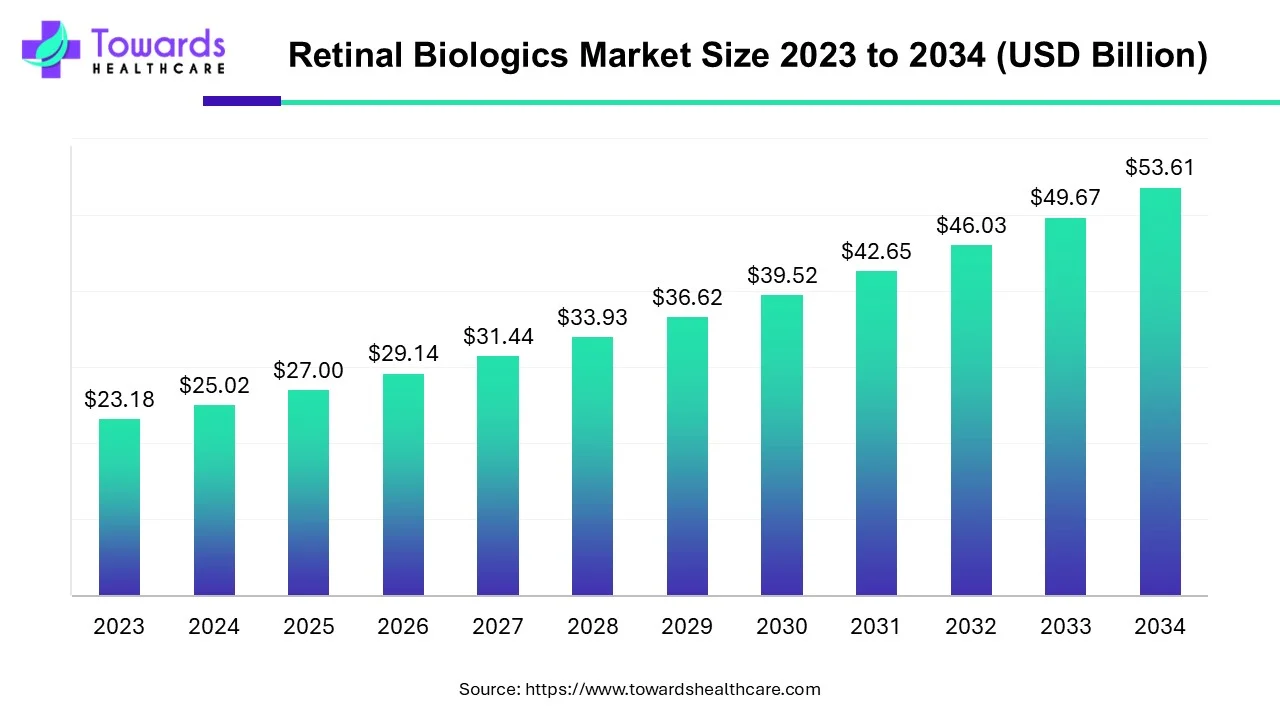
The global retinal biologics market is projected to grow from USD 27.0 billion in 2025 to USD 53.61 billion by 2034, expanding at a CAGR of 7.92% (2025–2034) as biologic therapies (especially VEGF-A antagonists and novel gene/antisense approaches) address rising burdens of age-related macular degeneration and diabetic retinopathy.
Download Free Sample: https://www.towardshealthcare.com/download-sample/5166
Market size
Base and forecast figures: Market value is USD 27.0B (2025) → USD 53.61B (2034). This implies near doubling of market value across the forecast window and reflects both volume growth and introduction/uptake of higher-value biologic therapies.
Compound growth rate: CAGR 7.92% — consistent growth driven by increasing disease prevalence, new drug approvals, and uptake of biosimilars and advanced delivery/therapy formats.
Price / value composition: A sizeable portion of market value is driven by high per-unit cost of biologics (complex production, R&D amortization), compounded by premium prices for novel agents and gene therapies.
Therapeutic class weighting: VEGF-A antagonists form a dominant share (largest drug-class contributor) — legacy biologics and newer entrants (bispecifics, combination biologics) add higher average selling prices.
Biosimilar impact on size composition: Entry of biosimilars (e.g., ranibizumab and aflibercept biosimilars) shifts market value mix — they increase access and volumes but may depress list prices, altering revenue distribution across originator vs biosimilar players.
Pipeline to market conversion: Late-stage pipeline programs and gene therapies (e.g., ABBV-RGX-314 program planned pivotal program) represent upside to market size if approved and broadly commercialized.
Geographic growth mix: North America contributes the largest absolute share; Asia-Pacific expected fastest relative growth (high population base + increasing healthcare infrastructure) — this affects regional revenue shares of the global USD figures.
Channel economics: Hospital pharmacy channel holds largest share historically — hospital procurement and inpatient/outpatient administration patterns influence where revenues are captured.
Reimbursement & payor effect: Public and private payor coverage decisions — especially for expensive biologics and gene therapies — materially influence realized market size versus theoretical demand.
Cost restraints & elasticity: High cost of biologics is a headwind that can cap uptake in price-sensitive regions; however, unmet need and sight-saving value create inelastic demand among covered patients.
Market trends
Dominance of VEGF-A antagonists: Anti-VEGF remains the therapeutic backbone for neovascular retinal disorders; agents like aflibercept, ranibizumab, bevacizumab (and newer bispecifics such as faricimab) continue to command largest market share.
Biosimilar and price competition: Introduction of biosimilars (BYOOVIZ, Yesafili, Cimerli, ABP-938 biosimilar approvals/launches) is expanding access and changing pricing dynamics; biosimilars reduce list price (BYOOVIZ listed ~40% below Lucentis historically) and shift market share.
Gene and long-duration therapies rising: Investigational programs (ABBV-RGX-314, OCU400, Kodiak’s candidates) signal movement toward gene-based or long-durational treatments that could reduce injection frequency and reshape treatment paradigms.
Regional acceleration in Asia-Pacific: Rapid expansion of healthcare infrastructure in China and India and aging populations drive fastest CAGR in Asia-Pacific.
Hospital pharmacies as primary distribution: Hospital pharmacy dominance indicates hospital/clinic administration models persist; hospital procurement controls reimbursement and usage.
Aging population drives demand: Epidemiologic shifts (increased centenarian figures, growing 60+/65+ cohorts) raise AMD and other age-related retinal disease prevalence, increasing lifetime treatment need.
Diabetes epidemic fuels DR demand: Rising diabetes prevalence and projected DR cases (presently ~103 million → projected 161 million by 2045 per cited meta-analysis) directly expand demand for retinal biologics.
Shift to personalized ophthalmology: Advances in diagnostics, sequencing, and translational research promote individualized therapies (gene therapy, modifier gene strategies) — enabling targeted treatment for inherited retinal disorders.
M&A, partnerships, licensing activity: Companies restructure portfolios via settlements and licensing (e.g., Biocon-Bayer settlement for Yesafili) and commercial transactions (Sandoz/Coherus) to accelerate market entry.
Cost and manufacturing complexity remain restraints: High R&D and manufacturing costs (biologics complexity; estimated development costs approaching ~$1.1B) constrain price reductions and may slow access in lower-income regions.
AI 10 deep point-wise roles/impacts on the retinal biologics market
Target identification and discovery acceleration
Subpoint: AI-driven analysis of omics and imaging datasets can uncover novel retinal disease targets and pathways (e.g., new angiogenic modulators beyond VEGF).
Explanation: By integrating genomics, transcriptomics and retinal imaging, AI finds patterns humans miss, shortening discovery timelines and increasing probability of high-value biologic candidates.
Biomarker discovery for patient stratification (personalized medicine enablement)
Subpoint: Machine learning models can identify molecular or imaging biomarkers that predict responder vs non-responder to specific biologics.
Explanation: Enables targeted trials, reduces trial size, and supports payer arguments for cost-effective personalized therapies, aligning with the market’s shift toward personalized ophthalmology.
Clinical trial optimization and virtual trial design
Subpoint: AI selects optimal inclusion criteria, simulates outcomes, and identifies high-yield sites/patients for retinal biologics trials (especially for rare inherited diseases).
Explanation: Reduces trial dropouts and shortens timelines — critical for expensive biologic programs (reducing the near-$1.1B risk).
Automated retinal image analysis for diagnosis, progression monitoring and endpoints
Subpoint: Deep learning algorithms read OCT and fundus images to grade disease severity, quantify fluid/leakage, and measure anatomical endpoints objectively.
Explanation: Provides scalable diagnostics in clinics, enables remote monitoring, and creates standardized endpoints for trials and reimbursement dossiers.
Predictive modeling for dosing regimens and durability
Subpoint: AI models predict individual pharmacodynamic responses, suggesting tailored dosing intervals (e.g., spacing intravitreal injections).
Explanation: Can reduce injection frequency, improve adherence, and support value demonstrations for long-acting gene therapies or bispecifics.
Manufacturing process optimization and batch quality prediction
Subpoint: Machine learning monitors bioreactor sensor data to predict yields, detect deviations, and optimize upstream/downstream processes.
Explanation: Mitigates batch variability (a key biologics challenge), improves yield and reduces cost per dose — directly addressing cost restraints.
Supply chain forecasting and demand planning
Subpoint: AI forecasts regional demand (age cohorts, diabetes prevalence, treatment penetration) to optimize inventory and reduce stockouts in hospital pharmacies.
Explanation: Ensures high-cost biologics are available where needed while minimizing wastage from expired vials.
Real-world evidence (RWE) generation and value demonstration
Subpoint: AI extracts structured insights from electronic health records and imaging archives to show long-term effectiveness and safety.
Explanation: Supports reimbursement negotiations and pharmacoeconomic arguments for expensive biologics and gene therapies.
Regulatory submission support and automated document generation
Subpoint: Natural language models draft parts of regulatory packages, summarize trial findings, and map evidence to regulatory questions.
Explanation: Speeds dossier preparation for approvals and biosimilar filings (reducing time to market).
Patient engagement, adherence prediction and tele-ophthalmology triage
Subpoint: AI chatbots and predictive models identify patients at high risk of non-adherence to injection schedules and trigger outreach or tele-visits.
Explanation: Improves clinical outcomes and helps maintain consistent treatment regimens, particularly important where frequent intravitreal injections are standard of care.
Regional insights
North America — largest absolute market share
Healthcare infrastructure & R&D: Strong clinical research ecosystem and payer systems that reimburse biologics enable high uptake.
Chronic disease burden & aging population: High prevalence of AMD and DR increases demand; forecasts show rising older cohorts and high economic cost of AMD (~USD 30B/year estimate referenced).
Commercial environment: Large commercial launches and rapid adoption of biosimilars/novel agents supported by distribution through hospital pharmacies and clinic networks.
Asia-Pacific — fastest CAGR (highest relative growth)
Demographic drivers: Large and aging populations in China, India, Japan accelerate absolute patient numbers.
Infrastructure investment: Rapid increases in hospital capacity and specialist availability (China’s vast network of hospitals and medical staff).
Access dynamics: Price sensitivity but expanding insurance coverage; biosimilars can play a rapid adoption role to scale volume.
Europe — growth through biosimilars and favorable regulations
Regulatory environment: Supportive biosimilar pathways (e.g., recent approvals like faricimab in EU) encourage competition and access.
Market nuance: National health systems emphasize cost-effectiveness and often negotiate prices, pushing biosimilar uptake.
Latin America — constrained by cost but large unmet need
Access limitations: Public budgets and procurement processes determine access; hospital pharmacy channel still key where available.
Opportunity: Rapid gains possible with lower-cost biosimilars and tiered pricing strategies.
Middle East & Africa — nascent uptake, high unmet need
Infrastructure variability: Limited specialist density in many countries; major urban centers lead adoption.
Access strategies: Tiered pricing, philanthropic programs, and tele-ophthalmology could accelerate adoption.
Cross-regional dynamics — role of biosimilar & gene therapy launches
Timing of launches & licensing agreements (e.g., Biocon-Bayer, Sandoz/Coherus) determines when lower-cost alternatives become available regionally.
Reimbursement heterogeneity will create regional differences in which novel therapies achieve commercial scale.
Market dynamics
Demand drivers: disease prevalence & aging (structural demand)
Evidence: Aging populations and diabetes prevalence (DR currently ~103M projected to 161M by 2045) drive sustained clinical need.
Supply drivers: innovation & pipeline
Evidence: Active pipeline (gene therapy, bispecifics, long-acting biologics like those from Kodiak, REGENXBIO collaborations) can shift standard of care.
Pricing & reimbursement pressures
Explanation: High development/manufacturing costs (~$1.1B) justify premium pricing; payors demand cost-effectiveness and RWE.
Competitive landscape: originators vs biosimilars
Effect: Biosimilar launches (BYOOVIZ, ABP-938, Yesafili, Cimerli) increase competition—volume up, price down for some molecules.
Distribution concentration in hospital pharmacies
Effect: Hospital procurement influences market access and negotiation power; hospitals centralize administration of intravitreal biologics.
Regulatory & intellectual property considerations
Effect: Patent expirations and settlements (e.g., Biocon-Bayer) change entry timing for biosimilars, affecting market dynamics.
Manufacturing complexity & batch variability risk
Effect: Complexity raises cost, raises barrier to smaller entrants, and creates quality consistency challenges.
Commercial partnerships & M&A activity
Effect: Licensing deals and acquisitions speed market access and scale commercialization (e.g., Sandoz/Coherus transaction).
Health economics & value demonstration importance
Effect: Providers and payors demand demonstration of reduced treatment burden (e.g., fewer injections) and long-term visual outcomes.
Technology convergence (AI, digital diagnostics, wearable/telemedicine)
Effect: These technologies can reduce care gaps, improve monitoring, and support rollouts in resource-limited settings — ultimately expanding addressable market.
Top 10 companies
AbbVie Inc. (U.S.)
Key product/program: ABBV-RGX-314 investigational program (gene therapy for wet AMD).
Overview: Large biopharma with robust R&D and commercial infrastructure; full-year 2024 revenue ~$56.334B (growth y/y).
Strength: Financial firepower, clinical development expertise, capability to run pivotal Phase 3 programs and global launches.
Amgen Inc. (U.S.)
Key product/program: ABP-938 — a biosimilar of EYLEA (aflibercept) approved by U.S. FDA.
Overview: Large biotech with proven biologics development and manufacturing capabilities.
Strength: Strong biologics manufacturing scale and established commercial channels for ophthalmology biosimilars.
Santen Pharmaceutical Co., Ltd (Japan)
Key product/program: Eylea® 8 mg (aflibercept recombinant solution) introduced/distributed in Japan (with Bayer Yakuhin marketing license).
Overview: Ophthalmology-focused specialist with regional market knowledge.
Strength: Deep ophthalmic focus and regional market relationships enabling targeted launches.
Biocon Biologics / Biocon (India)
Key product/program: Yesafili biosimilar of EYLEA; Biocon-Bayer settlement enabling Canada access.
Overview: Biosimilar leader leveraging cost advantages to expand access.
Strength: Cost-competitive manufacturing and partnerships to accelerate geographic entry.
Bayer AG (Germany)
Key product/program: Eylea originator marketer (marketing license holder in certain territories).
Overview: Large diversified healthcare company with strong ophthalmology franchise.
Strength: Global reach, established ophthalmology portfolio, and commercialization capacity.
Novartis AG (Switzerland)
Key product/program: Originator company for Lucentis (ranibizumab) historically; competitor in retinal space.
Overview: Global pharmaceuticals leader with scale and R&D.
Strength: Strong clinical development and commercialization, legacy retinal assets.
Kodiak Sciences Inc. (U.S.)
Key product/program: Pipeline candidates (tarcocimab, KSI-501, KSI-101) targeting combined mechanisms (e.g., anti-VEGF ± anti-inflammatory).
Overview: Clinical-stage specialty biotech focused on durable retinal therapeutics.
Strength: Innovative mechanistic approaches and late-stage pipeline candidates that could alter treatment paradigms.
AstraZeneca (U.K.)
Key product/program: Active in biologics, potential pipeline/partnership involvement in ophthalmology.
Overview: Large global pharma with broad biologics expertise.
Strength: R&D depth and global commercialization.
Amneal / Sun Pharmaceutical / Aurobindo / Teva / Fresenius / Merck / Pfizer — (examples from list)
Product/overview: These firms play roles as biosimilar developers, generic injectables suppliers, or global distributors.
Strength: Manufacturing scale, cost efficiency, distribution networks, and ability to support hospital pharmacy channels.
Ocugen, Inc. (U.S.)
Key product/program: OCU400 — gene-modifier platform being evaluated for Leber congenital amaurosis and RP (Phase 1/2 updates).
Overview: Small biotech focusing on inherited retinal diseases with gene-agnostic modifier approach.
Strength: Novel platform potentially addressing genetically heterogeneous retinal disorders.
Latest announcements
AbbVie & REGENXBIO (Jan 2025)
Announcement: Investigation of ABBV-RGX-314 in wet AMD patients; plan for pivotal (Phase 3) program in 2026.
Implication: If successful, a gene therapy that reduces injection burden could materially disrupt chronic anti-VEGF market economics and shift long-term revenue models.
Amgen — ABP-938 FDA approval (date noted)
Announcement: U.S. FDA approval of ABP-938 (aflibercept biosimilar) for certain angiogenic eye disorders.
Implication: Biosimilar competition to EYLEA expands access and compresses pricing for high-value aflibercept market share.
Santen (April 2024)
Announcement: Launch of Eylea® 8mg (114.3 mg/mL) in Japan (with Bayer Yakuhin marketing license).
Implication: New presentations/formulations target dosing convenience and regional distribution synergies.
Biocon Biologics — settlement with Regeneron/Bayer (Mar 2024)
Announcement: Agreement to allow sale of Yesafili in Canada; Biocon to introduce by July 1, 2025 latest.
Implication: Biosimilar entry timelines clarified, enabling market planners and payors to prepare for price competition.
Sandoz & Coherus (Jan 2024 deal)
Announcement: Sandoz paid $170M for biosimilar ranibizumab (Cimerli) assets and commercial support.
Implication: Strategic acquisitions accelerate biosimilar commercialization with ready inventory and sales infrastructure.
Ocugen (Sept 2023 / trial updates)
Announcement: Safety/efficacy updates for OCU400 in LCA and RP — extension data from high-dose cohorts.
Implication: Gene-modifier strategies progress through clinical stages, potentially enabling treatments for inherited retinal degeneration.
Biogen & Samsung Bioepis (June 2022)
Announcement: U.S. launch of BYOOVIZTM (ranibizumab biosimilar) at significant discount versus originator.
Implication: Demonstrates commercial viability of biosimilars in ophthalmology and their effect on pricing dynamics.
Recent developments
Multiple biosimilar launches and deals (BYOOVIZ, Yesafili, Cimerli, ABP-938) increasing competition and access.
Gene therapy programs advancing to pivotal planning (ABBV-RGX-314 planned Phase 3 in 2026).
Novel molecule pipelines (Kodiak’s tarcocimab, KSI series) targeting combined pathways and longer durability.
Commercial transactions accelerating market entry (Sandoz/Coherus purchase of Cimerli assets; Biocon-Bayer settlement).
Market expansion in APAC through launches and distribution agreements (Santen/Bayer in Japan; China hospital expansion metrics).
Clinical updates in inherited retinal disease programs (Ocugen’s OCU400) suggesting movement in rare disease segment.
Segments covered
By Drug Class
VEGF-A Antagonist: Primary therapy group — reductions in vascular leakage and vision restoration (Aflibercept 79–83% effect range; Ranibizumab 61–75%; Bevacizumab 59–68% as reported). Dominant revenue contributor.
TNF-A Inhibitor: Used for inflammatory retinal conditions (e.g., uveitis) — valuable in subsegment where inflammation is key driver.
Other biologic classes: Growth factors, immunomodulators, monoclonal antibodies and blood/plasma products — collectively expand therapeutic options beyond anti-VEGF.
By Indication
Macular degeneration (AMD): Largest indication share — aging population is the primary driver.
Diabetic retinopathy (DR): High unmet need as diabetes prevalence rises; DR affects 30–40% of diabetics and contributes materially to volume demand.
Uveitis and others: Smaller but clinically important subsets where immunomodulatory biologics are used.
By Distribution Channel
Hospital Pharmacies: Largest share — hospitals control in-clinic administration and large volume procurement.
Retail Pharmacies: Important for outpatient prescriptions in markets with community ophthalmology dispensing.
Online Pharmacies: Emerging channel for distribution of ancillary supplies or scripts where permitted.
By Region
North America: Largest share — strong R&D, reimbursement.
Europe: Significant growth with biosimilars and favorable regulation.
Asia-Pacific: Fastest CAGR — driven by population size and infrastructure expansion.
Latin America / MEA: Growing but constrained by access and budgets.
Top 5 FAQs
-
Q: What is the market size and growth rate for retinal biologics?
A: The market is projected to grow from USD 27.0B in 2025 to USD 53.61B by 2034, at a CAGR of 7.92% between 2025–2034. -
Q: Which drug class dominated the retinal biologics market in 2023?
A: VEGF-A antagonists dominated in 2023; they remain the cornerstone of treatment for neovascular retinal disorders. -
Q: What are the main demand drivers for the market?
A: Aging populations (higher AMD prevalence), rising diabetes and diabetic retinopathy burden (current ~103M DR cases projected to 161M by 2045), increasing healthcare infrastructure, and expanding biologic/gene therapy options. -
Q: How do biosimilars and new therapies affect the market?
A: Biosimilars (e.g., BYOOVIZ, Yesafili, Cimerli, ABP-938) expand access and lower list prices, increasing volume but compressing originator revenues; gene/long-acting therapies (e.g., ABBV-RGX-314) could reduce injection frequency and alter lifetime revenue per patient. -
Q: Which regions will show the fastest growth and who dominated in 2023?
A: North America dominated the market in 2023 (largest absolute share); Asia-Pacific is expected to grow at the fastest CAGR during the forecast period due to demographic scale and infrastructure expansion.
Access our exclusive, data-rich dashboard dedicated to the Therapeutics area Sector – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout Retinal Biologics Market Report Now at: https://www.towardshealthcare.com/checkout/5166
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest
