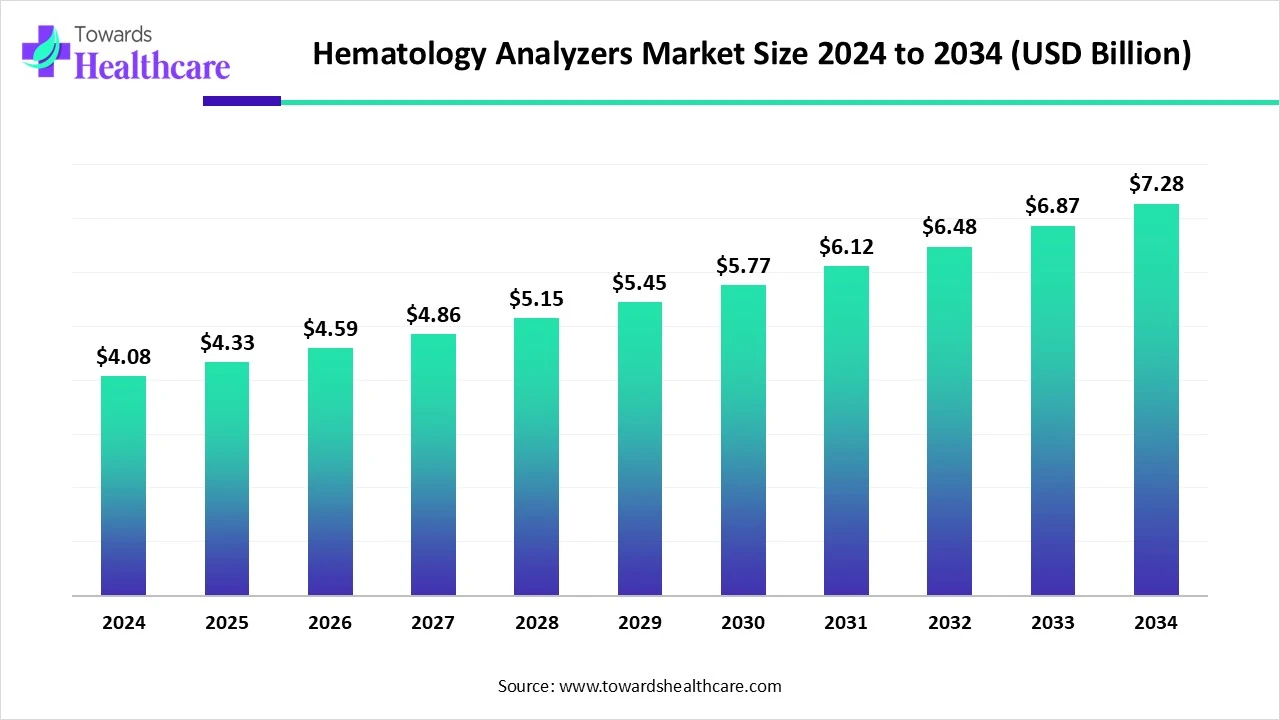
The global hematology analyzers market was US$ 4.08 billion in 2024, is US$ 4.33 billion in 2025, and is projected to reach US$ 7.28 billion by 2034 at a 5.97% CAGR (2025–2034), led by North America (41% share, 2024) and fastest growth in Asia Pacific.
Download the free sample and get the complete insights and forecasts report on this market @ https://www.towardshealthcare.com/download-sample/5838
Market Size
1) Baseline & Forecast
●2024: US$ 4.08 B → 2025: US$ 4.33 B → 2034: US$ 7.28 B (CAGR 5.97%, 2025–2034).
●Absolute growth addition 2025–2034: US$ 2.95 B.
2) Growth Drivers Shaping Dollars
●Clinical burden: Rising anemia, leukemias, coagulation disorders; aging cohorts increase routine CBC demand.
●Technology mix: Shift toward automated high-throughput, AI/ML-assisted analyzers, and POCT adds premium ASPs.
●Lab consolidation: Centralized reference labs and hospital networks expand installed base & reagent pull-through.
3) Product Mix Impact on Revenue
●5-part differential analyzers held 51% (2024)—main volume/value anchor.
●6-part & high-end forecast as fastest-growing tier (2025–2034), lifting blended ASPs.
4) Technology Mix Influence
●Flow-cytometry–based platforms commanded 46% (2024); AI/ML-integrated systems to outpace on growth.
5) Customer & Modality Effects
●Hospitals were 42% (2024) of spend; ASC/physician offices to grow fastest with POCT diffusion.
●Standalone systems led 2024 revenue; integrated chemistry/immunoassay combos to accelerate.
6) Regional Dollar Pools
●North America: largest 2024 pool (41%).
●Asia Pacific: fastest incremental dollar creation through 2034, supported by infrastructure build-out.
Market Trends
1) Automation & High Throughput
●Labs upgrade to high-throughput analyzers to handle chronic disease screening and oncology workflows.
2) AI/ML Integration
●Movement toward AI-driven differentials, morphology pre-classification, QC anomaly detection, and workflow orchestration.
3) Point-of-Care Expansion
●POCT CBC analyzers gain traction in clinics/EDs; faster triage and decentralized screening.
4) Strategic Partnerships
●Dec 2024: Axonia Medical partners with multiple OEMs to broaden lab equipment access.
●Apr 2024: PixCell Medical expands U.S. distribution of HemoScreen via Medline, Henry Schein, Thermo Fisher.
●Mar 2024: Sysmex–CellaVision alliance advances integrated hematology solutions.
5) Integrated Diagnostic Platforms
●Combined hematology + chem/immunoassay systems targeted for throughput and single-vendor service.
6) Preventive & Oncology Screening
●Preventive checkups and cancer pathways (pre-chemo, monitoring) sustain recurring CBC volumes.
7) Supply/Trade Dynamics
●China led exports (Oct 2023–Sep 2024; 59 exporters to 85 buyers; +19% growth); France #2 by shipments; Hong Kong exports grew +121% (May 2024–Apr 2025).
●Top importers include Indonesia (210 shipments), Uganda (64), and India (135).
8) Home Healthcare & Telemedicine
●Early interest in home phlebotomy/remote monitoring nudges demand for compact/microfluidic analyzers.
9) Regulatory & Access Push
●Government programs (e.g., Ayushman Bharat in India) and national lab networks improve awareness and test uptake.
10) Compact Instruments with ESR On-board
●HORIBA (Jun 2024) added compact models with ESR on board to reduce reflex testing steps.
10 Ways AI Impacts Hematology Analyzers
1) Differential Pre-classification
●AI models pre-classify WBC subtypes and flag atypical cells, cutting manual review time and inter-observer variability.
2) Image-assisted Morphology
●Vision models on digital slides integrate with analyzers to prioritize smears requiring pathologist review.
3) QC Drift Detection
●Unsupervised algorithms monitor reagent/instrument drift; trigger recalibration before failures.
4) Anomaly & Rare Event Detection
●Outlier detection surfaces blasts, schistocytes, or platelet clumps—improving early leukemia/hemolysis flags.
5) Reflex Testing Orchestration
●Rules engines auto-order retests/ESR/flow cytometry when patterns meet confidence thresholds.
6) Throughput Optimization
●Predictive batching and smart routing balance workload across multi-site lab networks.
7) Error Reduction & Traceability
●Intelligent checks catch sample ID mismatches, anticoagulant issues, or clotted samples before result release.
8) Personalized Baselines
●Patient-specific trend models (delta checks) differentiate biological change from noise—useful in oncology monitoring.
9) POCT Decision Support
●On-device AI in POCT analyzers guides non-specialist users, standardizing performance outside core labs.
10) Cost-to-Serve Analytics
●AI maps utilization, reagent consumption, and service cycles to optimize TCO and service contracts.
Regional Insights
North America (41% share, 2024)
Diagnostic Volume: High chronic disease screening and oncology workloads sustain high CBC/test densities.
Lab Consolidation: Centralized reference labs standardize analyzer fleets, boosting reagent annuities.
Innovation & Pilots: BD + Babson fingertip blood collection (Dec 2024) and AI pilots accelerate decentralized testing.
Asia Pacific (Fastest CAGR 2025–2034)
Infrastructure Build-out: India/China/SEA invest in labs; demand for standalone and mid-tier analyzers.
Disease Burden: Elevated anemia/thalassemia/cancers—especially rural India—raises testing needs.
Policy Levers: Programs like Ayushman Bharat expand awareness/access; private chains scale hubs.
Europe (Solid Growth; tech preference & POCT)
Tech Integration: Flow cytometry + AI + automation adopted to address staffing gaps.
POCT in Outpatient/Emergency: Demand for compact devices with ESR on-board (e.g., HORIBA updates, 2024).
Research Focus: Emphasis on translational medicine and precision diagnostics fuels high-end analyzer uptake.
China
Aging Demographics: Higher hematologic disease susceptibility; government promotes routine testing.
Digital Access: Online healthcare platforms expand therapy awareness and routine follow-ups.
India
Rising Disorders: Anemia/thalassemia prevalence drives CBC penetration beyond metros.
Public-Private Initiatives: Blood donation drives and national insurance coverage support diagnosis pipelines.
Latin America
Modernization: Brazil/Mexico labs upgrade analyzers; logistics improvements aid reagent continuity.
Access Challenges: Public procurement cycles can delay refresh; POCT fills service gaps.
Middle East & Africa
Selective Modernization: Tertiary centers adopt high-end systems; NGOs/private labs drive POCT.
Trade Patterns: Imports from China/HK/US supply growth markets like Uganda.
Market Dynamics
Drivers
Rising blood disorders (anemia, leukemia, coagulation) and aging populations expanding CBC volumes.
Technology advances: AI/ML integration, high-throughput automation, POCT, and microfluidics raise accuracy and speed.
Oncology & Preventive Health: Routine monitoring and screenings cement recurring demand.
Restraints
High upfront costs for advanced systems; budget constraints in developing markets.
Skilled personnel shortages for complex instruments; training/retention gaps.
Regulatory latency can slow the adoption of novel technologies.
Opportunities
6-part/high-end analyzers and integrated platforms for consolidated labs.
Home healthcare & telemedicine: miniaturized, cloud-connected devices for remote monitoring.
AI-enhanced accuracy and automated workflows; microfluidics/LoC innovations.
Top 10 Companies
Sysmex Corporation
Overview: Global IVD leader in hematology with strong presence across core labs and hospitals.
Key Products: 5-part/6-part analyzers; automation lines; digital morphology (with partners).
Strengths: Hematology focus & breadth, strong service network, integration with cell-image workflows.
Beckman Coulter (Danaher)
Overview: Diagnostics major with automation and informatics depth.
Key Products: Hematology analyzers integrated into automated tracks; middleware connectivity.
Strengths: Danaher operational rigor, enterprise lab contracts, interoperability with chemistry/immunoassay.
Abbott Laboratories
Overview: Broad diagnostics portfolio; extensive installed base in chemistry/immunoassay.
Key Products: Hematology systems and reagents aligned to core lab ecosystems.
Strengths: Cross-menu bundling, global distribution, strong POCT heritage.
Siemens Healthineers
Overview: Enterprise diagnostics and imaging giant with integrated lab solutions.
Key Products: Hematology within total lab automation and data platforms.
Strengths: End-to-end integration, hospital system relationships, digital/AI stack.
HORIBA Ltd.
Overview: Analytical technology leader with compact hematology systems.
Key Products: Benchtop analyzers incl. ESR on-board (2024 upgrades).
Strengths: Footprint in decentralized sites, user-friendly workflows, cost-effective models.
Mindray Medical
Overview: High-value medtech with strong growth in IVD.
Key Products: 3-part/5-part analyzers, automation modules, reagents.
Strengths: Value pricing, rapid innovation cycles, expanding global channel.
Roche Diagnostics
Overview: Leading diagnostics player with dominant chemistry/immunoassay footprint.
Key Products: Hematology solutions synergized with Roche middleware/IT.
Strengths: Enterprise access, strong oncology ecosystem, data platforms.
Bio-Rad Laboratories
Overview: Specialty diagnostics and QC leader.
Key Products: Hematology controls, QA/QC tools; specialty analyzers.
Strengths: QC credibility, research ties, specialty niche innovation.
Nihon Kohden
Overview: Clinical instrumentation specialist with lab/monitoring presence.
Key Products: Hematology analyzers for hospitals/clinics.
Strengths: Reliability, hospital channel strength, service quality.
Boule Diagnostics AB
Overview: Focused hematology company serving decentralized labs.
Key Products: Compact analyzers and reagents for routine CBCs.
Strengths: Ease of use, reagent security, affordability for emerging markets.
(Other active players in the market include: Roche, Diatron/STRATEC, PixCell, Dymind, Ortho Clinical, Drucker Diagnostics, Heska (veterinary), Agappe, Dirui, Erba Mannheim, Thermo Fisher—reagents/kits.)
Latest Announcements
Foundation Medicine & Sumitomo Pharma America (Feb 2025, U.S.)
Collaboration to accelerate investigational treatment for acute leukemia with NPM1 mutations or KMT2A rearrangements using FoundationOne® Heme—upstream signal that sustains high-acuity hematology testing demand.
BD & Babson Diagnostics (Dec 2024, U.S.)
Expansion of fingertip blood collection/testing for health systems—supports decentralized blood testing flows and POCT readiness.
Avanzanite Bioscience & Agios (Jun 2025, Europe)
Exclusive agreement to expand access to rare disease medicines—driving specialized hematology pathways needing precise CBC/platelet/coagulation monitoring.
India Blood Donation Drives (May 2025)
Sattva Group + Sankalp India Foundation drive city-wide blood donations—indirectly raising awareness and demand for hematology diagnostics.
Recent Developments
Sysmex America—XQ-320™ 3-part Differential (Mar 2025)
Extends entry segment with reliable CBC in low-volume labs, shortening TAT; strengthens laddered portfolio.
AbbVie—Venetoclax India Launch (Apr 2025)
Expands AML/CLL therapy options; oncology adoption increases pre-/on-treatment monitoring via CBC/differentials.
Novo Nordisk—Therapy Expansion (Mar 2025)
Moves beyond diabetes/obesity into blood & growth disorders—broadens hematology testing cohorts.
HORIBA—Compact ESR-Onboard Models (Jun 2024)
Reduces reflex testing by embedding ESR, improving workflow efficiency in decentralized settings.
Sysmex–CellaVision Alliance (Mar 2024)
End-to-end cell morphology + analyzer synergy; foundation for AI-assisted smear reviews.
PixCell Medical—U.S. Distribution Scale-up (Apr 2024)
HemoScreen POCT CBC expansion via leading distributors—accelerates clinic/office adoption.
Segments Covered
By Product Type
3-Part Differential Analyzers: Entry-level CBCs for smaller labs/clinics; attractive TCO.
5-Part Differential Analyzers (51% in 2024): Standard of care in hospitals; deeper WBC characterization (neutrophils, lymphocytes, monocytes, eosinophils, basophils).
6-Part & High-End: Adds immature granulocyte counts/advanced flags; fastest growth on oncology/critical care needs.
POC Hematology Analyzers: Decentralized settings; rapid triage; expanding via partnerships (e.g., PixCell).
Reagents & Consumables: Recurring revenue backbone; QC/controls critical for accreditation.
By Technology
Impedance-Based: Robust, cost-effective for routine CBC.
Flow Cytometry-Based (46% in 2024): Multiparameter cell analysis; superior specificity for complex cases.
Digital Imaging: Morphology digitization for remote review/AI assist.
AI/ML Integration (Fastest growth): Accuracy, automation, and triage gains across the workflow.
Microfluidics (Miniaturized/Portable): Low sample volumes; drives POCT and potential home-use evolution.
By Application
Complete Blood Count (CBC 58% in 2024): Cornerstone for screening/monitoring across conditions.
Hemoglobin & Hematocrit: Anemia management programs.
Reticulocyte Count: Bone marrow/therapy response insight.
White Cell Differential Count: Infection/oncology decision support.
Coagulation & Platelet Studies: Oncology/cardiovascular pathways; ties to advanced drug delivery monitoring.
By End User
Hospitals (42% in 2024): High acuity; need rapid, reliable results.
Clinical Diagnostic Labs: High-throughput hubs; automation heavy.
Research & Academics: Method development, translational studies.
Blood Banks: Donor screening, quality checks.
ASCs & Physician Offices (Fastest growth): POCT and ambulatory expansion.
By Modality
Standalone Analyzers (2024 leader): Preferred in hospitals/reference labs for throughput & accuracy.
Integrated Systems (Fastest growth): Combined with immunoassay/chemistry; single-workflow patient view.
By Region
North America: Largest share; advanced infrastructure, consolidation.
Asia Pacific: Fastest growth; infrastructure + disease burden.
Europe: Tech integration & POCT preference.
Latin America / MEA: Mix of modernization and POCT bridging access gaps.
Top 5 FAQs
1) What is the market size and growth outlook?
US$ 4.08 B (2024) → US$ 4.33 B (2025) → ~US$ 7.28 B (2034) at 5.97% CAGR (2025–2034).
2) Which region leads and which grows fastest?
North America led with 41% (2024); Asia Pacific grows fastest through 2034.
3) Which product and technology led in 2024?
5-part differential analyzers (51%) and flow cytometry–based technology (46%).
4) Who are the key end users today?
Hospitals dominate (42% in 2024); ASCs/physician offices show fastest growth (2025–2034).
5) Where is AI having the biggest impact?
Differential pre-classification, morphology imaging, QC drift detection, POCT decision support, and workflow automation, improving accuracy and reducing turnaround times and manual workload.
Access our exclusive, data-rich dashboard dedicated to the laboratory equipment sector – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Immediate Delivery Available | Buy This Premium Research @ https://www.towardshealthcare.com/checkout/5838
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Web: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest