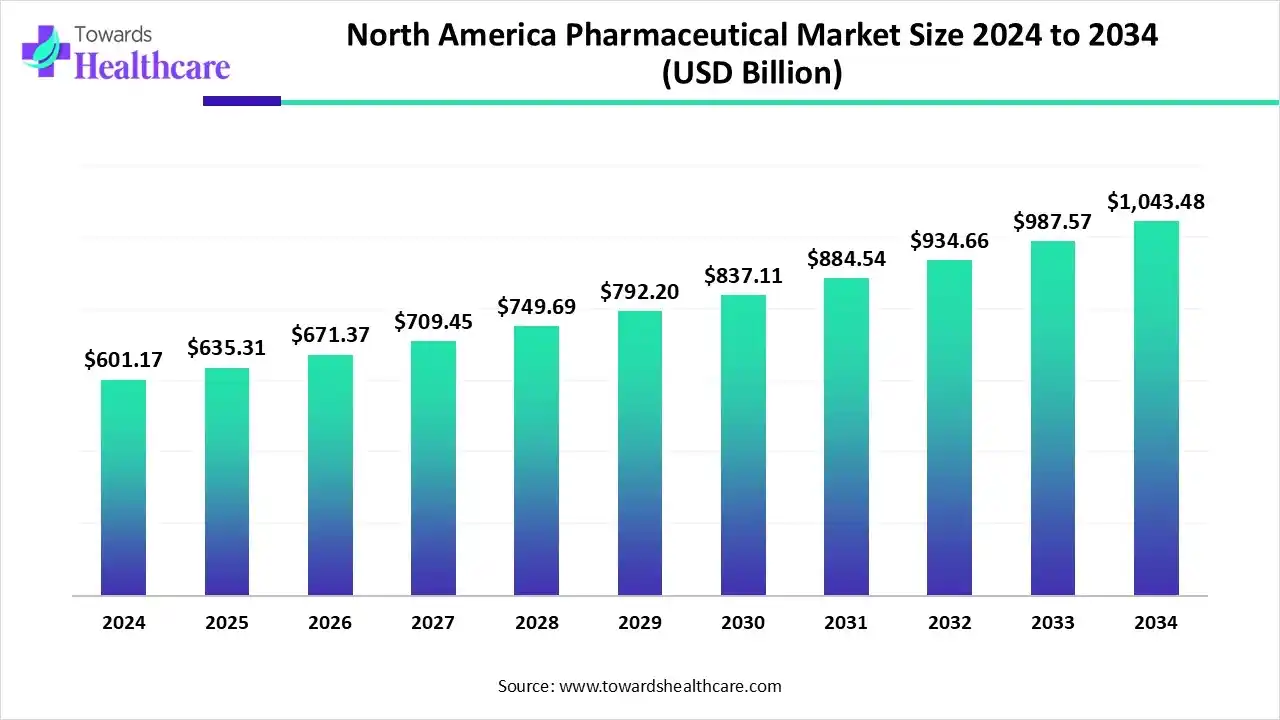
North America pharmaceutical market size reached US$ 601.17 billion in 2024, is US$ 635.31 billion in 2025, and is projected to hit US$ 1,043.48 billion by 2034 at a 5.67% CAGR (2025–2034), led by the U.S. with ~91% share in 2024, supported by aging demographics, chronic-disease burden, specialty/oncology leadership, biologics, gene/cell therapies, and expanding generics/biosimilars.
Download the free sample and get the complete insights and forecasts report on this market @ https://www.towardshealthcare.com/download-sample/6276
Market Size
Level-set
●2024 actual: US$ 601.17 B (baseline year; reflects high specialty mix and oncology strength).
●2025 size: US$ 635.31 B (transition to forecast period; integration of AI, supply-chain resiliency programs).
●2034 projection: US$ 1,043.48 B (5.67% CAGR from 2025).
Growth Drivers Embedded in the Numbers
●Population & disease mix: Rapid rise in elderly population and chronic conditions (diabetes, cardiometabolic, oncology) underpin consistent volume/value growth.
●Modality shift: Biologics, monoclonal antibodies, gene & cell therapies increase average selling prices and therapy duration.
●R&D intensity & approvals: Novel small molecules and advanced therapies sustain launch cadence, lifting peak-year sales curves.
Mix Highlights Behind the Totals
●Product type (2024): Branded small-molecule innovative drugs 34% share; specialty medicines/orphan & advanced therapies slated for the fastest CAGR.
●Therapeutic area (2024): Oncology 28% share; rare/orphan & gene therapies to grow quickest.
●Channel (2024): Retail pharmacies 45%; “other channels” (online, DTx, on-site dispensing) to scale fastest.
●Business model (2024): Innovator sales 62%; specialty pharmacy & hub services to outpace.
●Payer mix (2024): Commercial payers/insurers 48%; “other” category to rise fastest via rare-disease and trial-linked access.
Market Trends
Structural
Specialty tilt accelerates: Oncology, immunology, rare diseases dominate pipelines; CAR-T, bispecifics, therapeutic cancer vaccines scale indications.
Biosimilars & generics expand access: Broaden treatment coverage while reducing system costs; payers push biosimilar adoption.
Personalized medicine: Surge in companion diagnostics and precision dosing across oncology/CNS/metabolic care.
Investment & Industrial Policy
Roche (Apr 2025): US$ 50B planned U.S. spend across diagnostics & pharma (2025–2030) to deepen R&D/manufacturing.
AbbVie (Aug 2025): US$ 195M expansion in U.S. API manufacturing, shoring domestic supply.
Pricing, Access & Payer Behavior
U.S. initiatives (2025): Federal focus on drug price reduction, letters to 17 leading pharma, and supply-chain resilience actions.
Canada pharmacare (2024–2025): National moves toward universal pharmacare and provincial agreements to improve medicine access.
Commercial & Channel
Retail leadership (45% share 2024): Expanded medication management, POCT, vaccination, telepharmacy, remote monitoring.
Omnichannel growth: E-commerce + home delivery uptake; pilots for direct-to-patient complex therapies.
AI: 10 Deep Roles & Impacts
Target discovery & validation: Multi-omics + structure-function modeling to prioritize druggable proteins and de-risk early R&D.
De novo small-molecule design: Generative models optimize binding affinity, ADMET, and novelty, compressing design cycles.
Biologics engineering: Sequence-to-structure prediction for antibody affinity, developability, and immunogenicity risk.
Clinical trial design: Synthetic control arms, adaptive designs, and site selection using EHR/claims accelerate enrollment and power.
Patient stratification: ML-driven biomarker discovery enables precision cohorts and response prediction.
Safety signal detection: Continuous pharmacovigilance via NLP on real-world data to detect rare AEs earlier.
Manufacturing optimization: Predictive maintenance and process analytical technology (PAT) for yield, batch consistency, and cost control.
Supply-chain resilience: AI demand-sensing and multi-sourcing optimization reduce stock-outs; aligns with domestic API initiatives.
Commercial analytics: Next-best-action for payer negotiations, formulary access, and omnichannel HCP engagement.
Patient support & adherence: Intelligent care navigation, side-effect triage, and personalized dosing reminders improve outcomes and payer value.
Regional Insights
United States (91% share, 2024)
Scale & pipeline depth
Oncology leadership (28% market): Broad I/O, cell therapy, targeted agents; frequent label expansions.
Innovator sales (62% 2024): High launch values, robust specialty pharmacy & hubs.
Policy & access
Federal actions (2025): Price reduction focus; efforts to enhance API/Rx supply-chain resilience.
Payer dynamics: Commercial payers (48% 2024) drive utilization management, value-based contracts, and biosimilar conversions.
Industrial base
Capex & reshoring: Roche US$50B, AbbVie US$195M API; emphasis on domestic manufacturing and critical inputs.
Canada
Access expansion
Pharmacare initiatives (2024–2025): National framework and BC agreement to broaden free/affordable medications.
Market character
Heavier public role: Provincial plans shape formulary and pricing; steady uptake of biosimilars and vaccines.
Innovation spillover
Participation in clinical trials, regulatory alignment, and cross-border collaborations in specialty areas.
Market Dynamics
Drivers
Demographics & disease: Aging + chronic diseases expand treated populations.
Advanced modalities: Biologics, gene/cell therapies, and mAbs lift value mix.
Channel evolution: Retail pharmacies (45% 2024) plus omnichannel improve reach and persistence.
R&D investment: Large-scale commitments (e.g., US$50B, US$195M) and steady launch cadence.
Restraints
Rising R&D costs: Late-stage attrition and complex trials pressure ROI.
Regulatory stringency: Extended review times; post-market evidence requirements.
Pricing pressures: U.S. price scrutiny (2025 actions) and payer step-edits; Canada pharmacare cost controls.
Supply vulnerabilities: API dependencies; need for domestic resiliency upgrades.
Opportunities
AI-enabled productivity: Shorten discovery-to-IND timelines; smarter trials; predictive manufacturing.
Biosimilar wave: Cost relief + access expansion; payer alignment.
Rare/orphan & gene therapies: Fastest growth segment; novel hub services and outcomes-based contracts.
Direct-to-patient pilots: New service models in other channels with adherence support and real-world evidence capture.
Top 10 Companies
Pfizer Inc.
Overview: U.S. major with diversified vaccines, oncology, specialty medicines.
Products: Vaccines; anticoagulant Eliquis (co-marketed); oncology brands such as Adcetris, Padcev, Tivdak (commercial portfolio exposure).
Strength: Scale, launch execution, government partnerships; strong oncology and vaccines footprint.
Johnson & Johnson
Overview: Innovative pharma with immunology and oncology depth.
Products: DARZALEX, ERLEADA, CARVYKTI, RYBREVANT (oncology); TREMFYA, SIMPONI, Remicade (immunology legacy).
Strength: First-in-class/ best-in-class biologics, cell therapy entry (CARVYKTI), integrated patient services.
Merck & Co., Inc.
Overview: Vaccine and oncology leader.
Products: Gardasil/Gardasil 9 (HPV); Winrevair, Capvaxive, Enflonsia (approved vaccines/therapies).
Strength: Global vaccine platform, I/O franchise, payer traction.
AbbVie Inc.
Overview: Immunology powerhouse pivoting beyond Humira.
Products: Skyrizi, Rinvoq, Humira legacy.
Strength: Multiple immunology growth drivers; U.S. API capex (US$195M) to enhance supply.
Bristol Myers Squibb
Overview: Oncology, hematology, and cell therapy.
Products: Opdivo Qvantig, Breyanzi, Radiopharmaceuticals, Krazati.
Strength: Deep oncology pipeline, cell therapy scale-up, label expansion engine.
Eli Lilly and Company
Overview: Metabolic and obesity momentum; strong CNS/oncology pipeline.
Products: Advanced diabetes/obesity agents; specialty launches.
Strength: Category leadership in metabolic disease; precision medicine investments.
Amgen Inc.
Overview: Biologics pioneer with oncology, inflammation, bone health.
Products: Oncology and specialty biologics; biosimilar portfolio.
Strength: Manufacturing excellence in biologics; lifecycle maximization.
Gilead Sciences
Overview: Virology core; growing oncology.
Products: Antivirals; oncology entries via targeted and cell therapies.
Strength: Cash-generative virology, oncology diversification.
Novartis AG
Overview: Specialty innovator with oncology and gene therapy exposure.
Products: Targeted oncology; advanced therapies.
Strength: Precision medicine, global trial network, gene therapy know-how.
Roche
Overview: Integrated pharma-diagnostics.
Products: Oncology biologics; diagnostics tie-ins for companion tests.
Strength: US$50B U.S. investment plan (2025–2030); CDx leadership enabling personalized care.
(Also material players: Sanofi, AstraZeneca, Novo Nordisk, Biogen, Regeneron, Vertex, Moderna, Takeda, Bayer, Teva.)
Latest Announcements
Pfizer (Sep 2025): Agreement with U.S. government and Administration aimed at reducing drug costs for American patients; supports affordability narrative and payer alignment.
Sanofi (Sep 2025): US$ 625M added to Sanofi Ventures to expand biotech and digital health investments, signaling continued external innovation sourcing.
Recent Developments
Federal U.S. actions (Jul–Aug 2025): Initiatives to reduce drug prices, letters to 17 pharma leaders, and supply-chain resilience measures for APIs.
Roche (Apr 2025): US$ 50B U.S. diagnostics & pharmaceuticals investment commitment (five-year horizon).
AbbVie (Aug 2025): US$ 195M U.S. API manufacturing expansion.
Canada (Oct 2024 & Mar 2025): Legislation and pharmacare agreement with British Columbia for universal/free medication access in priority areas.
Segments Covered
By Product Type
Branded Small-Molecule Innovative Drugs (34% in 2024)
Clinical/Regulatory Mechanics
●Rapid approvals supported by well-defined endpoints and mature regulatory pathways (priority review, breakthrough tags).
●Lifecycle management via line extensions, fixed-dose combinations, and pediatric studies sustains share.
Economic Profile
●Lower COGS than biologics; attractive gross margins; pricing influenced by step-therapy and generic erosion timing.
●Patent cliffs still material—curve management relies on next-gen MoAs and indication expansions.
Market Role
●Backbone in cardiometabolic, CNS, oncology adjuncts; oral convenience drives adherence and retail channel pull-through.
2025–2034 Outlook
●Continues as high-volume anchor while specialty biologics take value mix; AI-driven design compresses early-cycle timelines.
Biologics & Monoclonal Antibodies
Clinical/Science
●High specificity; CDx-linked uptake in oncology/immunology; subcutaneous and on-body delivery improving persistence.
Operations
●Complex manufacturing (cell culture, purification) with capacity constraints; robust QA/QC and cold-chain needs.
Payer/Access
●Prior authorization common; outcomes evidence critical for renewals; site-of-care shifts (home/ambulatory) to manage cost.
2025–2034 Outlook
●Sustained growth on oncology, immunology, rare indications; ongoing shift to long-acting and self-administered formats.
Generics (Small Molecules)
Role in System
●Primary cost-containment lever enabling payer switches; high substitution rates post-exclusivity.
Market Mechanics
●Price compression through multi-sourcing; supply resilience programs emphasize domestic/API capacity.
2025–2034 Outlook
●Stable to expanding volumes; margins thin but scale players benefit from portfolio breadth and reliability.
Specialty Medicines / Orphan & Advanced Therapies (Cell & Gene) — Fastest Growth
Clinical/Value
●High clinical value in ultra-rare, oncology, hematology; curative intent in select gene/cell programs.
Commercial Model
Hub services, patient onboarding, and real-world outcomes tracking essential; emerging outcomes-based contracts.
Operational
●Personalized manufacturing, vein-to-vein logistics, and stringent chain-of-identity.
2025–2034 Outlook
●Pipeline depth + broader label expansions; payers adopt novel payment models to manage upfront cost.
Vaccines
Market Structure
●Demand stabilized by public procurement and immunization schedules; adult immunization broadening.
Operations
●Biologic manufacturing, seasonal planning, and strategic stockpiles; retailer administration expands access.
2025–2034 Outlook
●Steady growth from respiratory, oncology-prevention, and older-adult programs; combo and adjuvanted platforms scale.
OTC Medicines
Consumer Behavior
●Self-care momentum; retail & e-commerce dominate discovery and fulfillment; brand trust is key.
Regulatory/Market
●Rx-to-OTC switches can unlock step-change demand; private label pressures price.
2025–2034 Outlook
●Incremental growth via digital engagement, subscription models, and pharmacist-led services.
Biosimilars
Access & Economics
Price corridor compression vs. reference biologics; payer-driven adoption with interchangeability policies.
Market Mechanics
●Physician confidence rising with real-world evidence; tendering and formulary tiering steer share.
2025–2034 Outlook
●Broadening waves in oncology and immunology reduce system spend and reallocate budget toward advanced therapies.
By Therapeutic Area
Oncology (28% in 2024) — Dominant
Science/Modalities
I/O, CAR-T, bispecifics, targeted agents; combo regimens increase duration and depth of response.
Commercial
●Companion diagnostics gate access; treatment pathways align to value-based outcomes.
2025–2034 Outlook
●Continued label expansions; rise of tumor-agnostic indications and minimal residual disease (MRD) monitoring.
Cardiovascular & Metabolic
Growth Engine
Diabetes/obesity innovations drive category expansion; cardioprotective profiles expand eligible populations.
Market
●High primary-care footprint; adherence programs essential for outcomes and payer alignment.
CNS (Neurology/Psychiatry)
Innovation
●Precision sub-segments (neurodegeneration, rare epilepsies); digital endpoints and wearables enrich evidence.
Access
●Prior auth and step therapy typical; caregiver support impacts persistence.
Autoimmune/Immunology
Therapy Mix
Biologics & JAKs with differentiated safety/efficacy; switching dynamics with biosimilars intensify.
Care Setting
●Shift from infusion centers to at-home SC administration where feasible.
Infectious Diseases
Profile
●Vaccines/antivirals; preparedness cycles influence demand; stewardship affects antibiotic economics.
Respiratory & Allergies
Trend
●Biologics for severe asthma; device usability and adherence tech (smart inhalers) matter.
Rare/Orphan & Gene Therapies — Fastest Growth
Market
●Small populations, high value per patient; registries and long-term follow-up integral.
Payer
Installment/outcomes contracts increasingly utilized.
Others (Dermatology, GI, Ophthalmology, etc.)
Pattern
●Targeted launches with strong QoL impact; procedure-drug combos and sustained-release devices gain traction.
By Channel / Distribution
Retail Pharmacies (45% in 2024)
Scope Expansion
POCT, vaccinations, medication management, telepharmacy; adherence programs reduce discontinuations.
Digital
●Omnichannel, refill sync, home delivery; pharmacists as front-line navigators for chronic therapy.
Hospital / Health System
Use Case
●Complex infusions, acute starts, and pathway-driven oncology care; 340B-like dynamics (where applicable) influence mix.
Integration
●EMR-driven order sets and stewardship programs standardize use.
Specialty Pharmacies
Capabilities
●High-touch onboarding, benefits investigation, prior auth, REMS support, cold-chain logistics.
Value
●Generates real-world outcomes and persistence data for payer renewals.
Mail-Order / LTC / Institutional
Strength
●Chronic therapy continuity, synchronized dispensing, and cost-efficient logistics for stable regimens.
Other Channels (Fastest Growth)
Models
Online/DTx, employer on-site clinics, direct-to-patient pilots for select therapies.
Why It Scales
●Convenience + data capture enabling proactive adherence and pharmacovigilance.
By Business Model / Revenue Stream
Innovator Sales (62% in 2024)
Engine
●High launch values in oncology, obesity, diabetes, CNS, rare; patient-centric and omnichannel HCP engagement.
Risk
●Patent cliffs offset by next-wave launches and indication breadth.
CMO/CDMO
Role
●Flexible capacity for biologics/advanced modalities; tech-transfer expertise; resilience for domestic manufacturing.
Trend
●Strategic partnerships and co-investment to secure long-lead assets.
Generics & Volume
Economics
●Scale manufacturing; tenders and preferred positioning; quality/reliability key amid supply scrutiny.
Specialty Pharmacy & Hub Services (Fastest)
Capabilities
●Access orchestration, adherence, outcomes tracking; critical for cell/gene and high-touch biologics.
Vaccine & Public-Health Procurement
Stability
●Policy-linked multi-year demand; supports capacity planning and stockpiling.
By Buyer / Payer Type
Commercial Payers/Insurers (48% in 2024)
Tools
●Utilization management, prior auth, step edits, VBAs; aggressive biosimilar steering.
Evidence Needs
●Real-world outcomes and budget-impact models for renewals and tier placement.
Government/Public (Medicare, Medicaid; Provincial Plans)
Focus
●Affordability and equitable access; reference pricing and negotiated rates shape adoption.
Out-of-Pocket
Dynamics
●Cost burden in select categories; copay assistance and cards mitigate abandonment.
Private Employers/PBMs
Levers
●Plan design, formulary exclusions, center-of-excellence routing for high-cost therapies.
Other (Philanthropic, Trial Sponsors)
Use Cases
●Bridge access in rare diseases; coverage during trial/transition phases; standards for QA in procurement.
By Region
United States
Scale & Mix
●91% regional share (2024); largest pipeline and launch cadence; innovator sales 62% underpin value mix.
Policy & Market Shaping
●2025 federal focus on drug pricing and API supply resilience impacts contracting and sourcing.
Provider/Payer Behavior
●High specialty penetration (oncology, immunology); biosimilar conversions accelerate; VBAs more common.
Distribution
●Retail leadership, specialty hubs for complex therapies, and growth in direct-to-patient pilots.
Canada
Access Architecture
●Pharmacare steps (2024–2025) broaden essential medicines access; strong role for public procurement.
Adoption Patterns
Biosimilars advancing due to policy alignment; steady vaccine uptake.
Collaboration
●Participation in regional trials and regulatory harmonization to speed specialty adoption.
Cross-Segment KPIs & Execution Checklists
Access & Pricing
●Time-to-formulary post-approval, tier position, and prior-auth burden by plan.
Biosimilar adoption curves and net price evolution in protected classes.
Clinical & Evidence
●Companion diagnostic availability, post-market RWE, and label expansion cadence.
Operations
●Fill-rate, cold-chain integrity, site-of-care shifts, and hub case throughput.
Patient Outcomes
●Persistence at 3/6/12 months, PROs, and adherence interventions ROI.
Top 5 FAQs
1) What is the market size and growth outlook?
US$ 601.17B (2024) → US$ 635.31B (2025) → US$ 1,043.48B (2034) at 5.67% CAGR (2025–2034).
2) Which country leads the region?
United States with ~91% share (2024) driven by specialty/oncology, payer scale, and R&D intensity.
3) Which product segments matter most?
2024 leader: Branded small-molecule innovative drugs (34%).
Fastest growth: Specialty/orphan & advanced (cell & gene); biosimilars expanding access.
4) Which therapy areas dominate and grow fastest?
Dominant: Oncology (28% 2024).
Fastest growth: Rare/orphan & gene therapies (CRISPR/gene therapy approvals, R&D funding).
5) What policies and investments shape the next decade?
U.S.: 2025 federal actions on pricing and API resilience; large industrial commitments (US$50B, US$195M).
Canada: Universal pharmacare steps (2024–2025) broadening access.
Access our exclusive, data-rich dashboard dedicated to the pharmaceuticals sector – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Immediate Delivery Available | Buy This Premium Research @ https://www.towardshealthcare.com/checkout/6276
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Web: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest