A New Race Begins in Biomanufacturing
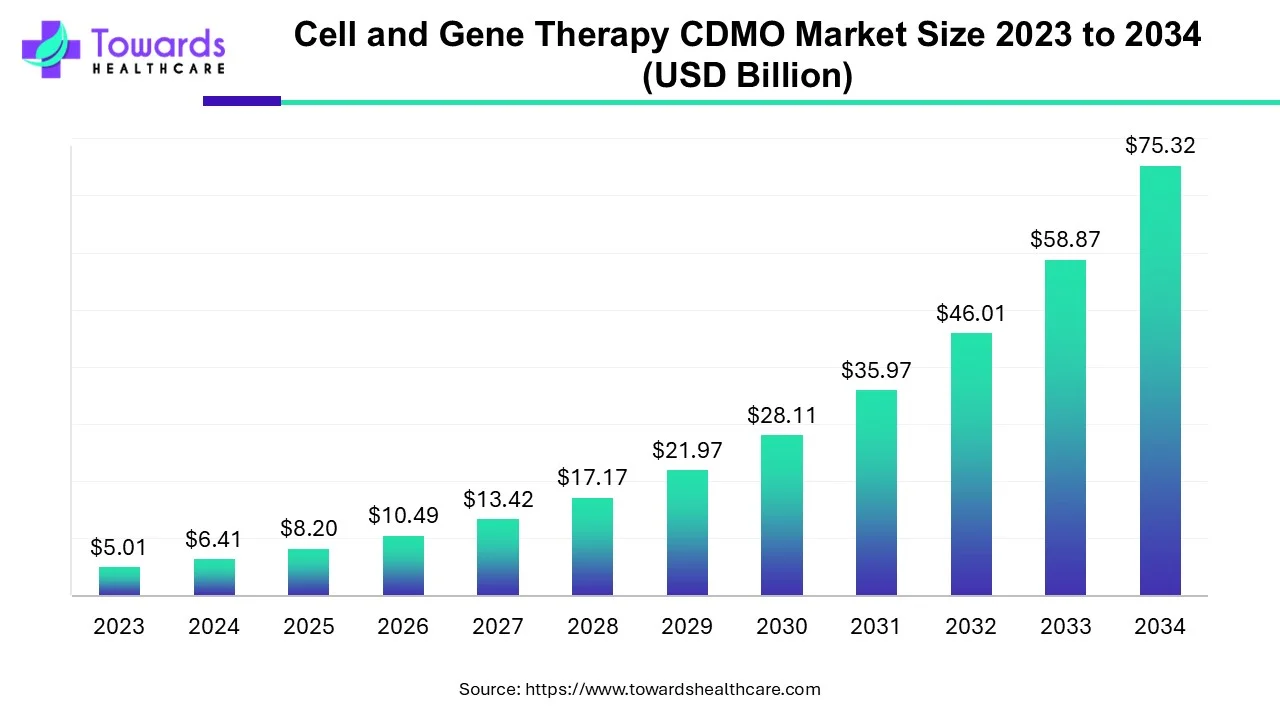
The global cell and gene therapy CDMO ecosystem is standing at a defining moment. The market, valued at just over USD 6.41 Billion in 2024, is moving at an unprecedented pace and is projected to cross USD 75.32 Billion by 2034. This is not a gradual shift but a profound acceleration driven by scientific breakthroughs, surging clinical activity, and the mounting pressure to transform research insights into real-world medical solutions. The world is now witnessing a rapid rise in diseases that demand personalized treatments, and this has pushed biotech innovators to depend more than ever on specialized outsourcing partners. CDMOs have evolved from supportive manufacturing units into strategic pillars of the therapeutic development ecosystem, reshaping how advanced therapies reach patients across the globe.
Download Sample:
https://www.towardshealthcare.com/download-sample/5452
Why the World Needs CDMOs More Than Ever
Cell and gene therapies have created new hope for patients suffering from rare diseases, genetic disorders, oncology indications, and life-threatening conditions that failed to respond to conventional treatments. These therapies rely on highly complex biological systems, often involving living cells or genetic material engineered to restore, replace, or correct patient biology. Such solutions require manufacturing environments that are as precise as they are innovative. This is where CDMOs enter the value chain, offering specialized infrastructure, deep expertise, and an operational architecture designed for therapies that are anything but standard.
The intense demand for these therapies has exposed the limitations of existing manufacturing capacity. Biotech startups often lack the internal capabilities to manage scale-up, regulatory compliance, viral vector production, and clinical batch manufacturing. Even larger pharmaceutical firms choose outsourcing to reduce capital burden and mitigate risk. As a result, the CDMO sector is no longer a complementary partner but an essential extension of the entire biopharmaceutical pipeline.
The CDMO Shift: From Support System to Strategic Engine
Across the last decade, CDMOs have transformed into highly adaptive organizations capable of operating at every step of therapy development. Their involvement now begins far before clinical trials and continues long after commercialization. From process development and viral vector engineering to GMP-grade manufacturing and regulatory documentation, CDMOs provide the backbone that holds the development journey together. Consolidation through mergers, acquisitions, and strategic alliances has strengthened this transformation. Each acquisition brings new technologies, expanded capacity, and specialized platforms that together accelerate the creation of therapies that once took years to develop.
The increasing reliance on CDMOs is also a result of a fundamental shift in biotech innovation. More than 90 percent of emerging biotech firms do not possess internal manufacturing capabilities. To stay competitive and meet the relentless demand for advanced therapies, they must operate with external partners who can deliver both speed and scalability. CDMOs, as a result, are becoming indispensable to the entire sector, especially in early research translation and late-stage commercial readiness.
The Era of Advanced Therapies Has Arrived
Cell and gene therapies have initiated a healthcare revolution, powered by living cells, genome editing tools, and engineered vectors. The potential of these treatments stretches far beyond symptom management. They offer curative possibilities for diseases previously deemed incurable. Yet, their promise also creates extraordinary pressure on the manufacturing ecosystem. Every therapy requires individualized processes, from customizing viral vectors to maintaining cell integrity throughout development.
These demands require an infrastructure built on precision, high automation, sterile environments, and specialized scientific expertise. CDMOs functioning in this space ensure that the complexity of living therapeutics does not become a barrier to accessibility. They modernize each step of the production pipeline, ensuring that both emerging innovators and global pharmaceutical leaders can deliver safe, effective treatments.
Innovation Takes the Lead as AI Reshapes Biomanufacturing
Artificial intelligence has become a driving force in reshaping bioprocessing, especially within the cell and gene therapy sector. Manufacturing unpredictability has long been one of the most significant challenges. Variations in cell viability, vector yields, contamination risks, and production bottlenecks can compromise entire batches. AI is addressing these challenges by analyzing millions of data points in real time to predict bottlenecks before they occur. AI-driven systems monitor biological processes continuously, identifying deviations and automating adjustments to maintain stability.
CDMOs integrating AI into their workflows are already experiencing enhanced throughput, more consistent yields, and reduced process failures. Predictive modeling is transforming decision-making, turning manufacturing into a more reliable and cost-efficient process. In an industry where every mistake is expensive and every delay impacts patient outcomes, AI has become a critical ally.
Demand Surges as Clinical Pipelines Expand
The growth of the cell and gene therapy CDMO market is tightly linked to the global rise in clinical trials. Today, dozens of therapy candidates are advancing across oncology, rare diseases, and infectious diseases. The FDA is reviewing a rising number of submissions, while regulatory bodies across Europe and Asia are encouraging the development of advanced therapy medicinal products. Biologics approvals have accelerated year over year, creating a pipeline that heavily depends on CDMO capacity.
The biotech sector’s increasing willingness to outsource is also driving market growth. Startups, academic spinouts, and early-stage innovators now recognize outsourcing as the fastest path to translate research into clinical programs. Reduced risk, lower cost, and access to state-of-the-art technologies make outsourcing a strategic imperative rather than an operational choice.
The High Cost of Innovation: A Structural Challenge
Despite extraordinary growth, the cell and gene therapy manufacturing landscape faces significant barriers. The complexity of developing therapies that rely on fragile living cells and customized genetic constructs makes manufacturing inherently challenging. Specialized infrastructure, advanced bioreactors, sterile environments, real-time monitoring systems, and highly trained personnel drive costs upward. The financial burden of building such facilities is often prohibitive for small companies. Even CDMOs themselves face steep investments to expand their capabilities.
The long development timelines and unpredictable nature of manufacturing add further complexity. Scaling personalized therapies from laboratory conditions to commercial volumes remains one of the biggest hurdles. These challenges limit market accessibility for smaller innovators, creating a competitive landscape dominated by firms with significant financial backing.
Collaboration Becomes the Fastest Path to Progress
While high costs and complexity remain barriers, collaborations have emerged as a powerful solution. Partnerships allow companies to share risk, pool expertise, and accelerate development timelines. Universities contribute scientific discoveries, biotech firms bring innovation, and CDMOs provide manufacturing and regulatory expertise. Together, these collaborations create ecosystems where discoveries move more smoothly through development and into clinical evaluation.
Such alliances not only reduce cost but also enhance operational efficiency. Knowledge transfer between partners strengthens process control, regulatory planning, and quality assurance. In an industry where delays cost millions and impact patient outcomes, partnership models are becoming the norm rather than the exception.
Why Oncology Dominates the Value Chain
Oncology remains the leading indication within the cell and gene therapy CDMO market. The global cancer burden continues to rise, with tens of millions of new cases reported annually. Conventional therapies often fall short in treating aggressive or late-stage cancers, driving the search for transformative treatments. Cell-based immunotherapies and genetically modified cells, especially CAR-T and TCR-T solutions, have reshaped cancer care.
CDMOs support oncology-focused therapies by ensuring scale-up, standardization, and regulatory compliance. Their role becomes even more critical as oncology pipelines grow larger, more complex, and more dependent on advanced manufacturing platforms capable of maintaining cell potency and viability. Every new cancer therapy candidate entering preclinical or clinical phases expands the role of CDMOs in global oncological innovation.
Rare Diseases Gain Momentum in the Advanced Therapy Landscape
While oncology leads in volume, rare diseases are emerging as one of the fastest-growing categories. Over three hundred million individuals suffer from rare conditions, and most have no approved treatments. Genetic origins underline nearly eighty percent of these conditions, making them ideal candidates for gene therapy interventions. The need for early diagnosis, targeted approaches, and curative outcomes pushes biopharmaceutical companies to invest heavily in rare disease pipelines.
CDMOs specializing in rare diseases must handle limited batch sizes, individualized process designs, and stringent regulatory expectations. Their expertise helps innovators move beyond conceptual research into actual therapeutic development. As awareness and diagnosis improve, the demand for rare disease therapies will accelerate even further, placing CDMOs at the center of this rapidly evolving landscape.
The Preclinical Stage Strengthens as a Strategic Foundation
Preclinical development has become a major driver of CDMO growth. Advances in scientific techniques, including gene editing, cell engineering, and vector optimization, have enabled researchers to design therapies with greater precision. A rising number of early-stage programs are entering the pipeline, creating pressure on CDMOs to provide specialized preclinical support.
This segment reflects the early experimentation and optimization needed to ensure that therapies move into clinical studies with well-defined mechanisms and safe manufacturing protocols. As more programs emerge across oncology, neurology, infectious diseases, and rare conditions, the preclinical stage will continue to shape the overall direction of the CDMO market.
Clinical Manufacturing Accelerates as New Approvals Loom
The clinical segment of CDMO services is expanding rapidly as more programs advance into human trials. Each new therapy requires meticulous quality control, validated processes, regulatory documentation, and scalable production. Investors are pouring resources into companies advancing clinical-ready products, anticipating that new approvals will reshape treatment landscapes across multiple regions.
As clinical trials multiply in the US, Europe, and Asia, CDMOs are adjusting their facilities to manage growing demand. The industry expects an increasing number of cell and gene therapy approvals over the next few years, reinforcing the need for experienced manufacturing partners capable of handling commercial-scale readiness.
Cell Therapy Continues to Lead as the Dominant Product Type
Cell therapy has captured a major share of the market due to the remarkable pace of innovation and clinical evidence supporting its effectiveness. Thousands of clinical and preclinical studies are underway, showcasing strong momentum in CAR-T therapies, stem cell therapies, and regenerative treatments. CDMOs play an indispensable role in enabling this growth by offering GMP-certified environments, expansion platforms, and specialized expertise.
The challenge moving forward lies in preparing for next-generation cell therapies. Researchers anticipate the emergence of more efficient, scalable, and targeted cell therapies. CDMOs must modernize their capabilities, integrate automation, and adopt new platforms to remain competitive.
Gene-Modified Cell Therapies Rise as New Lifelines
Gene-modified cell therapies represent the next evolutionary step in therapeutic innovation. These treatments combine cellular engineering with genetic modification, unlocking the ability to design highly targeted therapies for both malignant and non-malignant diseases. Their growing effectiveness in clinical studies has intensified investment into this field.
This category is expected to grow significantly in the coming decade, driven by advancements in delivery systems, improved editing technologies, and more efficient manufacturing methods. CDMOs offering specialized capabilities for gene-modified cell therapies will see strong demand as innovators pursue more ambitious therapeutic targets.
North America Leads Through Investment and Innovation
North America remains the global hub for cell and gene therapy development. Research institutions, clinical trial density, and high R&D expenditure have created a fertile environment for the CDMO sector. The region hosts hundreds of active trials and is home to some of the world’s most advanced manufacturing facilities. Large-scale investments in biomanufacturing, particularly in New Jersey, California, and Massachusetts, reinforce North America’s leadership.
The strong presence of biotech companies and regulatory clarity offered by the FDA also contribute to the region’s dominance. As funding continues to flow into startups and commercial manufacturing hubs, the region will remain a cornerstone for global growth.
Asia Pacific Emerges as the Fastest-Growing Region
The Asia Pacific region is rapidly gaining ground as the fastest-growing market for cell and gene therapy CDMOs. China leads this surge with substantial clinical activity, followed by Japan, South Korea, and India. Governments across the region are investing heavily in R&D infrastructure and regulatory reforms to attract global innovators.
Large patient populations, cost efficiencies, and growing collaborations between local and international research centers make Asia Pacific a strategic location for manufacturing expansion. As clinical trials increase, the demand for advanced CDMO services will scale accordingly.
Europe Reinforces Its Position with Scientific Excellence
Europe continues to strengthen its presence through scientific leadership, technological adoption, and growing investment in personalized medicine. The region has a vibrant ecosystem of biotech startups and academic institutions working on advanced therapy development. Regulatory support from the EMA enhances Europe’s ability to advance clinical research, while capital investments boost CDMO capacity.
The growing number of ATMP approvals across Europe reflects the region’s commitment to innovation and its ability to support advanced manufacturing capabilities.
A Future Defined by Scale, Speed, and Scientific Precision
The future of the cell and gene therapy CDMO market will be shaped by its capacity to scale rapidly, innovate continuously, and maintain the highest standards of safety and precision. Demand will continue to rise as more therapies enter the pipeline, patients seek targeted solutions, and biopharma companies push for faster commercialization.
CDMOs must embrace automation, AI-driven insights, modular facilities, flexible production systems, and strong global networks. Those capable of meeting these evolving demands will define the next era of advanced therapeutics.
Important Links
Access our exclusive, data-rich dashboard dedicated to the healthcare market – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout Cell and Gene Therapy CDMO Market Report Now at: https://www.towardshealthcare.com/checkout/5452
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest