For more than a decade, regenerative medicine has quietly shifted from an ambitious scientific idea into one of the most consequential forces shaping modern healthcare. What once lived largely in academic journals and experimental labs now stands at the center of clinical innovation, commercial strategy, and long-term healthcare planning. As someone who has observed this market evolve from its earliest translational breakthroughs to today’s advanced clinical pipelines, it is clear that regenerative medicine is no longer a future promise. It is a present reality with profound implications.
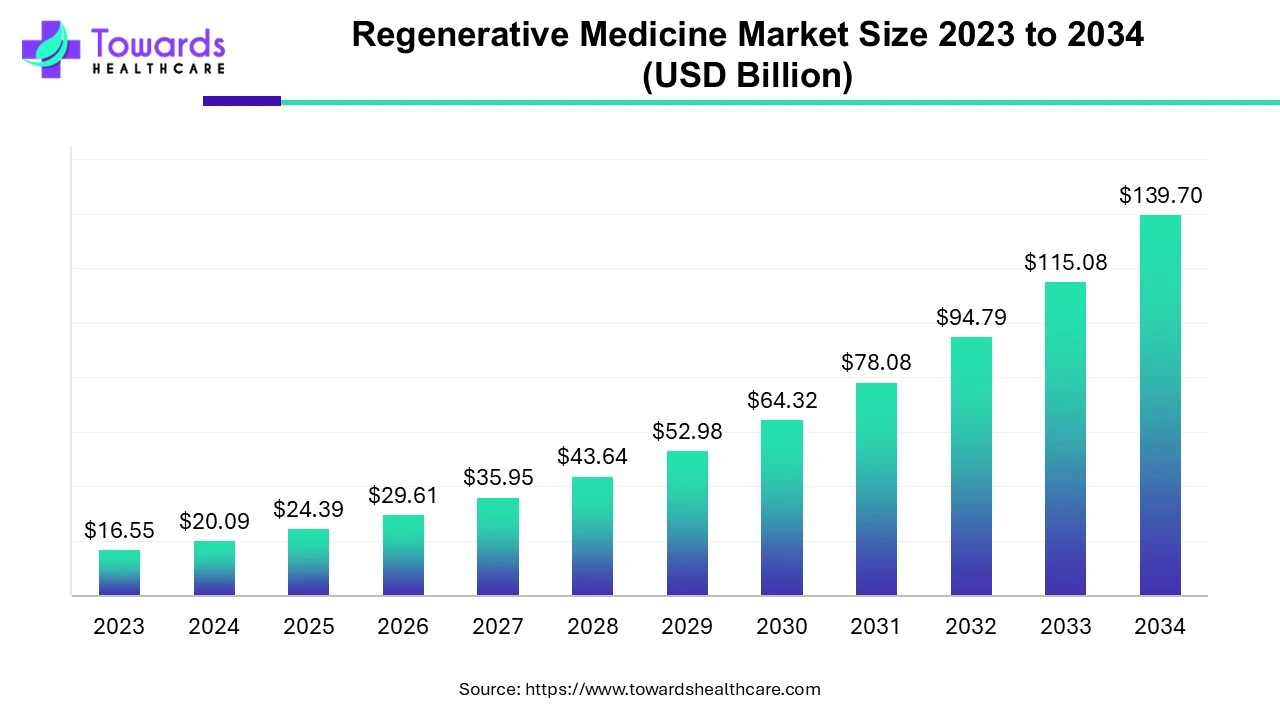
The global regenerative medicine market reflects this transformation. Projected to expand from USD 24.39 billion in 2025 to nearly USD 139.70 billion by 2034, the sector is advancing at a striking compound annual growth rate of over 21 percent. This growth is not driven by hype or speculative science. It is fueled by real clinical needs, rising chronic disease prevalence, expanding clinical trial success, and a growing recognition that traditional therapies alone cannot meet the demands of aging populations and complex conditions.
Download Free Sample and Get the Complete Report Easily at: https://www.towardshealthcare.com/download-sample/5092
At its core, regenerative medicine seeks to restore function rather than merely manage symptoms. It focuses on repairing, replacing, or regenerating damaged cells, tissues, and organs by working with the body’s own biological systems. This philosophy represents a fundamental departure from conventional treatment models and explains why clinicians, researchers, regulators, and investors increasingly view regenerative medicine as a cornerstone of next-generation healthcare.
From Organ Shortages to Biological Solutions
The origins of regenerative medicine are deeply tied to one of medicine’s most persistent challenges: organ and tissue loss. For decades, transplantation stood as the only viable solution for patients with end-stage organ failure. Yet donor shortages, immune rejection, long waiting lists, and lifelong immunosuppression created significant clinical and ethical limitations.
Regenerative medicine emerged as a response to these constraints. By combining engineering principles with life sciences, researchers began exploring ways to grow tissues, engineer grafts, and stimulate endogenous repair mechanisms. Over time, these approaches matured from theoretical models into FDA-approved therapies that are now commercially available.
The urgency behind this shift remains evident. Tens of thousands of patients continue to wait for kidney, liver, heart, and other organ transplants each year, while new patients are added to waiting lists at an alarming pace. Regenerative medicine does not simply aim to supplement transplantation. It seeks to reduce dependence on donor organs altogether by offering biologically integrated alternatives.
Why Chronic Diseases Are Accelerating Market Momentum
One of the most powerful forces driving regenerative medicine adoption is the global rise in chronic disorders. Cancer, autoimmune diseases, neurological conditions, hematological disorders, cardiovascular diseases, and musculoskeletal degeneration place immense strain on healthcare systems worldwide. Traditional pharmacological approaches often slow disease progression but rarely reverse damage.
Regenerative therapies address this gap by targeting the underlying biological deficits. Cell-based therapies can replace dysfunctional cells, gene therapies can correct inherited defects, and tissue-engineered constructs can restore structural integrity. As chronic diseases become more prevalent and patients live longer with complex conditions, demand for restorative treatments continues to grow.
This trend is especially evident in wound care, where regenerative medicine has demonstrated clear clinical and economic value. Chronic wounds, diabetic ulcers, and severe burns present persistent treatment challenges. Advanced cell-based grafts, tissue-engineered products, and biologically active dressings are transforming outcomes in this segment, which is now one of the fastest-growing application areas in the market.
Clinical Trials Are No Longer a Bottleneck
In the early years of regenerative medicine, limited clinical data constrained adoption and investment. That barrier has largely fallen. Today, the number of clinical trials evaluating regenerative therapies continues to expand across regions and indications. Governments, academic institutions, and private companies actively collaborate to move therapies from bench to bedside.
The maturation of clinical pipelines has changed industry perception. Many regenerative therapies have progressed into late-stage trials, signaling readiness for commercialization. This shift has encouraged pharmaceutical companies and medtech leaders to integrate regenerative strategies into their long-term portfolios rather than treating them as peripheral research projects.
Favorable regulatory frameworks have also played a critical role. Agencies increasingly recognize the unique nature of regenerative therapies and have introduced pathways designed to balance patient safety with innovation. While regulatory complexity remains a challenge, progress in this area has strengthened confidence across the ecosystem.
Cell-Based Therapies Take Center Stage
Among all product categories, cell-based products have established a dominant presence in the regenerative medicine market. Their success lies in their versatility and biological potency. These therapies include cellular immunotherapies, cancer vaccines, autologous and allogeneic stem cells, and progenitor cell products that can differentiate into specialized tissues.
Cell-based therapies excel because they actively participate in tissue formation and repair. Unlike inert implants or conventional drugs, living cells respond to their environment, integrate with host tissues, and adapt over time. This dynamic behavior aligns closely with the regenerative goals of restoring function and durability.
Autologous cell therapies, which use a patient’s own cells, reduce immune rejection risks but often require complex harvesting and expansion processes. Allogeneic therapies, derived from donor sources with low antigenicity, offer scalable, off-the-shelf solutions that improve accessibility and manufacturing efficiency. The coexistence of these models reflects the market’s diversity and adaptability.
Tissue Engineering Moves From Concept to Clinic
While cell-based products currently dominate, tissue-engineered products represent the fastest-growing segment in regenerative medicine. These therapies combine engineered cells, biomaterials, and bioactive signals to regenerate or replace damaged tissues. Advances in 3D bioprinting, scaffold design, and organ-on-a-chip technologies have accelerated progress in this field.
Tissue engineering addresses some of the most complex clinical challenges, including cartilage regeneration, bone defects, vascular repair, and soft tissue reconstruction. Researchers increasingly focus on mimicking native tissue architecture and promoting vascular integration, which is essential for long-term graft survival.
As manufacturing technologies improve and costs decline, tissue-engineered products are moving closer to routine clinical use. Their growth signals a broader shift toward integrated, multi-disciplinary solutions that combine biology, materials science, and engineering.
Musculoskeletal Applications Drive Real-World Adoption
Few areas illustrate the practical impact of regenerative medicine as clearly as musculoskeletal care. Conditions such as osteoarthritis, ligament injuries, tendon degeneration, and cartilage damage affect millions worldwide and often lead to chronic pain, reduced mobility, and diminished quality of life.
Regenerative approaches offer alternatives to invasive surgeries and joint replacements. Mesenchymal stem cells, growth factor therapies, scaffold-based implants, and autologous chondrocyte implantation techniques aim to restore tissue function rather than replace entire joints.
These therapies resonate strongly with patients and clinicians alike because they align with goals of faster recovery, reduced surgical risk, and long-term preservation of natural anatomy. As populations age and remain active longer, musculoskeletal regenerative solutions will continue to gain traction.
Neurology and Brain Injury Open New Frontiers
One of the most compelling emerging applications for regenerative medicine lies in neurology, particularly in the treatment of brain injuries. Traumatic brain injury and spinal cord injury impose lasting physical, cognitive, and emotional burdens that conventional therapies struggle to address.
Regenerative medicine introduces new possibilities by promoting neural repair, protecting existing neurons, and restoring damaged neural circuits. Stem cell therapies, tissue-engineered constructs, and neuroprotective strategies are increasingly explored in clinical research.
The growing prevalence of brain injuries, combined with heightened awareness and advocacy, has attracted significant investment and regulatory interest. Positive clinical trial outcomes in this area could fundamentally alter treatment paradigms and further expand the regenerative medicine market.
Stem Cell Therapy as a Catalyst for Market Expansion
Stem cell therapy stands as one of the most transformative applications within regenerative medicine. Its ability to generate specialized cells for tissue repair has captured global attention. Laboratories now routinely guide stem cells to become blood cells, heart muscle cells, or nerve cells, creating unprecedented therapeutic possibilities.
The market impact of stem cell therapy extends beyond clinical outcomes. Government funding, regulatory approvals, and public-private partnerships have accelerated innovation and commercialization. Landmark approvals of cell-based gene therapies demonstrate how rapidly the field is advancing.
At the same time, the industry continues to address challenges related to standardization, safety, and ethical considerations. Responsible development remains essential to sustaining long-term trust and growth.
Regulation Remains a Test of Maturity
Despite its progress, regenerative medicine faces regulatory hurdles that reflect its complexity. Smaller companies and academic groups often lack access to specialized regulatory expertise, creating delays and inefficiencies. Measurement standards, manufacturing consistency, and long-term safety data remain areas of active development.
However, these challenges also signal market maturation. As regulatory frameworks evolve and measurement science advances, regenerative medicine will benefit from greater predictability and scalability. Collaboration between regulators, industry, and research institutions will be critical in navigating this phase.
Regional Dynamics Shape Global Growth
Geographically, North America continues to lead the regenerative medicine market, supported by advanced healthcare infrastructure, high chronic disease prevalence, and strong investment in research and development. The region’s dominance reflects both scientific leadership and favorable reimbursement environments.
Asia-Pacific represents the fastest-growing region, driven by aging populations, rising healthcare expenditure, and increasing adoption of advanced therapies. Countries such as Japan, China, and India play distinct roles, combining innovation, manufacturing capacity, and expanding clinical demand.
Europe maintains a strong position through strategic investments, robust biotech ecosystems, and government support for gene and cell therapy development. Meanwhile, the Middle East and Africa show growing potential as research collaborations, medical tourism, and public funding expand.
Competition Fuels Innovation, Not Saturation
The competitive landscape in regenerative medicine remains dynamic rather than crowded. Established pharmaceutical and medtech companies coexist with agile biotech startups and academic spin-offs. Collaboration, rather than direct competition, often defines success in this market.
Partnerships accelerate discovery, reduce development risk, and enable commercialization at scale. As technologies converge and clinical evidence accumulates, competitive advantage increasingly depends on execution, manufacturing excellence, and regulatory strategy.
Looking Ahead: From Repair to Regeneration
Regenerative medicine has reached a pivotal moment. It has moved beyond experimental optimism and entered an era defined by clinical relevance and market credibility. The sector’s growth reflects more than financial projections; it reflects a broader shift in how medicine approaches healing.
Rather than asking how long patients can live with disease, regenerative medicine asks how fully function can be restored. This change in perspective carries profound implications for patients, healthcare systems, and society at large.
As research deepens, technologies mature, and collaboration expands, regenerative medicine will continue to redefine what is medically possible. The question is no longer whether this field will shape the future of healthcare, but how quickly its promise will become standard practice.
Access our exclusive, data-rich dashboard dedicated to the therapeutic area sector – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Important Links:
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout Regenerative Medicine Market Report Now at: https://www.towardshealthcare.com/checkout/5092
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest