Regulatory Compliance Is a Key Barrier to Entry in the 503A U.S. Compounding Market
Pharmacy compounding exists to meet patients’ unique needs, such as creating drug formulations in strengths, dosages, or forms not commercially available, or avoiding allergens and preservatives for patients with sensitivities.
Under 503A, compounding pharmacies fill prescriptions written for individual patients. A licensed pharmacist or physician compounds a drug for an identified patient after receiving a valid prescription. Pharmacies may prepare limited quantities in advance only if there is an established history with the patient or prescriber. 503A focuses on personalized medications on a case-by-case basis rather than mass production. This ensures patients with special needs, including pediatric formulations or rare dosage strengths, receive medications suited specifically to them.
Although 503A offers more flexibility than conventional drug manufacturing, it still imposes a heavy regulatory burden. Meeting its requirements in a compliant manner represents a major challenge for new or smaller pharmacies.
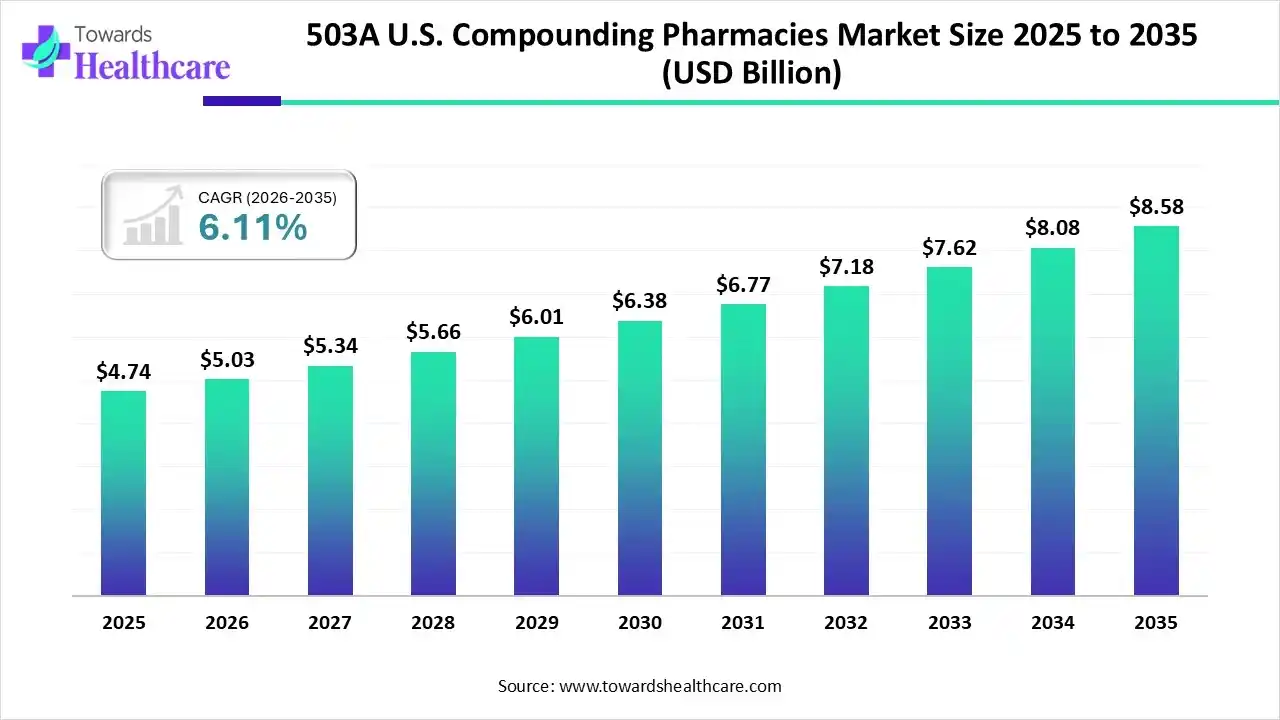
The 503A U.S. compounding pharmacies market is projected to reach USD 8.58 billion by 2035, growing from USD 5.03 billion in 2026, at a CAGR of 6.11% during the forecast period from 2026 to 2035, increasing demand for personalized, convenient and accessible care.
Download Free Sample: https://www.towardshealthcare.com/download-sample/5070
Legal Framework: 503A Requirements and Exemptions
When compounding under 503A, some regulatory requirements that apply to conventional drugs are waived. Pharmacies are exempt from certain manufacturing standards such as Current Good Manufacturing Practice, labeling with adequate directions for use, and premarket approval for the compounded drug. These exemptions reduce the complexity compared to full-scale manufacturing, making patient-specific compounding more feasible.
Despite these exemptions, 503A imposes strict conditions that pharmacies must meet. The drug must be compounded for a specific patient based on a valid prescription, or in limited amounts under narrowly defined conditions. Bulk drug substances must meet strict sourcing criteria, typically requiring an official monograph, inclusion in FDA-approved drugs, or placement on the approved bulks list. Compounded drugs cannot be essentially copies of commercially available drugs unless prescribed for significant patient-specific reasons. Pharmacies cannot promote specific compounded drugs and are restricted in interstate distribution. These rules ensure compliance and maintain patient safety.
Why Regulatory Requirements Create a High Barrier to Entry
The regulatory requirements impose substantial burdens on new or smaller pharmacies.
Infrastructure and Quality Control Needs
Even though 503A does not require full manufacturing compliance, compounding, especially sterile preparation, demands strict environmental control, proper storage of raw materials, validated procedures, and ongoing quality control. Many pharmacies follow standards such as USP <795> for nonsterile and USP <797> for sterile compounding. Meeting these standards requires specialized equipment, controlled environments, validated cleaning protocols, and regular monitoring. These capital and operational costs can be a major barrier for small pharmacies.
Skilled Personnel and Training Requirements
Compounding requires trained pharmacists who understand sterile technique, drug stability, proper labeling, recordkeeping, and patient-specific dosing. Maintaining compliance involves ongoing staff training, validation of processes, and meticulous documentation. Smaller pharmacies often lack the resources to hire or train enough qualified staff.
Challenges for New Entrants
New entrants in the 503A compounding market face several challenges. First, the financial burden is high. Establishing a compliant facility requires investment in clean rooms, equipment, monitoring systems, and sourcing verified drug substances. Second, regulatory compliance is complex and constantly evolving. Small pharmacies must stay updated on permitted bulk substances, monographs, distribution limits, and patient-specific prescription rules. Third, the business model is limited in scalability. 503A pharmacies cannot produce large batches or supply hospitals or clinics without prescriptions, restricting revenue potential. Fourth, risk management is demanding. Any lapse in sterility, documentation, or sourcing can lead to regulatory enforcement, product recalls, or shutdowns, which small pharmacies may not withstand.
These challenges mean that newcomers must carefully plan their operations, invest substantially, and commit to rigorous ongoing compliance to survive in the market.
Restrictions on Volume, Distribution, and Business Model
503A pharmacies must focus on patient-specific prescriptions and cannot produce large batches for stock or office use. They cannot supply hospitals or clinics without prescriptions and face limits on interstate distribution. This restricts scalability and revenue potential, making profitability difficult for new entrants.
Sourcing Constraints and Raw Material Compliance
503A pharmacies can only use bulk substances that meet specific criteria. Newer or niche substances, including certain peptides, are often off-limits unless approved. This limits the ability of small pharmacies to innovate or offer specialized formulations.
Regulatory Uncertainty and Compliance Risk
The compliance landscape is dynamic. Approved substances, monographs, and guidance may change, meaning that a formulation compliant today may become noncompliant tomorrow. Small pharmacies face higher risk from such regulatory changes, including potential business disruption.
Comparison: 503A vs. 503B: Why 503B Often Looks More Attractive for Scale?
To fully understand why 503A presents a barrier for small entrants, it helps to consider the alternative: 503B outsourcing facilities.
503B facilities operate under the same FD&C Act but are explicitly registered with the FDA. They are subject to more stringent regulations, including full compliance with current Good Manufacturing Practices, validated manufacturing processes, batch testing, and strict labeling and quality assurance protocols.
503B facilities can compound drugs in large batches without individual patient-specific prescriptions and supply hospitals, clinics, and other healthcare providers. They can also distribute across state lines without the distribution restrictions that 503A pharmacies typically face.
Because of these differences, 503B facilities have the scale and flexibility to act more like small manufacturers. They can produce larger volumes, spread fixed costs over many units, achieve economies of scale, and serve institutional customers rather than only individual patients. For many businesses, this model appears far more attractive from a growth and profitability standpoint.
By contrast, 503A pharmacies remain focused on small-batch, patient-specific compounding, similar to craftsmen supplying bespoke goods rather than factories producing commodity items. For a large-scale, growth-oriented business, this model may not provide sufficient return on investment or scalability.
Why Established Pharmacies Have a Competitive Edge
Compliance under 503A favors established pharmacies. Experienced pharmacies have already invested in clean rooms, equipment, environmental controls, and trained staff. They have developed standard operating procedures, documentation systems, and inspection experience. They can absorb compliance risks and operate efficiently.
New entrants face high upfront investment costs, lack institutional knowledge, and have limited ability to scale. Regulatory compliance, while essential for safety, creates a competitive advantage for established pharmacies while keeping smaller players out.
Market Impact of Regulatory Compliance
The 503A market tends to be dominated by well-established pharmacies. Distribution remains localized, and patients rely on individual pharmacies for personalized medications. Hospitals and clinics usually source drugs from 503B facilities, segmenting the market. Regulatory changes, including bulk substance approvals and interstate distribution rules, continue to shape opportunities and risks.
Established pharmacies increasingly partner with 503B facilities for complex or sterile compounding. Partnerships help them meet specialized demand while maintaining compliance, but they require careful management and additional resources.
Implications for New Entrants and Smaller Pharmacies
New or small pharmacies must recognize the challenges: substantial upfront investment, rigorous compliance systems, limited scalability, and the need for ongoing regulatory awareness. Partnerships with outsourcing facilities may be necessary for sterile or hazardous compounding, but these add complexity. Starting a 503A pharmacy requires realistic planning, resources, and dedication to maintaining compliance.
Importance of Regulatory Oversight
Regulations are necessary to protect patient safety. Compounded drugs are not FDA-approved and have not undergone rigorous testing. Without strict rules on sourcing, compounding environment, and documentation, there is a significant risk of contamination, dosing errors, or substandard products. Regulatory oversight ensures quality, safety, and accountability in the compounding process.
Access our exclusive, data-rich dashboard dedicated to the healthcare industry – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout 503A U.S. Compounding Pharmacies Market Report Now at: https://www.towardshealthcare.com/checkout/5070
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest