For more than four decades, western blotting has held a unique and often underestimated position in life science research and clinical diagnostics. Many technologies have come and gone, replaced by faster, flashier, or more automated alternatives. Western blotting, however, has persisted. Not because it is old, but because it remains fundamentally reliable. As we move toward 2035, the global western blotting market is no longer just surviving; it is evolving with purpose, discipline, and renewed relevance.
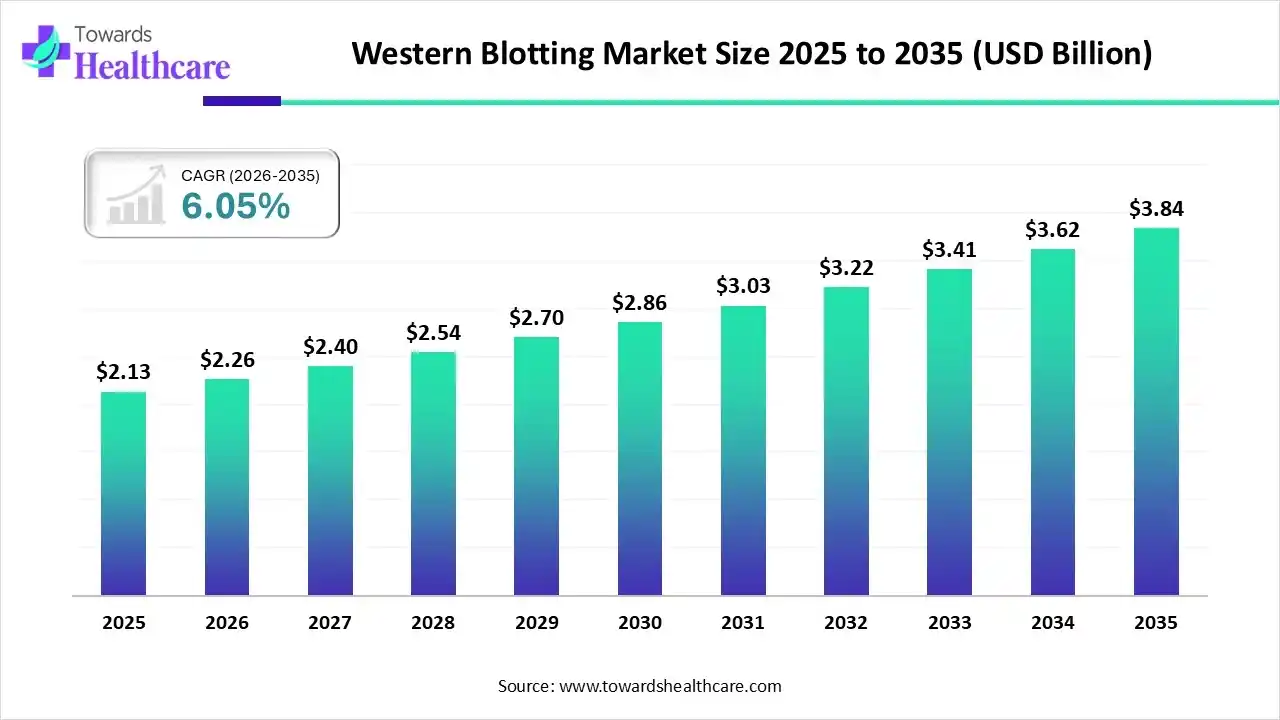
In 2025, the global western blotting market stood at USD 2.13 billion. By 2026, it reached USD 2.26 billion, and current projections place the market at approximately USD 3.84 billion by 2035. A steady compound annual growth rate of 6.05% over nearly a decade signals something important. This is not a hype-driven expansion. It reflects sustained scientific demand, real-world clinical utility, and structural shifts in biomedical research, diagnostics, and personalized medicine.
𝐃𝐨𝐰𝐧𝐥𝐨𝐚𝐝 𝐅𝐫𝐞𝐞 𝐒𝐚𝐦𝐩𝐥𝐞:
https://www.towardshealthcare.com/download-sample/5768
Western blotting has entered a phase where refinement matters more than reinvention. The coming decade will not be defined by whether the technique survives, but by how intelligently it adapts to new scientific and healthcare realities.
Why Western Blotting Still Matters in Modern Science
Western blotting continues to play a central role because it answers questions that many advanced technologies still struggle to resolve with the same clarity. Researchers rely on it to detect specific proteins, confirm molecular weight, validate antibody specificity, and semi-quantify protein expression. In disease research, especially in oncology, infectious diseases, and autoimmune disorders, western blotting remains a gold-standard confirmatory tool.
What truly differentiates western blotting is trust. Scientists trust the bands they see. Clinicians trust the confirmatory results. Regulators trust the method because its strengths and limitations are well understood. In a scientific ecosystem increasingly shaped by complex algorithms and black-box analytics, western blotting offers transparency. You see the result. You interpret it. You validate it.
This trust has become even more valuable as the demand for reproducibility in research intensifies. Over the past decade, concerns around irreproducible data have pushed laboratories back toward techniques that deliver consistency. Western blotting, when performed correctly, meets that expectation.
The Market’s Growth Story Is Rooted in Disease Burden
The expansion of the western blotting market closely mirrors global disease trends. Chronic disorders, infectious diseases, and genetic conditions are rising across regions. HIV remains a major driver, as western blotting continues to serve as a critical confirmatory test for HIV diagnosis in many healthcare systems. The growing global population living with HIV, combined with sustained public health investment, directly supports continued demand.
Beyond HIV, the rising incidence of cancer, neurodegenerative disorders, metabolic diseases, and autoimmune conditions has expanded the scope of protein-based diagnostics and research. Proteins sit at the center of disease pathways. Understanding them requires tools that can identify, separate, and validate protein expression with confidence. Western blotting fulfills that role.
As healthcare systems increasingly emphasize early diagnosis, disease monitoring, and precision medicine, protein-level insights have become indispensable. Genomics may tell us what could happen, but proteomics tells us what is happening now. This distinction will continue to anchor western blotting firmly in both research and diagnostics through 2035.
Research and Development Keep the Market Alive
The western blotting market does not grow in isolation. It grows because research grows. Global investment in biomedical research, proteomics, and translational science continues to expand, particularly in North America and Asia-Pacific. Academic institutions, government-funded laboratories, and private research organizations consistently rely on western blotting as part of their experimental workflows.
Pharmaceutical and biotechnology companies also play a crucial role. Drug discovery and development depend heavily on protein validation at multiple stages, from target identification to mechanism-of-action studies and biomarker validation. Western blotting remains one of the most widely accepted methods for confirming protein expression changes during preclinical and early clinical research.
As biologics, biosimilars, and targeted therapies dominate drug pipelines, the relevance of protein-focused analytical tools increases further. Western blotting does not compete with high-throughput technologies in scale, but it complements them in validation, which is often where regulatory and scientific confidence is built.
Consumables Quietly Drive Revenue Stability
One of the most overlooked aspects of the western blotting market is the dominance of consumables. Reagents, kits, membranes, antibodies, and buffers account for a significant share of global revenue, and they do so consistently. Instruments may represent technological progress, but consumables represent repeat demand.
Every western blot experiment requires consumables. They are not optional. This creates a stable, recurring revenue stream for manufacturers and suppliers. The availability of customized reagents and application-specific kits has further strengthened this segment. Laboratories increasingly seek standardized solutions that reduce variability and save time, and consumable providers have responded accordingly.
By 2035, consumables will continue to anchor the market, even as instruments evolve. Their dominance reflects the reality of laboratory workflows, where reliability, availability, and consistency matter more than novelty.
Instruments Are Changing How Researchers Work
While consumables dominate revenue, instruments represent the future of workflow efficiency. Over the past few years, western blotting instruments have become more compact, automated, and user-friendly. Imaging systems now offer higher sensitivity, improved dynamic range, and digital connectivity. Blotting systems have evolved to reduce processing time and minimize manual intervention.
The integration of artificial intelligence and machine learning into imaging and analysis software marks a turning point. These tools reduce human bias in band detection, normalize results more accurately, and accelerate data interpretation. For laboratories facing staff shortages or skill gaps, automation offers a practical solution.
By 2035, instruments will no longer be judged solely on performance. Ease of use, reproducibility, software intelligence, and integration with laboratory information systems will define purchasing decisions. This shift will particularly benefit diagnostic centers and high-throughput research environments.
Diagnostics Are Redefining the Market’s Direction
Historically, biomedical and biochemical research has dominated western blotting applications. That remains true today. However, disease diagnostics now represent the fastest-growing application segment. This shift reflects broader changes in healthcare delivery.
As populations age and chronic diseases rise, healthcare systems are moving diagnostics closer to patients. Western blotting, particularly when automated and standardized, fits well into this model for confirmatory testing. Diagnostic centers with appropriate infrastructure increasingly adopt advanced blotting systems to support infectious disease testing, autoimmune disorder diagnosis, and complex protein-based assays.
The demand for accuracy in diagnostics leaves little room for ambiguity. Western blotting’s ability to confirm results from screening tests reinforces its role as a diagnostic backbone rather than a legacy technique.
Academic Institutions Still Lead, but Others Are Catching Up
Academic and research institutes remain the largest end users of western blotting technologies. This dominance stems from the sheer volume of experiments conducted and the central role of protein analysis in basic and applied research. Funding from governments and private organizations sustains this demand.
However, diagnostic centers are emerging as the fastest-growing end-use segment. These facilities benefit from improved infrastructure, trained personnel, and growing patient volumes. As automated systems become more accessible, diagnostic centers will increasingly rely on western blotting for complex confirmatory testing.
Pharmaceutical and biotechnology companies occupy a strategic middle ground. Their demand fluctuates with pipeline activity, but their influence on innovation and standard-setting remains significant.
North America Sets the Pace, Asia-Pacific Sets the Speed
North America continues to dominate the western blotting market, accounting for nearly half of global revenue. Strong research funding, advanced infrastructure, and the presence of major industry players support this leadership. The region’s emphasis on proteomics, precision medicine, and translational research ensures sustained demand.
Asia-Pacific, however, represents the market’s fastest-growing region. Rising disease prevalence, expanding biotech sectors, and increasing government investment in research drive this growth. Countries such as China, Japan, and India are building long-term capabilities in proteomics and diagnostics, which will significantly shape global demand by 2035.
Europe maintains steady growth, supported by favorable government policies, public-private partnerships, and a strong focus on early disease detection. Meanwhile, the Middle East and Africa are emerging markets, where investments in genomics, proteomics, and healthcare infrastructure are gradually unlocking new opportunities.
Artificial Intelligence Is Not Replacing Western Blotting
There is a misconception that artificial intelligence will replace traditional laboratory techniques. In reality, AI is strengthening western blotting rather than threatening it. By automating image analysis, reducing human error, and improving reproducibility, AI enhances the technique’s credibility.
AI-driven software can detect subtle band variations, analyze complex patterns, and standardize interpretation across laboratories. This capability becomes particularly valuable in large-scale studies and diagnostic environments, where consistency is critical.
By 2035, AI will not redefine what western blotting is. It will redefine how efficiently and reliably it is performed.
Skill Gaps Remain a Real Constraint
Despite its growth, the western blotting market faces challenges. The technique remains labor-intensive and demands technical expertise. In many low- and middle-income regions, a shortage of skilled professionals limits adoption. Training requirements and funding constraints slow market penetration.
Automation and standardized kits help mitigate this issue, but they do not eliminate the need for fundamental scientific understanding. Addressing this gap will require coordinated efforts in education, training, and infrastructure development.
Miniaturization Signals the Next Evolution
Looking ahead, miniaturization represents one of the most promising opportunities in western blotting. Microfluidic platforms enable faster analysis, reduced reagent consumption, higher sensitivity, and multiplexing capabilities. These advancements address many limitations of conventional blotting, including sample wastage and long processing times.
Miniaturized systems also align with broader laboratory trends toward efficiency and sustainability. As research budgets tighten and environmental concerns grow, technologies that deliver more with less will gain traction.
By 2035, microfluidic western blotting may not replace traditional systems entirely, but it will expand the technique’s reach into new applications and settings.
The Road to 2035 Looks Steady, Not Speculative
The western blotting market’s projected growth to USD 3.84 billion by 2035 reflects maturity rather than speculation. This is a market built on trust, necessity, and continuous incremental improvement. It does not rely on disruptive hype. It relies on scientific demand.
Western blotting has proven its resilience by adapting without losing its core identity. It remains a technique that scientists understand, regulators respect, and clinicians rely on. As proteomics, diagnostics, and personalized medicine continue to evolve, western blotting will remain quietly essential.
The next decade will not be about whether western blotting survives. It will be about how confidently it continues to support science, medicine, and human health in a world that demands accuracy more than ever.
Access our exclusive, data-rich dashboard dedicated to the biotechnology industry – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout Western Blotting Market Report Now at: https://www.towardshealthcare.com/checkout/5768
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest