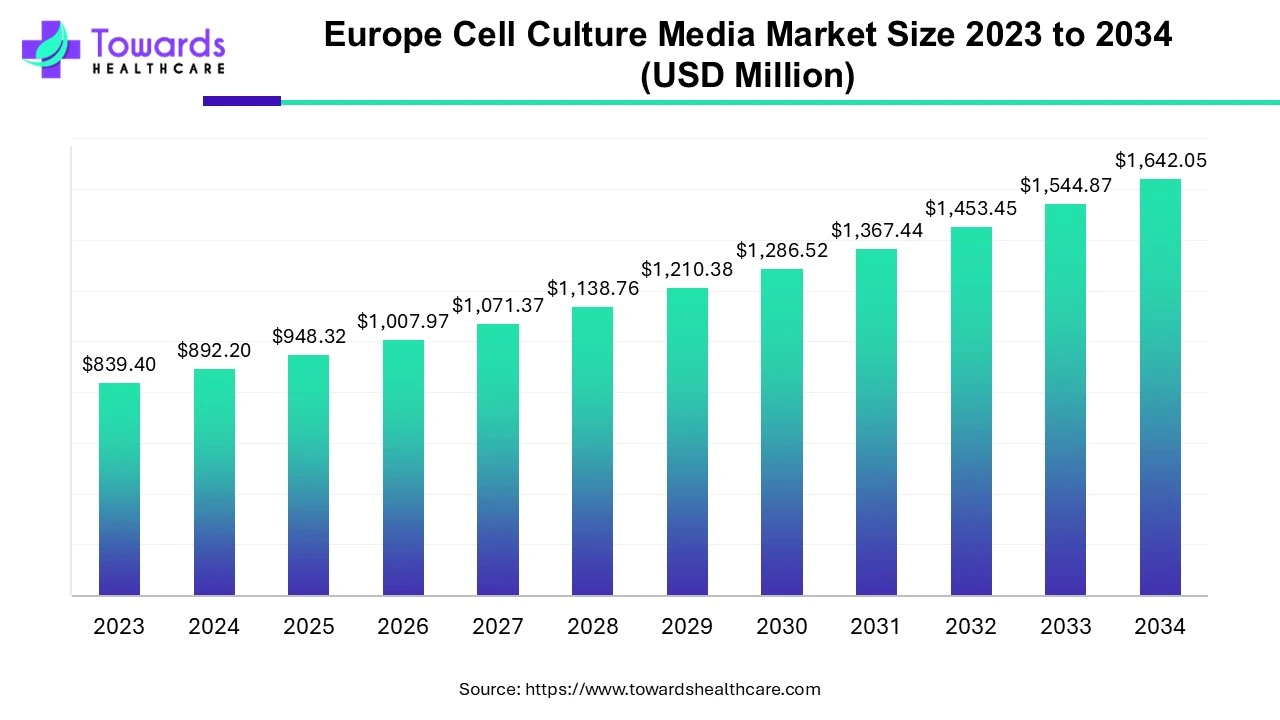
The Europe cell culture media market is projected to reach USD 1,642.05 million by 2034, growing from USD 948.32 million in 2025, at a CAGR of 6.29% (2025–2034), driven by rising R&D activities, technological advancements, and demand for serum-free and chemically defined media for safer, consistent, and high-quality biopharmaceutical production.
Download Free Sample of Europe Cell Culture Media Market Now and Get the Complete Report Easily at: https://www.towardshealthcare.com/download-sample/5027
Europe Cell Culture Media Market Size
2025 Market Value – USD 948.32 million; reflects growing R&D in biologics and vaccines manufacturing.
2034 Market Value – USD 1,642.05 million; robust expansion due to biopharma advancements and stem cell research funding.
CAGR (2025–2034) – 6.29%; steady growth driven by serum-free formulations and automation integration in cell culture systems.
Dominant Type Segment (2024) – Liquid media; accounts for the largest market share owing to ease in sterility and transport.
Fastest-Growing Type Segment – Semi-solid and solid media; supported by drug development and microbiology research demand.
Leading Product Segment (2024) – Serum-free media; preferred for animal-component-free formulations enhancing reproducibility.
Fastest-Growing Product Segment – Stem culture media; fueled by regenerative medicine and chronic disease management initiatives.
End-Use Leader (2024) – Pharmaceutical & biotechnology companies; attributed to advanced infrastructure and capital strength.
Fastest-Growing End-Use Segment – Hospitals & diagnostic laboratories; due to increasing biomarker identification and patient testing.
Regional Leader – France dominates the Europe market due to strong biotech ecosystem and government support for gene therapy.
Europe Cell Culture Media Market Trends
Massive Investments in R&D Infrastructure –
Merck’s €300 million investment (April 2024) to build an Advanced Research Center in Germany strengthens mRNA and antibody manufacturing.
Novartis’ €500 million investment (March 2024) to construct new cell culture facilities in Austria enhances microbial and monoclonal antibody production.
Transition to Serum-Free & Chemically Defined Media – Industry shifts from animal serum to defined, consistent media formulations for reproducibility and regulatory compliance.
Rise in 3D Cell Culture Techniques – Enables better physiological and morphological simulation, improving gene expression and cell-cell communication.
Expansion in Biopharmaceutical Manufacturing – Over 16.5 million liters of biopharmaceutical capacity globally depend on cell culture media inputs.
Emergence of Sustainable Media Components – Focus on plant-derived and recombinant proteins to replace animal-based ingredients.
Integration of AI in Media Optimization – AI tools enhance predictive modeling of nutrient needs and optimize bioreactor media formulations.
Increase in Regenerative Medicine Applications – Cell culture media used for stem cell expansion in tissue regeneration and therapy.
Regulatory Support for Clinical Trials in Europe – France and the UK easing regulatory frameworks for gene and cell therapy accelerates media demand.
EXCiPACT GMP Certification Trend – Merck’s 2024 certification establishes quality leadership in excipient manufacturing for media products.
Collaborative Networks for Innovation – Evonik’s 2024 global competence network integrates cross-continental expertise in cell culture solutions.
10 AI Impacts or Roles in the Europe Cell Culture Media Market
AI-Driven Media Composition Optimization – Algorithms predict optimal nutrient ratios for specific cell types, improving yield and consistency.
Automation in Media Formulation – Machine learning automates ingredient selection and batch preparation, reducing manual error and cycle time.
Predictive Quality Control Systems – AI detects potential contamination or deviation trends early, ensuring sterile and stable production environments.
Digital Twin Models for Bioreactors – AI simulates growth conditions virtually, allowing predictive adjustments for temperature, pH, and nutrient supply.
AI in Personalized Media Design – Machine learning algorithms can suggest customized formulations for specific stem cell lines or patient-derived cultures.
Optimization of Scale-Up Processes – AI helps in modeling nutrient diffusion and gas exchange for large-scale production, reducing experimental time.
Cost-Reduction via Predictive Analytics – AI optimizes resource utilization, minimizing reagent wastage and reducing production costs.
AI-Guided Biopharmaceutical Yield Forecasting – Predictive modeling estimates monoclonal antibody yield under varying media formulations.
Enhanced Data Management – AI organizes experimental data from thousands of culture runs for pattern recognition and R&D insights.
Automated Decision Support Systems – AI-enabled dashboards guide researchers on ideal media selection and timing for feeding or harvesting.
Regional Insights
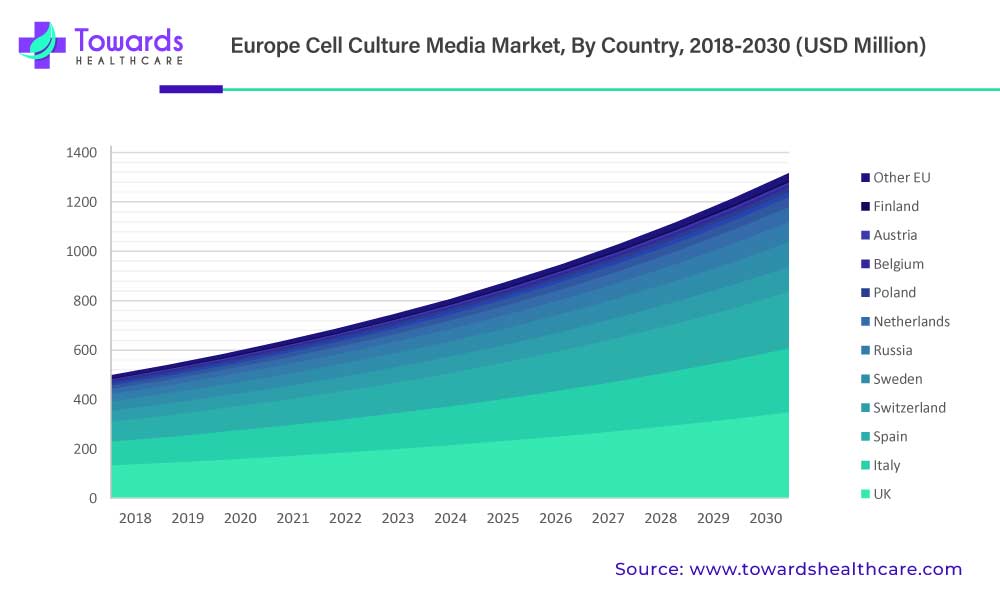
France: Market Leader in Europe
Strong Biotech Ecosystem: Home to advanced cell and gene therapy research hubs.
Government Incentives: France supports R&D collaborations between hospitals and private firms to accelerate cell therapy research.
Regulatory Ease: The French government promotes faster clinical trial approvals for gene therapy applications.
United Kingdom: Second-Largest Market
High Biopharma GVA: UK’s biopharma R&D adds ~£20 billion annually.
Government Funding: Over £100 million invested under the “Synthetic Biology for Growth” program.
Start-Up Ecosystem: Rapid growth of small biotech firms driving demand for consistent media.
Germany: Key Growth Engine
Merck’s €300 Million Center: Focused on mRNA and antibody manufacturing innovations.
Advanced Infrastructure: Strong base for chemical and bioprocess engineering, ensuring quality production of media.
Sweden:
Innovative Materials Research: Development of recombinant spider silk hydrogel for 3D media (Karolinska Institute, 2023).
High Research Quality: Integrating synthetic materials for cell culture applications.
Austria:
Novartis Investments: Building facilities in Kundi and Schaftenau for monoclonal and bispecific antibody production.
Spain & Italy:
Expanding Biopharma Facilities: Increased collaboration with global suppliers for vaccine media development.
Nordic Countries (Denmark & Norway):
Focus on Sustainable Media Sources: Use of eco-friendly materials and synthetic nutrients in culture formulations.
Market Dynamics
Drivers
Emergence of Cell-Based Vaccines: Faster, scalable production compared to egg-based methods for polio, rotavirus, and influenza vaccines.
Rise in Biopharmaceutical R&D: Increased adoption of serum-free media for monoclonal antibody and vaccine manufacturing.
Growing Investment in Research Centers: Merck and Novartis driving European expansion in biomanufacturing infrastructure.
AI & Automation Integration: Enhances media formulation precision and cost efficiency.
Restraints
High Cost of Regenerative Medicine: Limited supplier competition inflates prices for tissue engineering materials.
Complexity in Media Formulation: Challenges in balancing nutrient compositions for diverse cell types.
Opportunities
Evolution of 3D Cell Culture: Enables superior physiological modeling and advanced drug discovery.
Increased Government Funding: France, UK, and Germany supporting local biomanufacturing hubs.
Global Network Collaborations: Evonik’s initiative connects multi-continental R&D to boost innovation.
Top 10 Companies

Merck KGaA
Products: EXCiPACT-certified cell culture media, reagents, and bioprocess solutions.
Strength: Strong global footprint and certified GMP-compliant production sites.
Novartis AG
Products: Biopharma-grade cell media for antibody and vaccine production.
Strength: €500M investment in Austria facilities for microbial and mAb production expansion.
Sartorius AG
Products: Media filtration and bioreactor-integrated solutions.
Strength: Expertise in upstream bioprocess optimization.
FUJIFILM Irvine Scientific
Products: Advanced cell culture solutions for life sciences, IVF, and regenerative medicine.
Strength: Global innovation network and recent business expansion through Astorg acquisition.
Miltenyi Biotec B.V. & Co. KG
Products: Cell separation kits and custom culture media.
Strength: German biotechnology leader with strong immunotherapy applications.
denovoMATRIX
Products: Biomimetic matrices supporting serum-free 3D cultures.
Strength: Innovation in engineered scaffolds for enhanced stem cell culture.
PL BioScience
Products: Human platelet lysate-based media alternatives.
Strength: Pioneering xeno-free growth supplements replacing fetal bovine serum.
PAN-Biotech
Products: Specialty and classical media for academic and industrial R&D.
Strength: Strong product diversity and cost-effective manufacturing.
Eurobio Laboratories
Products: Classical media and custom media formulations.
Strength: Long-standing expertise in European laboratory reagent production.
Cherwell Laboratories Ltd.
Products: Microbiology media, ready-to-use culture plates, and media systems.
Strength: Reliable supplier for diagnostics and hospitals across Europe.
Latest Announcements
January 2025: FUJIFILM Irvine Scientific sold its Medical Media Business Unit to Astorg, expanding Astorg’s global MedTech portfolio in ART and IVF solutions.
September 2024: Merck achieved EXCiPACT cGMP certification for excipient-grade cell culture media, reinforcing industry leadership in quality assurance.
October 2024: Evonik launched a global competence network integrating experts across Europe, Asia, and the Americas to accelerate media optimization innovation.
Recent Developments
Merck (2024): EXCiPACT certification sets benchmark for GMP standards in cell culture media manufacturing.
Evonik (2024): Creation of multidisciplinary competence network for system solutions in upstream bioprocesses.
Karolinska Institute (2023): Development of spider silk-derived hydrogel for 3D cell culture scaffolding.
Novartis (2024): Construction of two new facilities for monoclonal and bispecific antibody production using microbial cell culture.
Merck (2024): €300 million research facility in Germany for advanced antibody and mRNA applications.
Segments Covered
1. By Type
1.1 Liquid Media
Definition & Role: Liquid media refers to nutrient-rich, fluid-based solutions that support microbial and cell growth in suspension. It is the most widely used media type in Europe due to its versatility in biopharmaceutical and microbial applications.
Market Dominance: The segment held the largest share in 2024, driven by its widespread use in large-scale cell and vaccine production.
Key Advantages:
Promotes faster cell growth compared to solid media due to even nutrient distribution.
Enables easy inoculation and transfer, reducing contamination risks.
Facilitates automation in bioreactors, making it suitable for industrial-scale biopharmaceutical manufacturing.
Applications: Used extensively in bioreactors, microbial fermentation, and suspension cultures for vaccine and monoclonal antibody production.
Growth Factors: The adoption of automated bioprocessing and serum-free formulations for scalable production drives the liquid media demand across Europe.
1.2 Semi-Solid & Solid Media
Definition & Role: Semi-solid and solid media are nutrient compositions containing agar or gelling agents, supporting the surface growth of cells and microbes.
Market Growth: Expected to witness significant expansion during 2025–2034 due to advancements in diagnostic microbiology and stem cell research.
Key Advantages:
Provides structural support for colony morphology studies and bacterial isolation.
Used in motility testing and screening of cell differentiation.
Promotes 3D growth environments, essential in regenerative medicine research.
Applications: Plays a critical role in drug screening, microbial identification, and organoid development.
Drivers: Increased investment in regenerative medicine and 3D culture applications enhances demand for solid-based nutrient systems in research laboratories.
2. By Product
2.1 Serum-Free Media
Definition & Role: Serum-free media excludes animal-derived serum, instead using defined formulations of hormones, growth factors, and nutrients.
Market Leadership: It led the Europe cell culture media market in 2024 due to better reproducibility and ethical acceptability.
Key Advantages:
Eliminates batch-to-batch variability associated with fetal bovine serum.
Simplifies downstream purification and GMP compliance.
Reduces the risk of contamination by viruses or prions.
Applications: Extensively used in monoclonal antibody (mAb) production, recombinant protein synthesis, and viral vector manufacturing.
Growth Factors: The shift toward xeno-free and animal-origin-free production enhances its adoption in European pharmaceutical and biotechnology firms.
2.2 Stem Cell Culture Media
Definition & Role: Specialized formulations designed to maintain stem cell pluripotency and proliferation under controlled conditions.
Growth Outlook: Expected to gain a significant market share by 2034 due to growing applications in regenerative medicine, gene therapy, and chronic disease treatments.
Key Advantages:
Supports self-renewal and lineage-specific differentiation.
Ensures optimal nutrient balance for sensitive stem cell lines.
Compatible with 3D scaffold environments used in tissue engineering.
Applications: Used in embryonic stem cell (ESC) culture, induced pluripotent stem cell (iPSC) expansion, and therapeutic transplantation research.
Drivers: The rising prevalence of chronic diseases and growing R&D funding for cell-based therapies in France, Germany, and the UK.
2.3 Classical Media & Specialty Media
Definition & Role: Classical media are well-established nutrient solutions such as DMEM, RPMI, and MEM, while specialty media are tailored formulations designed for specific cell types or functions.
Key Advantages:
Provides a cost-effective and standardized option for general cell growth and maintenance.
Specialty formulations are customized for cancer research, hybridoma culture, or transfection studies.
Applications: Used widely in diagnostic labs and research institutes for general cell line maintenance and testing.
Growth Factors: The continued demand for legacy research protocols ensures sustained usage across universities and contract research organizations (CROs).
2.4 Chemically Defined Media (CDM)
Definition & Role: Media with precisely known chemical compositions that eliminate any undefined biological components.
Market Potential: Witnessing increasing adoption due to its suitability for regulatory-compliant biomanufacturing.
Key Advantages:
Offers high reproducibility and minimal contamination risk.
Enables AI-driven optimization for specific cell lines.
Simplifies quality control and documentation for GMP processes.
Applications: Critical for cell line development, monoclonal antibody production, and vaccine manufacturing.
Drivers: Growing demand from large-scale biopharmaceutical producers and the trend toward fully synthetic media compositions in Europe.
3. By End-Use
3.1 Pharmaceutical & Biotechnology Companies
Dominance: Accounted for the largest share of the market in 2024.
Key Drivers:
Possess state-of-the-art R&D facilities and advanced bioreactor systems.
Increasing investments in biosimilars and monoclonal antibody development.
Utilize AI-integrated cell culture platforms for high-throughput screening and optimization.
Applications: Large-scale production of vaccines, mAbs, and recombinant proteins.
Country Focus: Germany and the UK house key pharma manufacturers and CDMOs actively adopting automated cell culture systems.
3.2 Hospitals & Diagnostic Laboratories
Growth Potential: Projected to show lucrative growth during 2025–2034.
Key Drivers:
Expanding use of cell-based diagnostic assays and biomarker analysis.
Hospitals increasingly employ cell culture media for cancer diagnostics and personalized medicine.
Rising patient volume and adoption of in-vitro testing tools enhance demand.
Applications: Used for tissue analysis, genetic screening, and pathogen detection.
Regional Insight: High adoption rate in France and the UK, supported by healthcare modernization and laboratory automation.
3.3 Research Institutes & Academic Centers
Role in Market: Serve as innovation hubs driving scientific breakthroughs in stem cell and gene therapy research.
Key Advantages:
Use of customized and specialty media for experimental optimization.
Collaboration with pharma companies and government agencies accelerates translational research.
Integration of AI-based data analysis tools for predictive modeling in cell behavior studies.
Regional Support: The European Union’s Horizon programs and national science councils fund multiple projects focusing on cell biology and regenerative medicine.
4. By Region
4.1 France – Market Leader in Europe
Market Position: France dominates the European cell culture media market due to a robust biotech ecosystem and government-backed initiatives.
Growth Drivers:
The French government supports rapid clinical trial approvals for gene therapy.
Strong collaboration between research institutions and private biotech firms.
Home to leading players like Eurobio Laboratories, Hepacult GmbH, and PL BioScience.
4.2 United Kingdom – Innovation Hub
Economic Impact: The UK’s biopharmaceutical R&D contributes approximately £20 billion in GVA annually.
Growth Factors:
Government’s Synthetic Biology for Growth initiative funding over £100 million in R&D.
Growing number of biotech startups adopting serum-free and defined media.
Increased collaboration between academia and CDMOs for mAb and vaccine manufacturing.
4.3 Germany – Major Production and R&D Center
Infrastructure Strength: Houses Merck’s €300 million Advanced Research Center for antibody and mRNA manufacturing.
Market Strengths:
Leader in chemically defined media production.
Key players like Sartorius AG and Miltenyi Biotec dominate R&D integration and bioprocess optimization.
4.4 Austria – Emerging Manufacturing Base
Strategic Investments: Novartis’ €500 million investment in Kundi and Schaftenau facilities to produce monoclonal and bispecific antibodies.
Impact: Strengthening the upstream cell culture supply chain in Central Europe.
4.5 Sweden – Research-Driven Market
Innovation: Development of recombinant spider silk hydrogel by Karolinska Institute (2023) for 3D media applications.
Focus: Enhancing cell culture scaffolds for tissue engineering and drug development research.
4.6 Southern Europe (Italy & Spain)
Trend: Expanding biopharmaceutical infrastructure and increasing collaboration with global suppliers.
Applications: Increased focus on vaccine and recombinant protein production using serum-free media.
4.7 Nordic Region (Denmark & Norway)
Market Strength: Focused on sustainable, plant-based media formulations to replace animal-derived products.
Innovation Focus: Adopting AI-powered monitoring tools for media optimization in bioprocess facilities.
Top 5 FAQs
-
What is the market size of the Europe cell culture media market?
The market was valued at USD 948.32 million in 2025 and is projected to reach USD 1,642.05 million by 2034, growing at a CAGR of 6.29%. -
Which segment led the market in 2024?
The liquid media type and serum-free product segment dominated due to superior sterility and animal-free composition. -
What factors are driving the market?
Rising biopharmaceutical R&D, AI-based optimization, government funding, and 3D cell culture evolution. -
Which country dominates the European market?
France leads the market, followed by the UK and Germany, driven by government support and biotech investment. -
What are the latest major developments?
Merck’s EXCiPACT GMP certification, Evonik’s global network launch, and Novartis’ €500M facility investments in Austria.
Access our exclusive, data-rich dashboard dedicated to the biotechnology Sector – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout Europe Cell Culture Media Market Report Now at: https://www.towardshealthcare.com/checkout/5027
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest