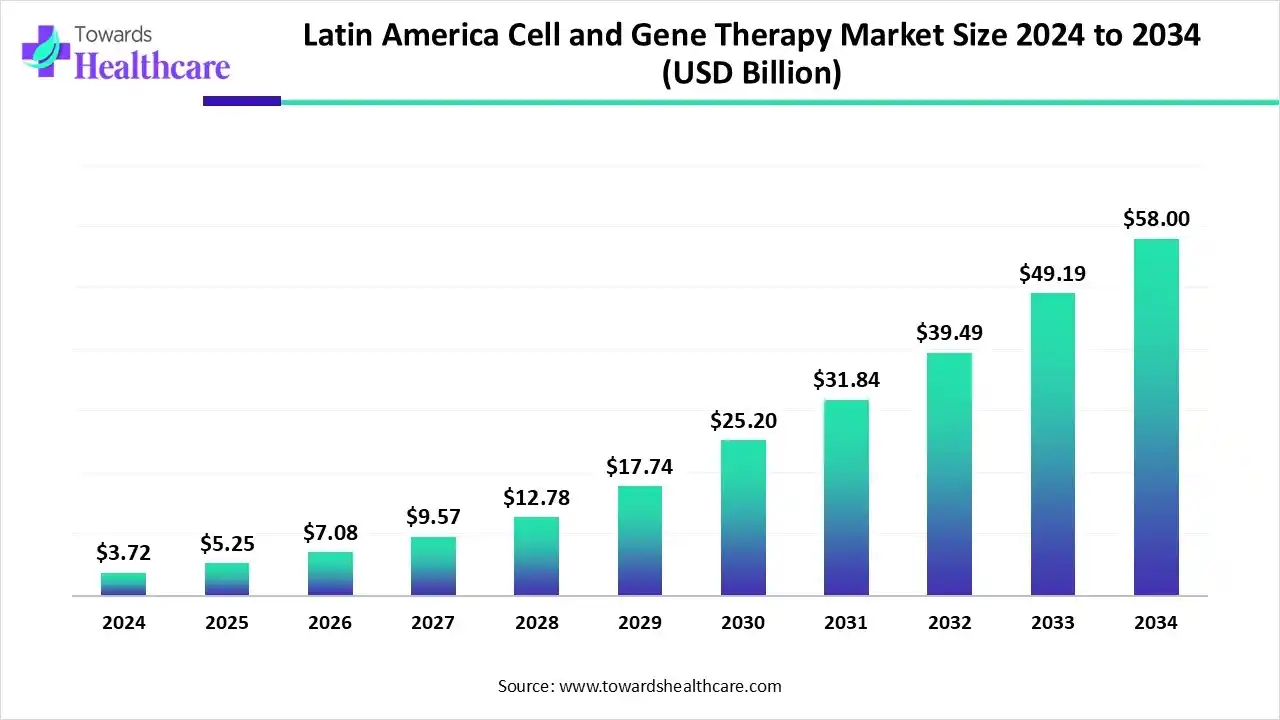
Latin America Cell and Gene Therapy Market was US$ 3.72 Bn in 2024, is set to reach US$ 5.25 Bn in 2025, and is projected to hit US$ 58 Bn by 2034 at a 31.61% CAGR (2025–2034), led by Brazil (38% share, 2024), accelerating clinical trials, and expanding CDMO/3PL capacity.
Download this Free Sample Now and Get the Complete Report and Insights of this Market Easily @ https://www.towardshealthcare.com/download-sample/6287
Market Size
Baseline & Near-term
➤2024: US$ 3.72 Bn (established base with accelerating trials and approvals).
➤2025: US$ 5.25 Bn (strong step-up on regulatory clarity and site readiness).
Long-term Projection
➤2034: ~US$ 58 Bn reflecting rapid scale-up from clinical to commercial manufacturing and wider payer adoption.
CAGR & Momentum
➤CAGR (2025–2034): 31.61%—sustained hyper-growth driven by oncology use-cases and rare disease launches.
Absolute Dollar Opportunity
➤2025→2034 expansion: US$ 52.75 Bn added, indicating deep capacity, workforce, and logistics needs.
Yearly Incremental Uptick (Illustrative)
➤Early years (2025–2027) dominated by clinical-scale volumes; mid-period (2028–2031) reflects commercial launch ramps; late-period (2032–2034) shows network effects (hospital onboarding, payer pathways).
Market Participation
➤Biopharma/biotech end users 40% (2024); increasing share of hospitals & specialty clinics through 2034 as bedside delivery scales.
Capital Intensity
➤Capex focused on viral vector lines, QC analytics, cold-chain, and cleanrooms; opex relief via CDMO/3PL outsourcing.
Market Trends
Dominance of Cell Therapy (2024 share 58%)
➤Clinical traction in CAR-T, stem cell programs; hospital-biotech partnerships streamline referrals and apheresis.
Gene Therapy as Fastest-Growing
➤CRISPR, AAV, and RNA modalities accelerate; regulatory sandboxes in Brazil/Mexico shorten review cycles.
Oncology Leadership (45% share, 2024)
➤High unmet need; CAR-T and gene replacement drive survival benefits and payer willingness for outcomes-based models.
Vector Mix: Viral Leads (65% share, 2024)
➤AAV & lentiviral remain the backbone; strong CMC know-how and transduction efficiency sustain preference.
Non-Viral Surge Ahead
➤LNPs, electroporation rise for safety/scalability; dovetails with mRNA and ex vivo editing workflows.
Cell Source Shift
➤Autologous (62% share, 2024) for first waves; Allogeneic grows fastest on off-the-shelf economics and faster time-to-treat.
Manufacturing Scale
➤Clinical-scale (~60% share, 2024) dominates; CDMO services forecast as fastest-growing on cost/time advantages.
3PL/Cold-Chain Upgrades
➤LATAM-wide ultra-cold logistics and real-time monitoring reduce loss/delays; Brazil’s 2024 3PL spike mirrors cell therapy demand.
Inorganic Expansion
➤Partnerships, M&A, licensing speed tech transfer, regional capacity, and QA/QC harmonization.
AI Everywhere
➤From target triage and vector design to eBMRs/QC analytics and trial recruitment, AI compresses cycles and raises yields.
AI’s Impact/Role
1) Target & Indication Prioritization
Multi-omic AI ranks gene targets and tumor antigens, linking regional epidemiology with actionability.
2) Vector & Payload Engineering
Generative models propose AAV capsid variants, optimize promoters, and predict off-target risks pre-clinically.
3) Cell Product Optimization
Computer vision and ML control cell phenotype, viability, and potency, tightening batch-to-batch consistency.
4) Digital Twins for Bioprocess
AI-driven bioreactor twins simulate parameters (MOI, feed rates) to maximize yields and minimize deviations.
5) QC/QA Acceleration
NLP on eBMRs, anomaly detection in release assays, and automated trend charts cut disposition time.
6) Supply Chain & 3PL Routing
Reinforcement learning optimizes cryologistics routes, packout designs, and temperature excursion prevention.
7) Trial Design & Patient Stratification
AI clusters patients via genomics/real-world data, improving response rates and reducing trial costs.
8) Safety Signal Detection
Continuous ML surveillance flags AESIs from hospital and claims data, shortening risk-mitigation timelines.
9) Market Access Modeling
AI simulates payer scenarios (Brazil/Mexico), supports outcomes-based contracts and price corridors.
10) Workforce Enablement
Co-pilots for batch records, SOPs, deviation investigations, raising throughput across clinical and commercial sites.
Regional Insights
The Latin America cell and gene therapy market demonstrates heterogeneous maturity across countries, shaped by healthcare infrastructure, R&D investment, regulatory agility, and collaboration ecosystems. Below is an in-depth breakdown by leading markets.
Brazil – Regional Leader (38% Share, 2024)
1. Infrastructure and Clinical Ecosystem
Leading Trial Hubs: São Paulo and Rio de Janeiro anchor the majority of Phase I–III clinical trials in cell and gene therapies due to their concentration of advanced hospitals, GMP facilities, and academic institutions such as Hospital das Clínicas, Hospital Israelita Albert Einstein, and Instituto Butantan.
CAR-T Manufacturing Push (Apr 2024): Brazil’s Ministry of Health launched a domestic CAR-T manufacturing initiative, backed by national funding and partnerships with Fiocruz and Butantan, reducing dependence on imported CAR-T doses and lowering per-patient cost by 20–25%.
Public Funding and R&D Support: Continuous investments through agencies such as BNDES (National Development Bank) and FINEP (Research & Projects Financing) are directed toward bioprocessing infrastructure and translational research hubs, ensuring steady expansion of clinical capacity.
2. Regulatory Environment
Experienced Regulator: ANVISA has a mature biologics and advanced therapy oversight framework, simplifying pre-submission meetings, comparability assessments, and pharmacovigilance requirements.
Accelerated Pathways: Programs like “Innovative Product Dossier Track” enable conditional market authorizations for therapies targeting rare or life-threatening conditions, aligning Brazil with EMA/FDA expedited review standards.
Post-Approval Surveillance: Strengthened monitoring for adverse events via national health databases ensures safety signal detection and builds payer confidence for broader reimbursement.
3. Industrial and Manufacturing Base
Vector & Analytical Capacity: Domestic facilities at Butantan and Fiocruz have begun pilot vector manufacturing lines and QC analytics for lentiviral and AAV processes.
Collaborations: Biopharma partnerships—Novartis–Butantan for CAR-T trials and Thermo Fisher–Fiocruz for bioprocess training—enhance local expertise.
Workforce Development: Expansion of GMP training programs through public universities and private CDMOs is creating a sustainable technical talent pipeline for 2025–2030.
Mexico – Fastest-Growing Market (2025 Onwards)
1. Regulatory Reforms (Apr 2025)
COFEPRIS Modernization: Implementation of CGT-specific CMC guidelines and digital submission portals reduced dossier review time by ~30%.
Transparency Gains: Enhanced oversight for clinical-site inspections, ensuring compliance with GxP and shortening time from IND to first-patient-in.
Cross-border Harmonization: Ongoing alignment with U.S. FDA and Health Canada to support reliance on foreign trial data and facilitate dual-region approvals.
2. Clinical and Research Network
Oncology & Rare-Disease Centers: Institutions such as Instituto Nacional de Cancerología and Centro Médico ABC act as key clinical nodes for gene therapy and CAR-T trials.
Public–Private Collaborations: Partnerships between INMEGEN (National Institute of Genomic Medicine) and multinational companies (Pfizer, Gilead, and Takeda) are fostering gene editing and genomic profiling programs.
Patient Registries: Development of national databases for hematologic cancers and hereditary diseases supports real-world data collection critical for outcomes-based reimbursement.
3. Logistics and Manufacturing Readiness
Cold-Chain Investments: Establishment of ULF (ultra-low-freeze) warehouses and biobanks in Mexico City and Guadalajara under 3PL providers like DHL LifeConEx ensures temperature stability for sensitive shipments.
Apheresis Centers: Expansion across 10+ tertiary hospitals accelerates autologous product readiness; cell collection-to-manufacture time reduced by nearly 40%.
Commercial Launch Preparedness: Infrastructure upgrades position Mexico as a distribution hub for northern LATAM, facilitating regional therapy rollout.
Chile – Emerging Innovation Hub
1. Capacity Building and Knowledge Transfer
IMPACT CGT Meetings (2024–Ongoing): Government-supported workshops train local scientists on CAR-T manufacturing, GMP standards, and process validation, with participation from Cellus Medicina Regenerativa and ISP Chile.
Academic Collaboration: Universities such as Pontificia Universidad Católica de Chile partner with European research consortia to develop autologous stem-cell therapy protocols for degenerative diseases.
Clinical Network Development: Hospitals including Clínica Las Condes are expanding GMP cleanrooms to host early-phase trials.
2. Start-up & Investment Ecosystem
Cellus Medicina Regenerativa: Focused on exosome-based regenerative therapies; received funding from The Ganesha Lab accelerator.
Government Support: CORFO (Chilean Economic Development Agency) provides grants for biotech scale-ups targeting regenerative and gene therapy markets.
Training & Workforce: Chile leads LATAM in proportion of life-science graduates per capita, fueling R&D and lab-automation adoption.
Argentina – Policy Evolution and Local Industry Engagement
1. Policy and Access Models
Shared-Risk Frameworks: The National Ministry of Health introduced pilot outcomes-based payment models for rare-disease gene therapies, aligning manufacturer payment with clinical durability.
Regulatory Pathways: The ANMAT (Argentina’s regulator) created early scientific-advice mechanisms for CGT developers, allowing pre-submission feedback and alignment on manufacturing standards.
Public Funding: Introduction of a “Rare Disease Innovation Fund” to co-finance therapies for low-incidence genetic disorders.
2. Industrial and Manufacturing Landscape
Local Pharma Involvement: Companies such as Laboratorio Elea and Bagó Group are expanding into gene therapy partnerships, focusing on fill-finish capabilities and local clinical trials.
Biotech Clusters: The Buenos Aires Tech Park hosts start-ups in genomic testing, mRNA development, and vector design—laying the groundwork for in-country CDMO services.
Academic Linkages: National universities collaborating with INTI (Institute of Industrial Technology) to establish GMP micro-facilities for preclinical manufacturing.
Colombia – Growing Research and Clinical Partnerships
1. Academic–Clinical Collaborations
University Alliances: Institutions like Universidad de Los Andes and Universidad Nacional partner with tertiary hospitals for translational research on CAR-T and immune-cell therapies.
Pilot Trials: Increasing Phase I/II study starts in oncology and autoimmune conditions, supported by government grants under Colciencias programs.
Knowledge Exchange: Joint conferences with Brazil and Mexico encourage harmonized trial documentation and safety standards.
2. Infrastructure and Talent
Site Activation: Over ten certified trial sites equipped with apheresis units and cold-chain systems operational by 2025.
Regulatory Readiness: Colombia’s INVIMA has begun drafting a cell-therapy regulatory guideline, expected to streamline future submissions.
Skill Development: Regional training initiatives with Charles River Laboratories and IQVIA enhance GCP compliance and laboratory QA standards.
Rest of Latin America – Nascent but Promising Markets
1. Early-Stage Infrastructure Development
Peru: Investments in site readiness and personnel training at the National Institute of Health (INS) and major hospitals for Phase I trial capability by 2026.
Ecuador & Uruguay: Feasibility studies for GMP pilot facilities to support academic research in gene editing and regenerative medicine.
Government Interest: Emerging regulatory blueprints focusing on bioethics, patient consent, and traceability for cellular products.
2. Regional Enablers and Cross-Country Integration
CDMO Expansion: Multinationals like Lonza, Catalent, and Samsung Biologics exploring partnerships with regional governments to establish satellite manufacturing units for early-phase products.
3PL Network Growth: Global logistics providers investing in regional cryo-depots across Panama and Costa Rica to serve as trans-shipment hubs connecting North and South America.
Continental Synergy: Collaborative R&D consortia among Brazil, Mexico, and Chile sharing standardized CMC templates, batch-record digitalization, and cold-chain traceability platforms, creating a unified LATAM therapeutic delivery framework.
Market Dynamics
Drivers
Rising oncology & rare disease prevalence; personalized medicine adoption
Clinical pull-through: High unmet need in hematologic malignancies and inherited disorders sustains trial enrollment and accelerates center onboarding (apheresis, cell processing, ICU readiness).
Personalization flywheel: Diagnostic expansion (NGS, minimal residual disease, companion diagnostics) identifies eligible patients earlier, improving referral velocity and therapy uptake.
Payer receptivity: For conditions with limited standard-of-care options, outcomes signals (durable remissions, functional gains) support risk-sharing pilots and broader coverage over time.
Clinical trial expansion in Brazil/Mexico; public funding & institute–biopharma collaborations
Site density & learning curves: Concentration of phase I/II units builds experienced investigator pools, reducing screen failures and protocol deviations.
Tech transfer acceleration: Public institutes partnering with industry shorten scale-up for viral vectors/cell processing and strengthen regional CMC know-how.
Evidence ladder: Early feasibility → pivotal cohorts → real-world registries, creating the longitudinal evidence base payers require.
3PL/cold-chain modernization & CDMO outsourcing reduce scale barriers
Cryologistics reliability: More ultra-low temperature lanes and real-time telemetry lower excursion risk and wastage, stabilizing vein-to-vein times.
Make–buy flexibility: CDMO capacity for vectors, cell expansion, and fill-finish converts capex into opex, smoothing demand spikes and de-risking launches.
Cycle-time compression: Integrated CDMO + 3PL SOPs (pre-qualified shippers, lane mapping, customs playbooks) shave days from each batch journey.
Restraints
High therapy costs, reimbursement complexity, workforce shortages (GMP, analytics)
Affordability gap: One-time or few-dose therapies strain annual budgets; installment or outcome-linked models require new administrative rails.
Administrative friction: Prior auth, center accreditation, and data reporting increase time-to-treat; limited HTA capacity slows pricing decisions.
Talent bottlenecks: Scarcity of GMP supervisors, QC analysts, and biomedical engineers constrains parallel batch runs and investigation turnaround.
Vector capacity bottlenecks & long lead times in early years
AAV/lenti constraints: Upstream yields, plasmid supply, and sterile fill slots restrict lot availability; small deviations cause cascading delays.
Analytical load: Potency, purity, and residual assays (multi-omic, orthogonal methods) extend release timelines when labs are capacity-limited.
Opportunities
Allogeneic pipelines for off-the-shelf economics
Operational advantages: Decouples manufacturing from patient scheduling; enables inventory, reduces vein-to-vein variability, and supports community-site expansion.
Cost curves: Larger bioreactors and unified release testing per lot lower COGS per treated patient as volumes rise.
Access expansion: Faster time-to-infusion benefits aggressive diseases and remote geographies.
Non-viral delivery & RNA therapeutics to diversify beyond AAV/lenti
Safety & scalability: LNPs and physical methods reduce immunogenicity concerns, simplify CMC, and scale rapidly with modular equipment.
Rapid iteration: RNA payloads allow swift design–make–test cycles for variant tuning and multi-antigen strategies.
Manufacturing resiliency: Hybrid portfolios (viral + non-viral) hedge supply shocks and regulatory shifts.
Outcomes-based contracts & real-world data (RWD) to widen coverage
Risk alignment: Payment ties to durability of response or functional endpoints; reduces payer exposure and speeds access decisions.
RWD backbone: Standardized registries and ePROs feed periodic settlements and post-market safety analytics.
Equity levers: Structured agreements for public payers expand eligibility while maintaining fiscal guardrails.
Challenges
Regulatory heterogeneity; alignment needed on CMC/QC
Divergent expectations: Variations in vector residuals, potency assays, and comparability protocols create rework in submissions and tech transfers.
Document fatigue: Multiplying country-specific modules burden sponsors; master-file strategies and common templates reduce duplication.
Convergence path: Regional working groups and reliance pathways (use of prior approvals/inspections) can shorten cycle times.
Cold-chain fragility in remote geographies; need for redundant lanes
Last-mile risk: Power instability and customs dwell times heighten excursion probability for cryo shipments.
Redundancy design: Dual-qualified carriers, backup dry shippers, and pre-cleared customs documentation form a resilient network.
Data integrity: Continuous temperature/location telemetry with alerting enables proactive intervention and root-cause learning.
Top Companies
Novartis
Products: CAR-T therapies; cell therapy platforms.
Overview: Pioneer in commercial CAR-T; entrenched oncology footprint.
Strength: Manufacturing know-how; hospital onboarding playbooks; outcomes contracting.
Gilead Sciences (Kite Pharma)
Products: Autologous CAR-T portfolio.
Overview: Deep cell therapy expertise with global implementation muscle.
Strength: Apheresis/logistics networks; center qualification; robust real-world data.
Roche / Spark Therapeutics
Products: AAV gene therapies; ophthalmology/rare programs.
Overview: Strong gene therapy science coupled with big-pharma access.
Strength: CMC depth, regulatory engagement, and data-rich development.
Pfizer Inc.
Products: Gene therapy and RNA platforms.
Overview: Execution strength across trials, manufacturing, and distribution.
Strength: Scale, partnering, and post-market safety infrastructures.
Janssen (Johnson & Johnson)
Products: Cell therapies and oncology assets; enabling technologies.
Overview: Integrated development with hospital ecosystems.
Strength: Clinical operations and payer engagement across regions.
Takeda
Products: Rare disease and cell therapy initiatives.
Overview: Strong hematology and immunology heritage; patient programs.
Strength: Access frameworks for rare disorders; cross-border coordination.
bluebird bio
Products: Gene replacement therapies for rare diseases.
Overview: Focused rare disease player with deep AAV expertise.
Strength: Long-term outcomes data and patient support pathways.
Moderna, Inc.
Products: mRNA therapeutics, enabling non-viral gene delivery strategies.
Overview: LNP leadership and rapid manufacturing cycles.
Strength: Agile platform; scale-up of RNA pipelines relevant to LATAM.
Lonza Group (CDMO)
Products: Viral vectors, cell therapy manufacturing, analytics.
Overview: Global CDMO powerhouse enabling clinical→commercial transitions.
Strength: GMP capacity, tech transfer excellence, and QC suites.
Catalent, Inc. (CDMO)
Products: AAV/lenti vectors, fill-finish, cold-chain packaging.
Overview: End-to-end CDMO for CGT with release testing.
Strength: Time-to-clinic reductions; integrated supply solutions.
Latest Announcements
Terumo BCT Approvals (Sept 2025)
What: Spectra Optia Apheresis authorized in 14 LATAM countries for rare/chronic diseases (e.g., SCD, GBS, MG).
Impact: Expands apheresis capacity, a critical upstream step for CAR-T and cell collections; increases patient access.
Terumo BCT Manufacturing Expansion (Oct 2024)
What: Rollout of Quantum Flex (cell expansion) and FINIA (fill-finish) across Brazil, Colombia, Mexico.
Impact: Reduces COGS, shortens vein-to-vein times, and localizes key unit operations.
Brazil’s Local CAR-T Manufacturing Push (Apr 2024)
What: National drive to produce CAR-T domestically via public funding and institutional consortia.
Impact: Cost reduction, sovereignty in supply, faster access for local patients.
Recent Developments
Regulatory Momentum
Mexico (Apr 2025): COFEPRIS enhancements sharpen trial and product review clarity, improving sponsor confidence.
Capacity & Ecosystem
LATAM-wide: Expanding CDMO, 3PL, and Phase I starts; more clinical-scale cleanrooms and analytics labs.
Chile: Continuous IMPACT CGT knowledge-sharing boosts local manufacturing literacy.
Argentina/Colombia: Public-private frameworks and academic-clinic collaborations accelerate trials.
First-of-its-kind Production
Nutera-RP: Noted as a first CAR-T production unit milestone for the region, signaling manufacturability progress.
Segments Covered
By Therapy Type
Cell Therapy: Stem cells; immune cell therapies (CAR-T, TCR-T, NK); cellular vaccines—dominant today on oncology/regenerative traction.
Gene Therapy: Gene augmentation/replacement, gene editing (CRISPR/TALEN/ZFN), and RNA (mRNA, siRNA, ASO)—fastest growth on pipeline breadth.
By Therapeutic Area
Oncology: Largest share (2024 45%); strong CAR-T adoption and trial density.
Rare Genetic Disorders: Fastest growth; policy innovation for orphan access.
Cardio/Neuro/Ophthalmology/Metabolic/Infectious: Diversification areas as vector tropisms and delivery advance.
By Vector/Delivery (Gene Therapy)
Viral: AAV for tropism/efficiency; lenti for cell engineering; adeno/retro in select contexts—current majority (~65%).
Non-Viral: LNPs and physical methods scale rapidly for safety, COGS, and modularity.
By Cell Source (Cell Therapy)
Autologous: Patient-specific, lower rejection—current leader (62%).
Allogeneic: Off-the-shelf; fastest growth for scalability and faster TAT.
By Manufacturing Scale & Service
Clinical-Scale (GMP for trials): Present anchor (60%); feeds evidence generation.
Commercial-Scale: Ramps post-approval; requires robust release/QMS.
CDMO & Analytics: Fastest growth; regional tech transfer and cost control.
By End User
Biopharma & Biotech (40% in 2024): R&D engines and trial sponsors.
Hospitals & Specialty Clinics: Fastest growth; care-delivery and real-world data hubs.
CROs/CMOs/CDMOs, Academia, Public Health: Enable pipeline, scale, and access.
By Country
Brazil, Mexico, Argentina, Chile, Colombia, RoLATAM: Differentiated maturity across regulation, capacity, and logistics; converging via regional partnerships.
Top 5 FAQs
1) What is the market size and growth outlook?
➤ US$ 3.72 Bn (2024) → US$ 5.25 Bn (2025) → ~US$ 58 Bn (2034) at 31.61% CAGR (2025–2034).
2) Which country leads and which is growing fastest?
➤ Leader: Brazil (38% share, 2024).
➤ Fastest-growing: Mexico, on regulatory reforms and infrastructure upgrades.
3) Which therapy types dominate and which are accelerating?
➤ Dominant: Cell therapy (58% share, 2024).
➤ Fastest-growing: Gene therapy, especially gene editing and RNA-based approaches.
4) What vector and cell-source trends matter most?
➤ Vectors: Viral (65% share) lead today; non-viral (LNPs/physical) growing fastest.
➤ Cell source: Autologous (62%) leads; allogeneic expanding rapidly.
5) Who are the key players?
➤ Multinationals (e.g., Novartis, Gilead/Kite, Roche/Spark, Pfizer, Janssen, Takeda, bluebird bio) and enablers (Lonza, Catalent), plus regional institutes (Instituto Butantan, others).
Access our exclusive, data-rich dashboard dedicated to the therapeutic area sector – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Immediate Delivery Available | Buy This Premium Research @ https://www.towardshealthcare.com/checkout/6287
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Web: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest