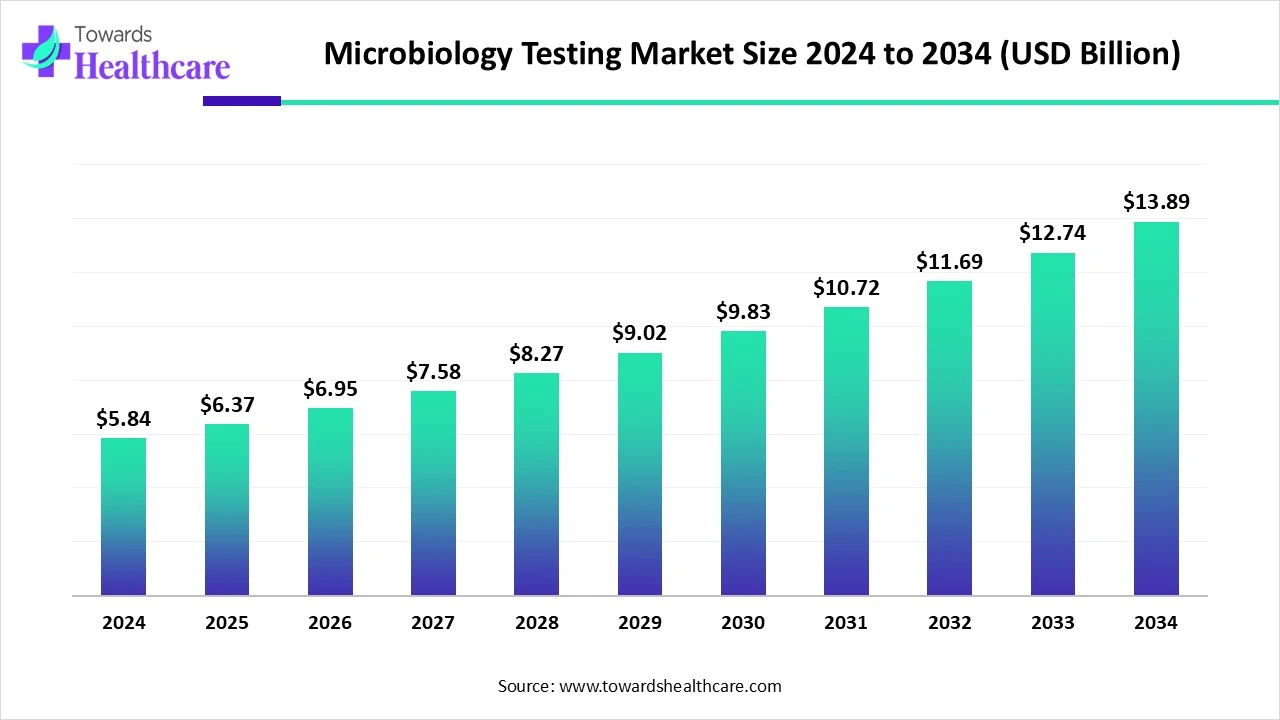
The global microbiology testing market was valued at USD 5.84 billion in 2024, grew to USD 6.37 billion in 2025, and is projected to reach USD 13.89 billion by 2034, expanding at a CAGR of 9.04% (2025–2034), with North America leading (46% share) and Asia Pacific emerging as the fastest-growing region.
Download the free sample and get the complete insights and forecasts report on this market @ https://www.towardshealthcare.com/download-sample/5629
Market Size
◈2024 Market Value: USD 5.84 billion.
◈2025 Market Value: USD 6.37 billion (YoY growth driven by hospital testing and advanced instrument adoption).
◈2034 Projection: USD 13.89 billion.
◈CAGR (2025–2034): 9.04% — steady mid-high growth due to demand in healthcare, food safety, and pharma.
◈Regional Leader (2024): North America at 46% share.
◈Fastest Growing Region: Asia Pacific due to healthcare expansion, population size, and public health investments.
◈Dominant Product (2024): Instruments (PCR machines, autoclaves, incubators, etc.).
◈Fastest-Growing Product Segment: Reagents (used in microbial classification, identification, antibiotic testing).
Market Trends (Deep Analysis)
R&D & Grants Boosting Microbiome Research
◈Feb 2025: Zymo Research launched the Master the Microbiome Grant ($5,000 credits) to advance metagenomics and microbiome sciences.
AI-driven Innovations in Factory Quality Control
◈Feb 2025: Spore.Bio raised €22M Series A to scale AI-based microbial testing for industrial quality control.
Growing Role of Rapid Diagnostics
◈Rapid testing (PCR, biosensors, molecular diagnostics) gaining ground for infectious disease containment and hospital decision-making.
Food Safety as a Global Driver
◈WHO: 600M people fall ill annually, 420,000 deaths from foodborne illness → increasing global adoption of microbiology testing.
Emerging Disease Threats Driving Demand
◈Rising viral outbreaks (COVID-19, influenza variants) and antibiotic resistance push demand for bacterial/viral diagnostics.
Technological Advancement
◈Biosensors, metagenomics, and automation in microbiology labs expanding test accuracy and speed.
AI’s Impact on the Microbiology Testing Market (Deep-Dive)
Accelerated Diagnostics
◈From Days to Hours: Traditional culture-based bacterial identification takes 24–72 hours; AI-powered image recognition and machine learning algorithms can analyze growth patterns, MALDI-TOF spectra, or genomic signatures in minutes.
Clinical Relevance:
◈Early detection of sepsis-causing pathogens reduces mortality by 30–40%.
◈AI-aided MALDI-TOF platforms identify rare pathogens that standard protocols may miss.
◈Market Implication: Hospitals see reduced ICU stays and costs, fueling adoption of AI-powered platforms despite high upfront investment.
Antibiotic Resistance Prediction
◈Problem: AMR is projected to cause 10 million deaths annually by 2050. Traditional AST (antibiotic susceptibility testing) takes 2–3 days.
AI Edge:
◈Deep learning models interpret whole-genome sequencing (WGS) and metagenomic data to forecast resistance even before cultures show results.
◈AI platforms like PathAI and IBM’s Deep Genomics are exploring predictive AMR surveillance.
Public Health Angle:
◈Early warning systems for carbapenem-resistant Enterobacteriaceae (CRE) and MRSA outbreaks.
◈Data integration with global surveillance networks (WHO’s GLASS, CDC’s NARMS).
◈Market Implication: Pharma companies can design narrow-spectrum, precision antibiotics targeting predicted resistance profiles.
Drug Development Support
AI + Microbiome:
◈The human microbiome influences drug metabolism, immune response, and efficacy of antibiotics or cancer immunotherapies.
◈AI maps microbe–host interactions to identify new therapeutic targets.
Applications:
◈Screening billions of microbial metabolites for novel antibiotic candidates.
◈Identifying microbiome markers predicting patient response to checkpoint inhibitors in oncology.
◈Strategic Implication: Biopharma pipelines shorten by years, lowering R&D costs while improving trial success rates.
Epidemiological Monitoring
AI-Driven Surveillance:
◈Real-time analysis of hospital lab data, EHRs, and wastewater sequencing to detect outbreak signals.
◈Machine learning models predict spread dynamics of influenza, TB, and COVID-19 variants.
◈Case Example: During COVID-19, AI models helped forecast ICU demand and detect emerging Omicron clusters before traditional surveillance caught up.
◈Market Implication: Governments and NGOs fund AI-backed platforms for biosecurity and global health preparedness.
Automation in Testing Labs
◈Challenge: Global shortage of clinical microbiologists and skilled technicians.
AI Role:
◈Automated plate reading, colony picking, and slide interpretation.
◈Smart analyzers integrate robotics + AI to handle high-volume testing with minimal human intervention.
Examples:
◈BD Kiestra™ total lab automation system.
◈Copan WASPLab with AI-assisted plate reading.
◈Impact: Efficiency gains up to 40% reduction in turnaround time; scalability without proportionally increasing workforce.
Industrial Applications
◈Food & Beverage: AI systems scan microbial genomic data to detect contaminants (Listeria, Salmonella, E. coli) in real time during production.
Pharmaceutical Manufacturing:
◈AI-enabled environmental monitoring in cleanrooms → prevents contamination before batch loss.
◈Predictive models identify contamination risk hotspots in supply chains.
Regulatory Compliance:
◈Faster, AI-backed QC aligns with FDA’s Quality by Design (QbD) and EMA’s Annex 1 guidelines for sterile manufacturing.
◈Market Implication: Adoption in F&B and pharma QC labs ensures not just safety but also cost savings from avoiding recalls.
Regional Insights
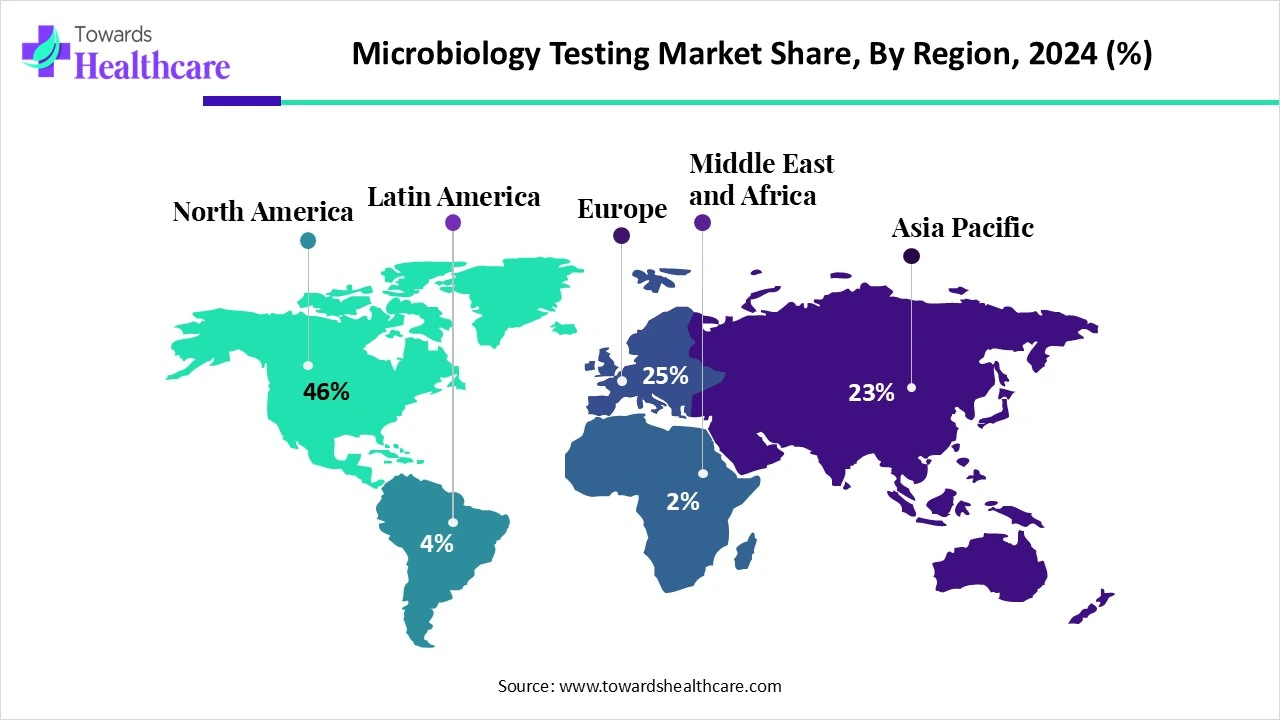
1. North America (46% Market Share in 2024)
◈North America remains the global leader in microbiology testing, driven by advanced healthcare infrastructure, robust funding, and a concentration of leading global diagnostic companies.
Strengths
◈Advanced healthcare infrastructure with well-established diagnostic laboratories.
◈Presence of leading players such as BD, Thermo Fisher, Abbott, and Roche, ensuring early adoption of cutting-edge technologies.
◈Strong regulatory framework (FDA, CDC, NIH) that enforces stringent food and clinical safety testing standards.
Key Drivers
◈High prevalence of infectious diseases including TB, influenza, HIV, and hospital-acquired infections.
◈Adoption of microbiology testing as a core hospital diagnostic service — diagnostics guide 60–70% of medical decisions in the region.
◈Strong government investments into pandemic preparedness and antibiotic resistance management.
United States
◈The Global Health Security and Diplomacy Bureau (GHSD) leads international collaboration to prevent, detect, and respond to infectious diseases.
◈Heavy investment in AI-driven and rapid diagnostic technologies to manage drug resistance and pandemics.
Canada
◈Focus on enteric infections linked to international travel.
◈Expansion of HIV and STBBI (sexually transmitted and blood-borne infections) testing, with emphasis on Indigenous and rural populations.
◈Government allocated $26.5M (2022–2024) for HIV self-testing kits and community programs.
2. Asia Pacific (Fastest Growing Region)
◈Asia Pacific is expected to outpace other regions in growth, driven by rapid healthcare modernization, population growth, and increasing food safety regulations.
China
◈Target by 2030: establish world-leading epidemic monitoring systems and early warning labs.
◈Heavy investment in laboratory networks across ministries for infectious disease control.
India
◈Longstanding CDC partnerships enhance outbreak response, epidemiological surveillance, and lab infrastructure.
◈Focus on combating vector-borne and water-borne infections with microbiology testing expansion.
Japan
◈Significant investments in modernizing healthcare labs and scaling up infectious disease preparedness.
◈High adoption of automation and AI-driven microbiology instruments in hospitals and research centers.
Overall Regional Drivers
◈Expansion of healthcare facilities in both public and private sectors.
◈Growing focus on food safety regulations amid rising export and domestic consumption.
◈Strong pharmaceutical and biotechnology presence fueling demand for drug safety and microbial monitoring.
3. Europe (Strong Growth)
◈Europe’s market is driven by strict regulations, an aging population vulnerable to infections, and rising antibiotic resistance concerns.
Germany
◈Leads antimicrobial resistance (AMR) management via DART 2030 strategy.
◈Focus on cross-sector collaboration across agriculture, veterinary medicine, and healthcare to limit AMR.
United Kingdom
◈20-year National Action Plan (NAP) against AMR, aiming to reduce overuse of antibiotics in humans and livestock.
◈Investment in next-gen diagnostic tools for early infection detection and antibiotic stewardship.
Key Drivers
◈Stringent food safety and healthcare regulations boosting microbiology testing demand.
◈Aging population driving higher demand for hospital-based microbiology diagnostics.
◈Rising cases of drug-resistant pathogens prompting stronger lab investments.
4. Latin America
◈Latin America shows strong growth potential due to rising healthcare spending, infectious disease prevalence, and increased collaborations with global testing companies.
Brazil
◈Listed in two WHO high TB-burden lists (general TB and HIV-associated TB).
◈TB prevalence: >150 cases per 100,000 annually.
◈Government prioritizes advanced diagnostic technologies to manage TB/HIV dual burden.
Mexico
◈TB prevalence: 23 cases per 100,000 population (2023).
◈Presence of global testing service providers such as SGS SA, Intertek, and Mérieux NutriSciences.
◈Rising demand for food safety and clinical diagnostics to support export and healthcare goals.
Key Driver
◈Rising infectious disease prevalence, coupled with growing healthcare spending and adoption of advanced testing technologies.
5. Middle East & Africa (MEA)
◈MEA is emerging as a growth hub, supported by government investments, expansion of diagnostic labs, and adoption of AI-based testing solutions.
United Arab Emirates (UAE)
◈2024: Smart Salem launched AI-powered metagenomic WGS microbiome test for comprehensive microbial diversity analysis.
◈Strong emphasis on AI and personalized medicine integration in diagnostics.
South Africa
◈High TB burden: 280,000 TB cases and 55,000 deaths (2023).
◈National SDG goal: eradicate TB by 2030 (80% reduction in infections, 90% reduction in deaths).
◈Increased funding into public health diagnostic infrastructure.
Key Drivers
◈Rising prevalence of infectious diseases across the region.
◈Strong government support for healthcare modernization and lab expansion.
◈Growing demand for point-of-care testing in rural and underserved areas.
Market Dynamics
Drivers
Food Safety Awareness
◈WHO: 600M people fall ill, 420,000 deaths annually due to contaminated food.
◈Drives adoption of rapid microbial testing in food and beverage industries.
Infectious Disease Prevalence
◈Growing cases of TB, HIV, influenza, COVID-19 variants increasing demand for bacterial and viral tests.
Technological Advancements
◈Adoption of PCR, biosensors, AI-based diagnostics, and molecular testing across clinical and industrial settings.
Restraints
Lack of Skilled Workforce
◈Advanced clinical microbiology testing requires highly trained professionals, limiting adoption in low-resource regions.
High-Tech Complexity
◈Sophisticated equipment (PCR, metagenomics platforms) face real-time technical issues and cost barriers.
Opportunities
AI & Automation
◈Automated microbiology platforms reducing turnaround times and reliance on human expertise.
Expanding Applications
◈Environmental monitoring, pharmaceutical testing, and food safety creating new revenue streams.
Government Investments
◈National strategies in China, India, EU, and Africa expanding lab infrastructure and epidemic monitoring systems.
Top Companies

1. Bio-Rad Laboratories Inc.
◈Strength: Wide molecular diagnostics portfolio.
◈Focus: Clinical diagnostics and advanced research.
2. Abbott Laboratories
◈Strength: Strong infectious disease testing platforms.
◈Focus: Hospital diagnostics and rapid tests.
3. Becton Dickinson (BD)
◈Strength: Market leader in diagnostic instruments & consumables.
◈Focus: Automated microbiology solutions for hospitals.
4. Roche
◈Strength: Advanced molecular diagnostics and virology expertise.
◈Focus: Clinical testing and oncology microbiology integration.
5. Thermo Fisher Scientific
◈Strength: Broad microbiology instrument portfolio.
◈Focus: R&D and advanced lab automation.
6. bioMérieux SA
◈Revenue (2024): €4B, with €1.33B from microbiology.
◈Strength: Global in vitro diagnostics leader.
◈Focus: Infectious disease detection and food safety.
7. Intertek Group plc
◈Revenue (2024): £3.39B, Health & Safety up 7.9%.
◈Strength: Assurance, Testing, Inspection, and Certification (ATIC).
◈Focus: Food and clinical microbiology services.
Latest Announcements
◈Jan 2025: RID launched BSIDx device for bloodstream infections (reduces death risk by 8% per hour delay in antibiotic treatment).
◈Feb 2025: Spore.Bio raised €22M Series A for AI-powered microbiological quality control in industries.
◈Feb 2025: Zymo Research announced $5,000 Microbiome Grant to support microbial and metagenomics research.
Recent Developments
◈India (Feb 2024): PM Modi inaugurated food microbiology lab in Kakkanad with FSSAI funding (₹4.5 Cr).
◈India (Apr 2025): Kerala launched advanced microbiology public health lab in Thiruvananthapuram.
◈UAE (Jun 2024): Smart Salem launched AI-powered metagenomic WGS microbiome test.