For decades, small-molecule drugs dominated medicine shelves and therapeutic strategies. That dominance has shifted. Antibodies drug, once considered complex, expensive, and niche, have moved into the center of global healthcare. What started as a scientific experiment in immunology has evolved into one of the most influential and commercially significant segments of the pharmaceutical industry.
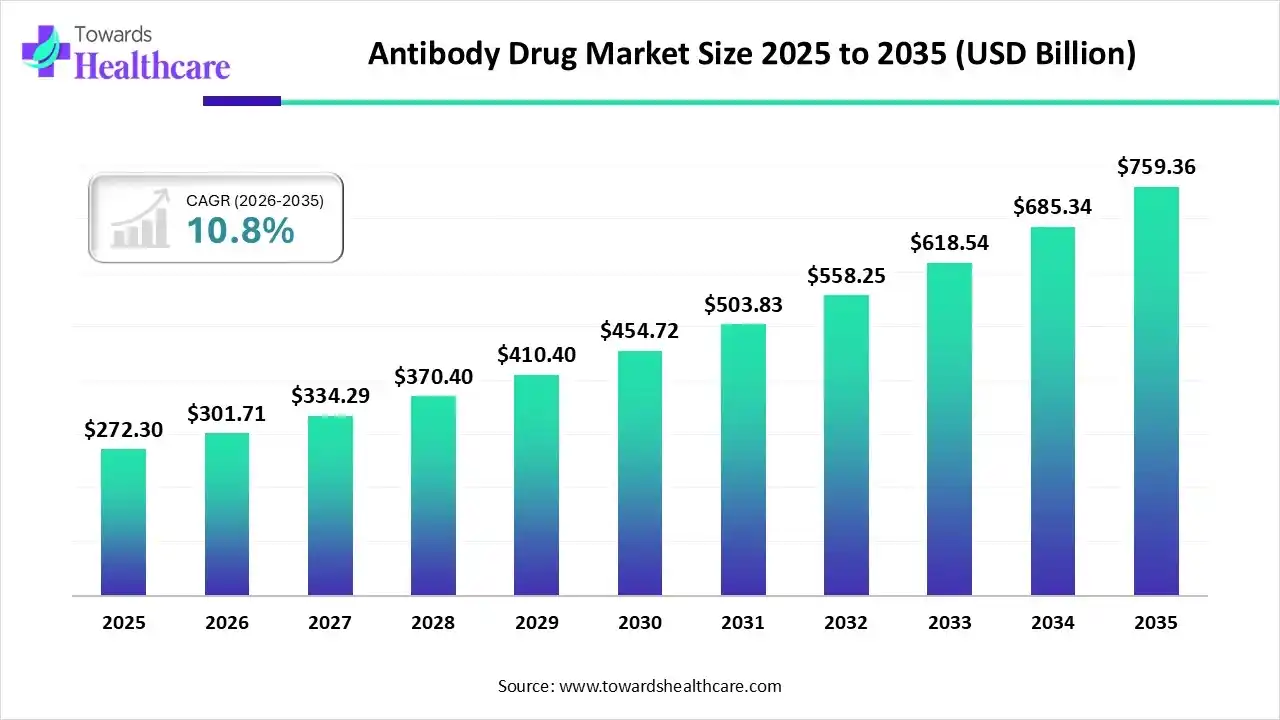
By 2025, the global antibodies drug market reached USD 272.3 billion, and the trajectory ahead suggests something far bigger than incremental growth. By 2035, the market is expected to approach USD 759.36 billion, expanding at a CAGR of 10.8 percent.
𝐃𝐨𝐰𝐧𝐥𝐨𝐚𝐝 𝐅𝐫𝐞𝐞 𝐒𝐚𝐦𝐩𝐥𝐞:
https://www.towardshealthcare.com/download-sample/5009
This growth is not accidental. It reflects deep structural changes in disease patterns, medical science, healthcare economics, and patient expectations. Antibody drugs are no longer viewed as last-resort therapies. They are increasingly positioned as first-line, precision-driven solutions for diseases that once had limited or ineffective treatment options.
Why Antibody Drugs Matter More Than Ever
Antibody drugs sit at the intersection of biology and engineering. Unlike traditional chemical drugs that broadly affect biological pathways, antibodies are designed to recognize specific targets with remarkable precision. This specificity allows physicians to intervene directly in disease mechanisms rather than simply managing symptoms.
Cancer, autoimmune disorders, inflammatory diseases, and rare genetic conditions have driven much of this shift. As these diseases grow more prevalent and complex, healthcare systems demand therapies that offer higher efficacy, better safety profiles, and long-term disease control. Antibody drugs meet those expectations more consistently than many conventional therapies.
The rising confidence of regulators also signals maturity. More than 100 antibody-based therapies have already received regulatory approval, and biologics now represent roughly one-fifth of all new drug approvals annually. This trend reflects scientific confidence as well as regulatory comfort with antibody platforms.
From a Single Breakthrough to a Global Industry
The story of antibody drugs began modestly. In 1986, Orthoclone OKT3 became the first licensed monoclonal antibody, approved to prevent kidney transplant rejection. It was effective but imperfect. Patients experienced immune reactions because the antibody originated from murine sources. Those limitations shaped decades of innovation.
Today’s antibody drugs look very different. Human and humanized monoclonal antibodies dominate the market, significantly reducing immunogenicity while improving pharmacokinetics and therapeutic durability. The transition from murine to human antibodies reflects one of the most important technological evolutions in modern medicine.
That evolution also explains why human monoclonal antibodies now hold the dominant share by source and continue to grow at the fastest rate. Pharmaceutical companies no longer see antibody development as experimental. They see it as foundational.
Monoclonal Antibodies Still Lead, but the Next Wave Has Arrived
Monoclonal antibodies remain the backbone of the market. Their applications stretch from oncology and rheumatology to infectious diseases and cardiology. Physicians value their predictability, regulators trust their development pathways, and patients benefit from their targeted action.
However, the market’s most dynamic momentum comes from antibody-drug conjugates. ADCs combine the targeting ability of antibodies with the potency of cytotoxic drugs. This dual mechanism allows clinicians to deliver highly potent payloads directly to diseased cells while sparing healthy tissue.
This approach changes the therapeutic equation, particularly in oncology. ADCs address one of cancer treatment’s oldest challenges: how to kill tumor cells without overwhelming systemic toxicity. With more ADCs advancing through clinical trials and regulatory frameworks becoming increasingly supportive, this segment is positioned for rapid expansion.
Cancer Remains the Epicenter of Demand
Cancer continues to dominate the antibody drug market, not because of hype, but because of unmet medical need. Rising incidence rates, aging populations, and improved diagnostic capabilities ensure sustained demand. More importantly, antibody therapies have demonstrated tangible improvements in survival rates and quality of life across multiple cancer types.
Lung cancer, breast cancer, colorectal cancer, and hematological malignancies have seen particularly strong adoption of antibody-based treatments. These therapies not only target tumor antigens but also enhance immune system activity, shifting cancer treatment toward long-term disease management rather than short-term remission.
The market’s fastest-growing indication, however, lies in autoimmune diseases. Conditions such as psoriasis, rheumatoid arthritis, multiple sclerosis, and inflammatory bowel disease demand long-term therapy. Antibody drugs address the underlying immune dysregulation driving these diseases, offering sustained control rather than episodic relief.
Distribution Channels Are Quietly Transforming Access
Hospital pharmacies have traditionally dominated antibody drug distribution, and that dominance remains intact. Many antibody therapies require controlled administration, monitoring, and specialized handling. Hospitals offer the infrastructure necessary to support these requirements.
At the same time, online pharmacies represent a structural shift rather than a short-term trend. As formulations improve and subcutaneous administration becomes more common, patients increasingly expect convenience alongside efficacy. Digital healthcare platforms, home delivery, and online consultations are reshaping how antibody drugs reach patients, particularly in chronic disease management.
This shift also reflects broader consumer behavior changes in healthcare. Patients want autonomy, transparency, and flexibility. Distribution models that align with those expectations will shape market dynamics over the next decade.
North America Sets the Pace, Asia-Pacific Sets the Speed
North America continues to lead the global antibody drug market due to advanced healthcare infrastructure, high healthcare spending, and strong R&D ecosystems. The region’s aging population and high cancer burden sustain long-term demand, while early adoption of innovative therapies accelerates commercialization.
Asia-Pacific tells a different but equally compelling story. Rapid urbanization, growing middle-class populations, and increasing chronic disease prevalence create a fertile environment for antibody drug adoption. Governments across China, India, and Japan actively support biopharmaceutical manufacturing, clinical research, and regulatory modernization.
China’s accelerating approvals, India’s manufacturing incentives, and Japan’s long-term drug development strategies collectively position Asia-Pacific as the fastest-growing region in the market. This growth reflects both domestic demand and global supply chain integration.
Europe’s Quiet Strength in Antibody Innovation
Europe rarely dominates headlines, yet it remains a critical pillar of antibody drug development. Strong academic research, established pharmaceutical players, and supportive regulatory frameworks allow Europe to punch above its weight in innovation.
Countries such as Germany, the UK, and France play a central role in oncology and immunology research. The region’s focus on early diagnosis, rare diseases, and advanced biologics sustains steady demand for antibody therapies. Recent regulatory approvals in the UK further demonstrate Europe’s commitment to accelerating patient access without compromising safety.
Latin America, Middle East, and Africa Are No Longer Peripheral
Emerging markets increasingly influence global antibody drug strategies. Latin America’s growing biologics demand, combined with expanding biosimilar development, improves accessibility and affordability. Brazil, in particular, stands out for its manufacturing capabilities and rising R&D investments.
In the Middle East and Africa, government-led healthcare reforms and rising awareness of rare and genetic diseases reshape market potential. Countries such as the UAE are modernizing pharmaceutical regulation and infrastructure, signaling long-term commitment to advanced therapies.
These regions may not lead in absolute market size today, but they represent critical growth engines for the next decade.
AI Is Changing How Antibody Drugs Are Discovered and Made
Artificial intelligence no longer sits on the sidelines of drug development. In antibody research, AI and machine learning accelerate discovery, optimize molecular design, and reduce development timelines. Algorithms predict antigen-antibody interactions, identify favorable mutations, and simulate patient-specific responses.
Manufacturing also benefits. AI-driven process optimization improves yield, reduces errors, and enhances quality control at scale. These efficiencies matter in a market where production complexity and cost remain persistent challenges.
As AI tools mature, they will not replace human expertise but amplify it. The result will be faster innovation cycles and more personalized antibody therapies.
Biosimilars Are Reshaping the Cost Equation
Cost remains one of the antibody drug market’s most significant barriers. Monoclonal antibodies are expensive to develop, manufacture, and distribute. For many healthcare systems, especially in low- and middle-income countries, affordability limits access.
Biosimilar monoclonal antibodies address this challenge directly. Priced significantly lower than originator biologics, biosimilars expand patient access while maintaining clinical efficacy. Regulatory agencies now possess clearer frameworks for biosimilar evaluation, accelerating market entry.
As more blockbuster antibody drugs face patent expiration, biosimilars will play an increasingly central role in balancing innovation with sustainability.
Regulatory Rigor Remains a Double-Edged Sword
Strict regulatory requirements protect patient safety, but they also slow market entry and increase development risk. Clinical trial failures, delayed approvals, and post-marketing surveillance obligations create financial and operational pressure for developers.
Despite these challenges, regulatory rigor ultimately strengthens the market. Clear standards foster trust among clinicians, patients, and payers. Over time, this trust translates into broader adoption and sustained market growth.
Functional Limitations Still Demand Scientific Attention
Antibody drugs are powerful, but not perfect. Issues such as limited tissue penetration, suboptimal pharmacokinetics, and immune system interactions continue to challenge researchers. Early-generation antibodies highlighted these limitations, and while modern designs have addressed many concerns, further refinement remains essential.
Ongoing research into novel linkers, improved payloads, and next-generation antibody formats demonstrates the industry’s commitment to overcoming these barriers rather than accepting them.
A Market Defined by Long-Term Commitment
The antibody drug market does not thrive on short-term trends. It grows through sustained investment, scientific discipline, and clinical validation. Companies currently support hundreds of monoclonal antibody programs across early and late-stage development, with oncology dominating the pipeline.
This long-term commitment signals confidence not only in market returns but in the therapeutic value antibodies deliver. Industry leaders increasingly view antibody platforms as strategic assets rather than individual products.
The Road Ahead for Antibody Drugs
Antibody drugs have moved beyond novelty. They represent a mature, resilient, and innovation-driven market segment that continues to redefine modern medicine. Growth through 2035 will depend on scientific breakthroughs, regulatory agility, manufacturing efficiency, and equitable access.
What makes this market compelling is not just its size, but its purpose. Antibody drugs address diseases that shape human longevity, quality of life, and healthcare sustainability. As science advances and systems adapt, antibody therapies will likely become even more integral to global health strategies.