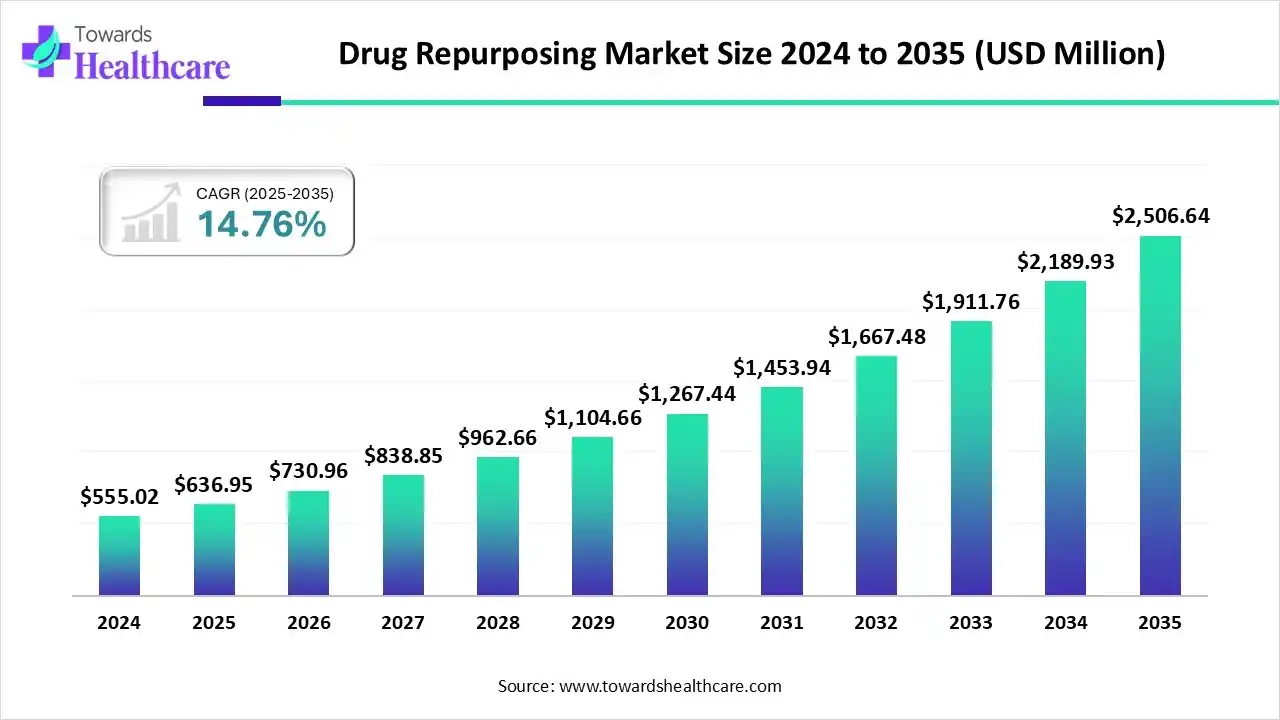
The global drug repurposing market has quietly transformed from a niche scientific strategy into one of the most credible growth engines in modern pharmaceutical development. In 2025, the market stands at approximately USD 636.95 million, but this figure only hints at what lies ahead. By 2035, projections point toward a valuation exceeding USD 2.5 billion, driven by a strong compound annual growth rate of nearly 15 percent. This growth does not emerge from hype or speculative science. It reflects a structural shift in how the industry thinks about risk, time, cost, and therapeutic innovation.
Download Free Sample Now and Get the Complete Report Easily at: https://www.towardshealthcare.com/download-sample/6424
Drug repurposing, also referred to as drug repositioning, focuses on identifying new clinical uses for existing drugs, including approved, shelved, or investigational compounds. Unlike traditional drug discovery, which can take over a decade and billions of dollars to deliver a single approved therapy, repurposing leverages known safety profiles and clinical histories. This approach compresses timelines, lowers failure rates, and offers faster relief to patients facing diseases with limited or no treatment options.
What makes this market particularly compelling is its alignment with today’s healthcare reality. The global burden of chronic diseases, rare disorders, neurodegenerative conditions, and multiple cancers continues to rise. At the same time, healthcare systems face mounting cost pressures, regulatory scrutiny, and demands for faster innovation. Drug repurposing sits at the intersection of these challenges, offering a pragmatic and scalable solution.
Why the World Is Turning Back to Existing Drugs
The surge in interest around drug repurposing is not accidental. Pharmaceutical companies, biotech startups, regulators, and academic institutions increasingly recognize that the traditional pipeline model is unsustainable on its own. Attrition rates remain high, especially in late-stage clinical trials, where failures carry enormous financial and emotional costs.
Repurposing addresses this problem directly. When a compound already has established pharmacokinetics, toxicity data, and human exposure records, researchers can redirect resources toward understanding disease mechanisms rather than starting from scratch. This advantage becomes especially powerful in rare and neglected diseases, where patient populations are small and commercial incentives for de novo drug discovery remain limited.
Cancer, neurological disorders, autoimmune diseases, and metabolic conditions have become focal points for repurposing strategies. In oncology, for instance, drugs initially developed for diabetes or cardiovascular disease now show promise in modulating tumor growth pathways. In neurology, compounds once abandoned for lack of efficacy in one indication are finding renewed relevance in conditions such as Parkinson’s disease, spinal cord injury, and rare neuroinflammatory disorders.
Technology Is Redefining What Repurposing Can Achieve
While serendipity once played a major role in drug repurposing, today’s market is driven by data, algorithms, and computational precision. Artificial intelligence, machine learning, and deep learning models now sit at the core of repurposing workflows. These technologies analyze massive biological datasets, including genomics, transcriptomics, proteomics, clinical trial records, and real-world evidence, to uncover hidden connections between drugs and diseases.
Knowledge graphs and graph neural networks have emerged as particularly powerful tools. They allow researchers to model complex biological relationships in a way that mirrors real cellular systems. By mapping interactions among genes, proteins, pathways, and drug targets, these models can predict new therapeutic opportunities that human analysis alone would miss.
Generative AI and large language models further accelerate hypothesis generation. By scanning decades of biomedical literature, patents, and clinical notes, these systems identify non-obvious patterns and suggest repurposing candidates with unprecedented speed. At the same time, advances in high-throughput screening, organoids, and 3D cell culture models provide more physiologically relevant environments to validate computational predictions. This combination of digital intelligence and biological realism is reshaping drug development from end to end.
FDA Momentum Signals a New Regulatory Era
Regulatory support plays a critical role in shaping any pharmaceutical market, and drug repurposing has gained meaningful traction in this area. The U.S. FDA has actively supported repurposed drugs through pathways such as the 505(b)(2) application, which allows developers to rely on existing data while submitting targeted new evidence for additional indications.
Recent approvals underscore this momentum. Tirzepatide, originally developed for type 2 diabetes, received approval for obstructive sleep apnea, highlighting how metabolic drugs can extend into entirely new therapeutic territories. Nogapendekin alfa inbakicept-pmln transitioned from non-alcoholic steatohepatitis research into primary biliary cholangitis. Even established drugs like bumetanide have found refined applications through alternative delivery methods.
These approvals do more than expand treatment options. They send a clear signal to investors and innovators that repurposing represents a viable and regulator-supported development strategy rather than a secondary or opportunistic approach.
Disease-Centric Thinking Takes the Lead
Among various repurposing strategies, disease-centric approaches dominated the market in 2024. This model starts with unmet clinical needs rather than molecular targets, making it particularly relevant for rare and neglected conditions. By integrating clinical data, genetic insights, and epidemiological trends, companies can identify candidate drugs that modulate disease-relevant pathways, even if those drugs were not designed with that condition in mind.
This approach resonates with payers and regulators alike because it focuses on patient outcomes. It also aligns well with AI-driven platforms capable of zero-shot or few-shot predictions, where models extrapolate therapeutic potential for diseases with limited historical data.
At the same time, target-centric strategies are gaining momentum. By focusing on specific molecular targets implicated across multiple diseases, researchers can systematically evaluate existing drugs that interact with those targets. This approach opens doors to cross-therapeutic repurposing, where a compound from oncology may prove effective in neurodegeneration or autoimmune disorders.
Therapeutic Boundaries Are Starting to Blur
Historically, most repurposed drugs remained within the same therapeutic area, primarily because safety, dosing, and clinical pathways were already well understood. This trend continued to dominate the market in 2024, as companies prioritized lower-risk life-cycle management strategies.
However, the market is rapidly evolving. Cross-therapeutic repurposing is gaining credibility as biological insights deepen and technology improves. Immunology drugs now intersect with oncology through combination therapies. Metabolic agents influence neurological and inflammatory pathways. Nanotechnology and advanced drug delivery systems further enhance the feasibility of repositioning drugs across therapeutic boundaries.
This shift reflects a more holistic understanding of disease biology, where rigid categorizations no longer constrain innovation.
Biologics Lead Today, but Small Molecules Are Catching Up
In 2024, biologics captured the largest share of the drug repurposing market. Their specificity, potency, and relevance to complex diseases make them attractive candidates for repositioning, especially in oncology and immunology. Strategic collaborations and advances in biologics manufacturing continue to strengthen this segment.
Yet small molecules are emerging as a powerful growth driver. Their established safety profiles, oral bioavailability, and lower manufacturing costs make them ideal candidates for rapid repurposing. Researchers increasingly explore small molecules that influence DNA repair, epigenetic regulation, and metabolic signaling, unlocking new therapeutic possibilities in cancer and rare diseases.
Regional Dynamics Reflect Local Strengths
North America remains the dominant force in the global drug repurposing market. The region benefits from advanced research infrastructure, strong venture capital ecosystems, and a regulatory environment that actively supports repurposing initiatives. Partnerships between AI startups, academic institutions, and large pharmaceutical companies accelerate innovation and commercialization.
Asia Pacific, however, represents the fastest-growing region. Countries such as China, India, South Korea, and Japan combine large patient populations with expanding pharmaceutical manufacturing capabilities. Governments increasingly promote AI-driven healthcare innovation as a strategic priority, making repurposing an attractive and cost-effective development pathway.
Europe continues to play a vital role through collaborative research initiatives and regulatory reforms. EU-funded programs and evolving pharmaceutical legislation create new opportunities for both commercial and non-profit repurposing efforts. Meanwhile, South America, the Middle East, and Africa are leveraging repurposing to strengthen local healthcare resilience, reduce import dependency, and build domestic manufacturing capabilities.
The Value Chain Reveals a Collaborative Ecosystem
Drug repurposing thrives on collaboration. From computational discovery and preclinical validation to clinical trials and patient support, the value chain spans academia, startups, pharmaceutical giants, hospitals, and regulators. AI-driven companies identify candidates, research institutions validate mechanisms, pharmaceutical firms manage development and scale, and healthcare providers deliver therapies to patients faster than ever before.
This interconnected ecosystem reduces duplication of effort and spreads risk, making innovation more sustainable over the long term.
Industry Leaders Are Betting Big on Repurposing
Major pharmaceutical companies such as AbbVie and Novartis integrate drug repurposing deeply into their R&D strategies. With vast libraries of proprietary compounds and strong investments in AI and data convergence, these organizations use repurposing to extend product life cycles, accelerate pipeline development, and address unmet medical needs.
At the same time, specialized biotech firms and AI-first startups bring agility and computational expertise to the table. Their focus on systematic drug repositioning, quantum computing applications, and advanced analytics complements the scale and clinical experience of established players.
A Market Driven by Urgency, Not Trend
What ultimately sets the drug repurposing market apart is urgency. Patients cannot wait for decade-long development cycles. Healthcare systems cannot absorb unsustainable costs. Regulators cannot ignore the potential of faster, safer innovation. Drug repurposing responds to these realities with a practical, science-driven solution.
As technology continues to evolve and biological understanding deepens, the line between old and new drugs will blur further. The question is no longer whether existing drugs can deliver new cures, but how quickly the industry can unlock their full potential.
The coming decade will define drug repurposing not as an alternative strategy, but as a central pillar of global drug development.
Access our exclusive, data-rich dashboard dedicated to the pharmaceutical industry – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout Drug Repurposing Market Report Now at: https://www.towardshealthcare.com/checkout/6424
Become a valued research partner with us - https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest