Is the 3D Bioprinting Market Ready to Solve the Global Organ Shortage?

For more than a decade, I have watched 3D bioprinting evolve from an ambitious laboratory concept into a technology that is steadily redefining how we think about human tissues, organs, and disease modeling. What once sounded like science fiction has now entered a phase of practical experimentation, clinical relevance, and serious commercial interest.
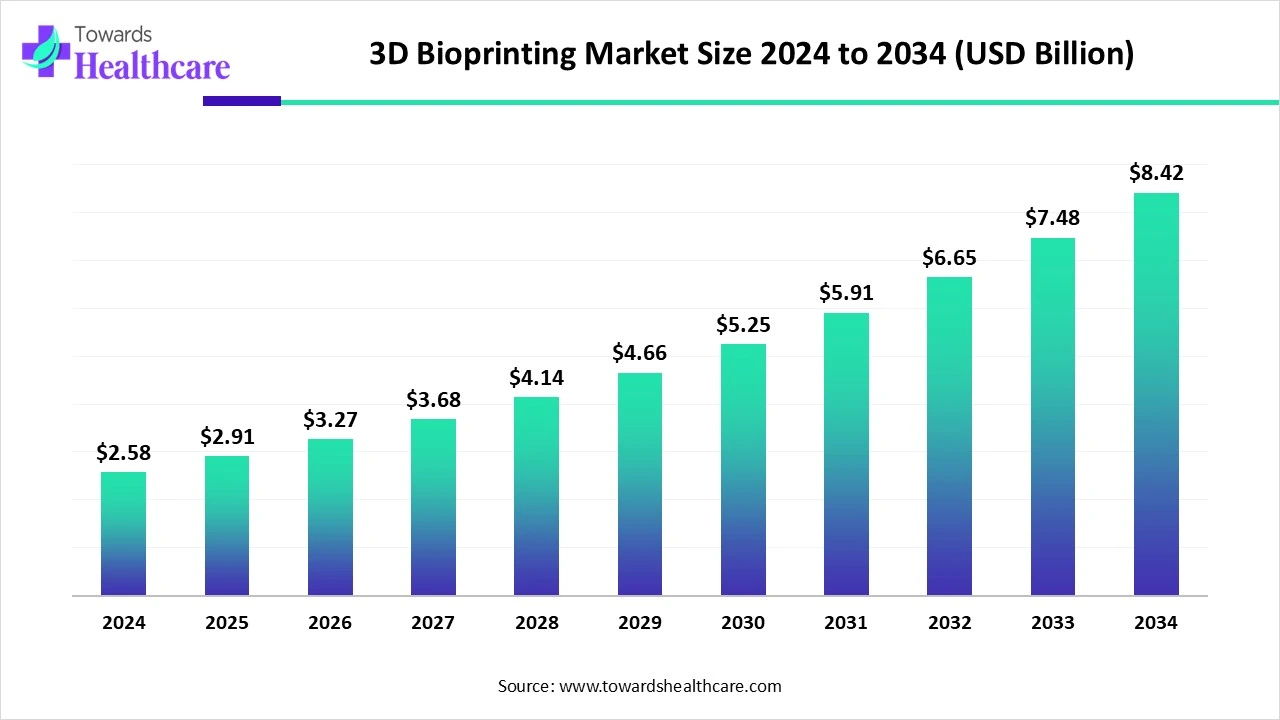
The global 3D bioprinting market, valued at around USD 2.58 billion in 2024 and projected to grow to approximately USD 8.42 billion by 2034, reflects not hype but a measured confidence built on scientific progress and real-world demand.
Download Free Sample and Get the Complete Report Easily at: https://www.towardshealthcare.com/download-sample/5784
This market is not growing because it is fashionable. It is expanding because traditional healthcare systems face structural problems that conventional solutions cannot solve. Organ shortages persist across the world, drug development remains expensive and slow, and personalized medicine still struggles to move beyond theory. 3D bioprinting sits at the intersection of these challenges, offering a new way to build, test, and repair biological systems rather than merely treat symptoms.
From Plastic Prototypes to Living Structures
The most common misunderstanding about 3D bioprinting is that it is simply an extension of conventional 3D printing. In reality, the leap from printing inert materials to printing living tissues required a complete rethinking of materials, processes, and outcomes. Instead of plastics or metals, bioprinters work with bioinks composed of living cells, biomaterials, and growth factors. These elements must survive the printing process, interact with each other, and mature into functional tissue.
Over the years, the industry has learned how to better mimic the microarchitecture of natural tissues. Early bioprinted constructs often lacked structural integrity or long-term cell viability. Today, improved inkjet-based systems allow precise placement of cells at relatively low cost, which explains why this technology dominated the market in 2024. At the same time, newer approaches such as magnetic levitation are gaining momentum because they allow cells to assemble without mechanical stress, preserving their biological function.
This shift from structural accuracy to biological performance marks a critical turning point. The conversation is no longer about whether tissues can be printed, but about how closely they can replicate natural behavior inside the human body.
Why Medicine Became the Center of Gravity
Medical applications dominate the 3D bioprinting market for a simple reason: the unmet need is immense. Organ transplantation systems worldwide operate under permanent scarcity. Waiting lists grow longer each year, while donor availability fails to keep pace. Bioprinted tissues offer a potential path to reduce this gap by creating patient-specific constructs that lower rejection risks and improve outcomes.
Beyond transplantation, the medical sector has embraced bioprinting for drug testing, disease modeling, and regenerative therapies. Traditional drug testing relies heavily on animal models or simplified cell cultures, both of which often fail to predict human responses accurately. Bioprinted tissues provide a more realistic environment, allowing researchers to observe how drugs interact with human-like structures before entering clinical trials.
This is why tissue and organ generation stands out as the fastest-growing application sub-segment. It aligns perfectly with the broader shift toward personalized medicine, where treatments are tailored to individual genetic and biological profiles rather than standardized averages.
Asia Pacific’s Rise Signals a Global Shift
Asia Pacific’s leadership in the 3D bioprinting market in 2024 is not accidental. The region combines large patient populations, expanding healthcare infrastructure, and strong government interest in advanced manufacturing technologies. Countries such as China and India view bioprinting not only as a medical innovation but as a strategic industry that can reduce long-term healthcare costs and dependency on imports.
In China, government-backed research initiatives and funding programs have accelerated academic and industrial collaboration. The focus extends beyond transplantation to include disease modeling and pharmaceutical research. India’s growth follows a different but equally compelling logic. Rising chronic disease rates, a strong medical tourism sector, and cost-efficient production capabilities have created an environment where bioprinting can scale faster than in many Western markets.
These regional dynamics suggest that innovation in bioprinting will no longer remain concentrated in a handful of countries. Instead, we are witnessing the early stages of a globally distributed innovation ecosystem.
North America’s Strength Lies in Integration
While Asia Pacific leads in market share, North America continues to play a defining role in technological integration and commercialization. The region benefits from mature healthcare systems, advanced research institutions, and a well-developed startup ecosystem. What sets North America apart is its ability to combine bioprinting with adjacent technologies such as artificial intelligence, advanced imaging, and automation.
AI integration has moved bioprinting from a largely manual, trial-and-error process toward a data-driven discipline. Algorithms now assist in selecting optimal bioink compositions, adjusting printing parameters in real time, and reconstructing complex anatomical models from medical images. This reduces variability, improves reproducibility, and shortens development cycles.
In practical terms, this means faster iteration, lower failure rates, and greater confidence in scaling bioprinted solutions from lab to clinic.
Europe’s Research-Driven Momentum
Europe’s role in the 3D bioprinting landscape reflects its strong academic and regulatory foundations. The region places significant emphasis on safety, ethical considerations, and long-term clinical validation. Research institutions across Germany, the UK, and other European countries have focused heavily on developing new bioinks, improving tissue complexity, and establishing standardized protocols.
This research-first approach may slow commercialization in the short term, but it builds trust and credibility that are essential for long-term adoption. Regulatory clarity, particularly around regenerative medicine and personalized therapies, gives European innovators a stable framework within which to operate.
Bioinks Are Becoming the True Differentiator
If bioprinters are the engines of this industry, bioinks are its fuel. Over the past few years, innovation has shifted decisively toward improving bioink performance. Early formulations often struggled with issues such as poor cell viability, limited mechanical strength, or complex crosslinking requirements.
Recent developments have focused on making bioinks more biocompatible, easier to handle, and suitable for a wider range of tissues. Advances in plant-based and synthetic bioinks aim to improve scalability and consistency, addressing one of the biggest barriers to commercialization. The ability to mix cells directly into bioinks without harsh curing processes represents a significant step forward for regenerative medicine.
As bioinks become more specialized, they will increasingly define competitive advantage within the market.
Investment Patterns Reflect Long-Term Confidence
Venture capital and private equity interest in 3D bioprinting has matured alongside the technology itself. Early investments targeted proof-of-concept startups. Today, funding increasingly supports platforms with clear pathways to clinical or industrial application. Investors recognize that bioprinting is not a quick-return industry, but one with the potential to reshape healthcare economics over decades.
Startups focusing on proprietary printing technologies, AI integration, or niche applications such as organ-on-chip systems have attracted sustained interest. This capital influx has helped transform experimental tools into robust platforms capable of meeting regulatory and clinical standards.
The Infrastructure Challenge Remains Real
Despite its promise, 3D bioprinting faces undeniable constraints. High equipment costs, the need for sterile environments, and reliance on skilled personnel limit accessibility, particularly in lower-resource settings. These barriers slow adoption and concentrate advanced capabilities within specialized centers.
However, history suggests that such constraints often diminish over time. As technologies mature, costs fall, workflows simplify, and expertise spreads. The current phase mirrors early developments in medical imaging and genomic sequencing, both of which were once prohibitively expensive and are now widely accessible.
AI Is Redefining What Is Possible
The integration of artificial intelligence into 3D bioprinting represents one of the most profound shifts I have witnessed. AI does not merely automate existing processes; it changes how problems are approached. By learning from historical data, AI systems can predict optimal printing conditions, identify defects before they occur, and adapt designs to patient-specific needs.
This capability transforms bioprinting into a responsive system rather than a static one. It opens the door to printing complex structures that would be nearly impossible to design manually, particularly when dealing with irregular defect sites or highly customized tissues.
Beyond Transplants: The Broader Impact
While organ transplantation captures public imagination, the broader impact of 3D bioprinting may emerge elsewhere. Disease modeling, fertility research, dental applications, and pharmaceutical testing already benefit from bioprinted structures. These use cases generate value long before fully functional organs become clinically routine.
Each incremental success builds confidence among regulators, clinicians, and investors. Over time, these cumulative advances will lower the barriers to more ambitious applications.
Looking Ahead Without Illusions
After years in this field, I remain optimistic but realistic. 3D bioprinting will not solve the global organ shortage overnight, nor will it replace traditional medical manufacturing in the near term. What it will do is gradually redefine how we approach biological repair, testing, and personalization.
The projected growth to USD 8.42 billion by 2034 reflects steady progress rather than speculative frenzy. It suggests a technology finding its place within healthcare systems, supported by science, policy, and patient need.
The real story of 3D bioprinting is not about printing organs tomorrow. It is about building the foundation for a future where medicine moves from replacement to regeneration, from generalized treatment to biological precision. And that transformation, while slow, is already underway.
Access our exclusive, data-rich dashboard dedicated to the biotechnology industry – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout 3D Bioprinting Market Report Now at: https://www.towardshealthcare.com/checkout/5784
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest
Table of Contents
Toggle



