
The cumulative effect is a new clinical tool that operates like a live feed of tumor behavior, something that traditional biopsy could never offer.
When AI Enters the Equation: The New Frontier in Liquid Biopsy
Artificial intelligence has begun reshaping the liquid biopsy ecosystem in ways that were unimaginable ten years ago. AI can recognize molecular patterns hidden deep within raw sequence data, predict therapy response based on mutation combinations, and reduce analytical errors typically associated with manual interpretation.
The integration of AI gives liquid biopsy tests an edge in accuracy and speed. Algorithms can analyze thousands of genetic markers simultaneously, identify weak signals, and classify mutation profiles with high confidence. This is particularly useful in cancers like triple-negative breast cancer, where biomarker identification is historically difficult.
AI-diagnostics partnerships are emerging at a rapid pace, merging computational intelligence with biological insight. These collaborations are building pipelines capable of handling high-complexity data while generating clinically actionable reports almost instantly. AI does not replace the science behind liquid biopsy; it elevates it.
The combination of digital pathology, machine learning models, and cloud-based genomic workflows is beginning to transform cancer diagnostics into a technology-driven discipline. In this landscape, liquid biopsy becomes not just a test, but a data engine.
The Rising Global Momentum and the Push from Health Systems
Governments and public health systems worldwide are paying closer attention to liquid biopsy’s potential to reduce healthcare burden and improve early detection. Countries with rising cancer incidence are exploring ways to integrate non-invasive diagnostics into national screening programs.
In nations like India, large-scale initiatives under programs similar to Ayushman Bharat aim to support cancer care for economically vulnerable populations. As policymakers recognize the value of early and minimally invasive diagnostics, liquid biopsy becomes a natural fit for enhancing accessibility and reducing long-term treatment costs.
Healthcare institutions also view liquid biopsy as a facilitator of faster decision-making. Instead of waiting weeks for tissue biopsy analysis, oncologists can shorten treatment timelines, personalize choices more effectively, and monitor response with unprecedented clarity. This systemic shift fuels market expansion more strongly than commercial demand alone.
Beyond the Buzz: The Real Challenges Slowing Liquid Biopsy Adoption
Despite its promise, the liquid biopsy landscape still faces several scientific and operational hurdles that require attention. Early-stage tumor detection remains one of the field’s most persistent challenges. Tumors in their initial stages shed minimal circulating DNA, making accurate identification difficult even with highly sensitive technologies.
Another recognized limitation lies in determining the tumor’s exact anatomical origin. Liquid biopsy reveals genetic mutations circulating in the bloodstream but does not pinpoint the physical location of the tumor. This creates reliance on imaging scans and other diagnostic modalities for comprehensive assessment.
The field also grapples with technical inconsistencies. Variation in sample collection, processing methods, and analytical platforms can affect test reproducibility. False positives and false negatives, while reducing over time, continue to require rigorous validation.
Moreover, the cost of advanced sequencing or multi-panel mutation analysis still poses accessibility challenges in low-income regions. The promise of universal adoption hinges on closing this affordability gap.
However, none of these challenges negate the value of liquid biopsy. In fact, they serve as focal points for ongoing innovation, regulatory refinement, and clinical trial activity.
The Unfolding Opportunity: A New Era in Personalized Oncology
The rapid expansion of liquid biopsy aligns with the broader global movement toward personalized medicine. Tumors evolve, mutate, and diversify throughout their lifecycle. A one-time tissue biopsy cannot capture this dynamic progression. Liquid biopsy, with its ability to offer repeated insights, becomes essential in guiding long-term therapy strategies.
This gives rise to new possibilities in oncology care. Patients may benefit from earlier interventions, better treatment alignment, and more accurate surveillance after remission. Clinicians gain an unprecedented level of control over decision-making. And healthcare systems move toward a future where outcomes improve not just because treatments evolve, but because diagnostics evolve faster.
The future also holds promise for integrating liquid biopsy into large-scale population screening. As technologies mature and costs decline, liquid biopsy could emerge as a mainstream method for early cancer detection, particularly for hard-to-detect cancers with historically poor survival rates.
The implications extend beyond cancer. Rapid progress in reproductive health applications, transplant medicine, infectious disease monitoring, and autoimmune disorder evaluation hints that liquid biopsy’s molecular principles are far from limited to oncology alone.
A Market Growing Not Just in Value, but in Vision
When analysts discuss the liquid biopsy market reaching more than USD 27 billion within the next decade, they often frame it as an economic milestone. But the real story lies in what this growth represents, a shift from invasive diagnostics to minimally invasive molecular intelligence. A shift from delayed treatment decisions to real-time intervention. A shift from fragmented tumor sampling to comprehensive molecular mapping.
The market is expanding because clinicians increasingly trust the technology. It is expanding because patients prefer comfort over complication. It is expanding because scientific breakthroughs keep raising the ceiling of what is possible. And it is expanding because healthcare systems recognize that early detection is not an expense, it is an investment.
Instruments currently dominate the market, but the rapid rise of kits and reagents suggests that liquid biopsy is becoming more modular, more accessible, and more adaptable. Blood remains the dominant sample type, but technological innovations may broaden the scope to include saliva, urine, and other non-blood samples, widening the diagnostic frontier further.
Hospitals and laboratories continue to lead in adoption, while specialty clinics grow rapidly as precision oncology becomes a mainstream approach rather than a luxury reserved for major institutions.
Where the Future Points: The Path from Potential to Standard Practice
The journey of liquid biopsy mirrors the trajectory of many transformative medical innovations: skepticism in the beginning, gradual acceptance by researchers, cautious adoption by clinicians, confident integration into treatment plans, and eventual recognition as a standard tool.
We are now positioned between confident integration and worldwide standardization. The next decade will determine how far liquid biopsy spreads into everyday clinical practice. If current trends continue, liquid biopsy may soon stand alongside imaging and blood testing as one of the essential components of cancer care.
Its potential lies not only in what it offers today, but in what it promises tomorrow: a future where cancer is detected earlier, treated more accurately, monitored more efficiently, and understood more deeply.
Is This the Beginning of a New Diagnostic Era?
Liquid biopsy represents a rare convergence of technological maturity, clinical relevance, and patient-centered design. It brings together genomics, molecular biology, computational intelligence, and real-world clinical needs. It removes barriers that once slowed cancer diagnosis and replaces them with a method rooted in comfort, speed, and scientific precision.
For those who have observed this field evolve over the past ten years, the transformation feels nothing short of historic. The world of oncology is moving toward rapid, minimally invasive, data-rich diagnostics, and liquid biopsy stands at the center of that movement.
As the market marches toward 2035 with increasing confidence, one question remains: are we witnessing a technological evolution, or the opening chapter of a diagnostic revolution that will redefine how humanity understands cancer? The answer, as always, lies in the science, and in the countless lives that stand to benefit from it.
Access our exclusive, data-rich dashboard dedicated to the Biotechnology Industry – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Important and Quick Links:
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout Liquid Biopsy Market Report Now at: https://www.towardshealthcare.com/checkout/5031
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest