The IV bags market is no longer a quiet, background segment of healthcare. For decades, intravenous bags were treated as basic consumables, essential, reliable, and largely unchanged. Today, that assumption no longer holds. The market is entering a phase of structural transformation shaped by sustainability demands, digital intelligence, supply chain resilience, and shifting care models. From hospital ICUs to home-based infusion centers, IV bags are becoming smarter, safer, and more strategically important than ever before.
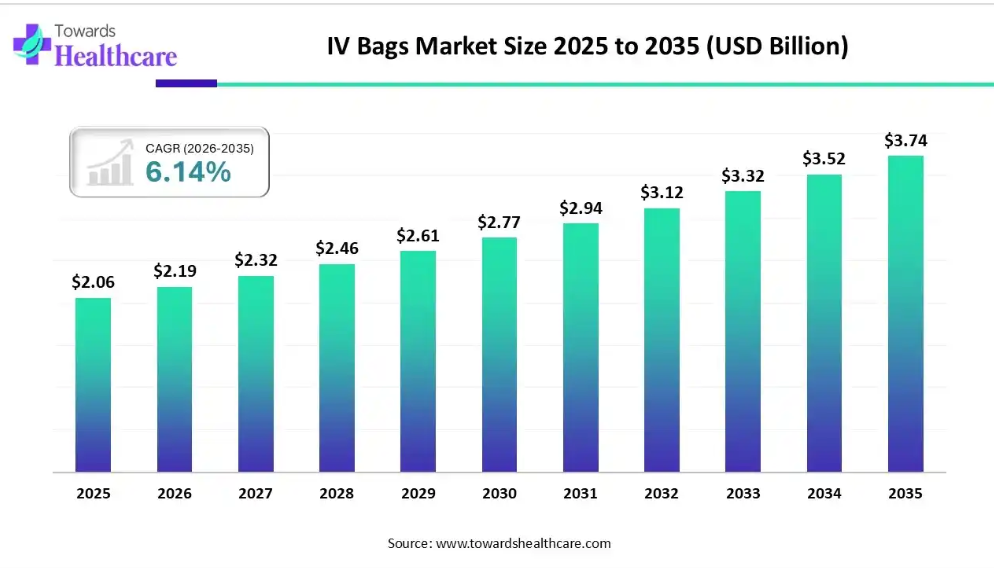
With the market valued at approximately USD 2.19 billion in 2026 and projected to reach USD 3.74 billion by 2035 at a CAGR of 6.14%, growth is steady but meaningful. More importantly, this growth reflects deep changes in how healthcare systems function, how patients receive treatment, and how manufacturers respond to regulatory, environmental, and technological pressures. After more than a decade of observing medical device markets, it is clear that IV bags are no longer just containers for fluids; they are becoming part of a broader infusion ecosystem.
Download Free Sample: https://www.towardshealthcare.com/download-sample/5379
Why IV Bags Still Matter More Than Most People Realize
IV therapy remains one of the most widely used clinical interventions worldwide. Whether treating dehydration, managing electrolyte imbalance, supporting chemotherapy, or delivering parenteral nutrition, IV bags sit at the center of patient care. A significant majority of hospitalized patients receive IV fluids at some point during treatment, and this dependency increases sharply in ICUs, oncology wards, surgical recovery units, and emergency care.
What makes IV bags particularly critical is their universality. Unlike specialized medical devices used by specific departments, IV bags cross nearly every clinical boundary. They are used in tertiary hospitals, rural clinics, ambulances, home care settings, and disaster response units. As healthcare systems expand access and decentralize care, the demand for reliable, safe, and adaptable IV bags continues to rise.
A Market Defined by Scale, Stability, and Subtle Innovation
Unlike high-risk implantable devices, IV bags operate in a high-volume, low-margin environment. This reality has historically limited radical innovation. However, recent disruptions, most notably global supply shortages and environmental regulations, have forced manufacturers to rethink design, materials, and production strategies.
North America continues to dominate the global market, driven by advanced healthcare infrastructure, high hospitalization rates, and strict quality standards. At the same time, Asia-Pacific has emerged as the fastest-growing region, fueled by expanding healthcare access, rising chronic disease prevalence, and strong domestic manufacturing capabilities in countries such as India and China.
This dual-market dynamic has created an interesting contrast. Mature markets prioritize quality, safety, and compliance, while emerging markets focus on scale, affordability, and capacity expansion. Together, they are reshaping how IV bags are produced, distributed, and improved.
Material Choices Are No Longer Just a Cost Decision
Polyvinyl chloride has long dominated the IV bags market due to its flexibility, transparency, durability, and cost efficiency. PVC-based IV bags remain widely used for parenteral nutrition, hydration solutions, and antibiotic delivery. Their ability to withstand sterilization and maintain structural integrity under varying conditions has made them the industry standard for decades.
However, the market is clearly shifting. Environmental sustainability and patient safety concerns have accelerated interest in non-PVC alternatives, particularly polyethylene and polypropylene. These materials eliminate exposure to phthalates, reduce chlorine-related byproducts, and offer better recyclability. Polyethylene, in particular, has gained traction due to its chemical resistance, durability, and compatibility with gamma irradiation.
This transition is not abrupt, but it is unmistakable. Hospitals, regulators, and manufacturers increasingly view material selection as a long-term risk management decision rather than a short-term cost calculation. Over time, this shift is expected to redefine competitive differentiation in the IV bags market.
Size and Simplicity Continue to Drive Clinical Preference
Despite technological advances, clinical workflows still value simplicity. This explains why IV bags in the 250 ml to 500 ml range continue to dominate global usage. These sizes align well with common infusion rates, are easy to handle, and reduce storage and transportation challenges. In high-volume hospital environments, operational efficiency often matters as much as clinical performance.
Similarly, single-chamber IV bags maintain their leading position due to ease of use, reduced contamination risk, and straightforward administration. While multi-chamber bags offer advantages for complex formulations, single-chamber designs remain the preferred choice for most routine therapies.
These preferences highlight an important insight: innovation in the IV bags market must enhance usability without adding complexity. Products that disrupt established clinical routines without clear benefits face slow adoption, regardless of technological sophistication.
Parenteral Nutrition Keeps the Market Anchored
Among all applications, parenteral nutrition remains the most dominant driver of IV bag demand. Patients with gastrointestinal disorders, post-surgical complications, malnutrition, or advanced cancer often rely on IV-based nutrition for survival and recovery. As global cancer rates rise and surgical volumes increase, the need for safe and reliable parenteral nutrition delivery continues to grow.
This segment places particularly high demands on IV bag quality. Compatibility with complex nutrient mixtures, resistance to chemical interaction, and strict sterility requirements make material integrity and manufacturing precision critical. As a result, parenteral nutrition applications often act as a benchmark for product innovation and regulatory scrutiny.
The Quiet Rise of Smart IV Bags
Artificial intelligence and sensor-based monitoring are beginning to influence even the most traditional medical consumables. In the IV bags market, AI integration is emerging as a practical solution to long-standing challenges rather than a futuristic concept.
Smart monitoring systems can track fluid levels in real time, alert nursing staff before bags run empty, and reduce the risk of dosing errors. AI-driven visual inspection systems enhance quality control during manufacturing by identifying defects that human inspection might miss. Robotics improves consistency, reduces contamination risk, and increases throughput in high-volume production environments.
While fully autonomous IV bag systems are not yet mainstream, incremental digital integration is already improving safety, efficiency, and reliability across the value chain. Over the next decade, these technologies are expected to move from pilot projects to standard practice, particularly in high-resource healthcare systems.
Supply Chain Lessons That Changed the Industry
The COVID-19 pandemic exposed a critical vulnerability in global healthcare supply chains, and IV bags were among the most affected products. Sudden surges in demand, factory shutdowns, logistical bottlenecks, and geopolitical disruptions led to shortages that directly impacted patient care.
In response, governments and private investors have committed substantial resources to strengthening local manufacturing capacity and supply chain resilience. Large-scale investments in IV fluid and IV bag production facilities across North America, Asia, the Middle East, and Australia reflect a strategic shift toward self-sufficiency and redundancy.
These investments are not temporary reactions. They represent a long-term rethinking of how essential medical supplies are produced and distributed. For the IV bags market, this has translated into capacity expansion, technology upgrades, and a renewed focus on risk mitigation.
Environmental Pressure Is Reshaping Product Strategy
Sustainability is no longer a secondary consideration in healthcare manufacturing. Regulatory restrictions on single-use plastics, combined with growing awareness of medical waste, have placed IV bag manufacturers under increasing pressure to innovate responsibly.
Non-PVC materials, recyclable designs, and take-back or recycling programs are gaining attention. Initiatives that divert used IV bags from landfills and repurpose materials for non-medical applications signal a shift toward circular economy principles. While these efforts are still evolving, they are changing how manufacturers approach product lifecycle management.
Importantly, sustainability in IV bags is closely tied to patient safety. Eliminating harmful plasticizers, reducing chemical exposure, and ensuring material stability align environmental goals with clinical outcomes. This alignment strengthens the business case for sustainable innovation.
Regional Dynamics Reveal Different Growth Stories
North America’s dominance reflects mature healthcare systems with high utilization rates, advanced infusion practices, and strong regulatory oversight. Frequent IV therapy usage, particularly in hospitals and home care settings, continues to sustain demand.
Asia-Pacific tells a different story, driven by demographic change and infrastructure expansion. Aging populations, rising cancer and gastrointestinal disease rates, and government-backed healthcare initiatives are accelerating IV bag adoption. Countries like India and China are also emerging as global manufacturing hubs, supplying both domestic and international markets.
Europe balances technological sophistication with sustainability leadership, while regions such as Latin America and the Middle East are experiencing steady growth fueled by healthcare expansion, medical tourism, and government investment. Together, these regional patterns underscore the global relevance of the IV bags market.
Competition Built on Reliability, Not Hype
The IV bags market is characterized by established players with strong manufacturing expertise and regulatory compliance capabilities. Competition revolves around quality consistency, supply reliability, material innovation, and long-term customer relationships rather than aggressive marketing or rapid product turnover.
Recent product launches and regulatory approvals demonstrate a clear emphasis on ready-to-use formulations, latex-free designs, and non-PVC containers. These developments reflect a cautious but deliberate approach to innovation, one that prioritizes safety, compliance, and clinical acceptance over novelty.
Looking Ahead: Evolution Without Disruption
The future of the IV bags market will not be defined by dramatic breakthroughs but by continuous, thoughtful improvement. Smarter monitoring, safer materials, more resilient supply chains, and environmentally responsible practices will gradually reshape the industry.
What makes this market particularly interesting is its stability. Demand is not speculative; it is rooted in fundamental healthcare needs. At the same time, the expectations placed on IV bags are rising. Healthcare systems now expect these products to support digital workflows, sustainability goals, and patient safety standards simultaneously.
From the perspective of long-term market observation, the IV bags industry is entering a new era, not because it is reinventing itself overnight, but because it is finally receiving the strategic attention it deserves. In a healthcare landscape increasingly defined by complexity and accountability, even the simplest tools must evolve. IV bags are doing exactly that, quietly but decisively.
Access our exclusive, data-rich dashboard dedicated to the therapeutics area sector — built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout IV bags Market Report Now at: https://www.towardshealthcare.com/checkout/5379
Become a valued research partner with us — https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region — +44 778 256 0738
North America Region — +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Pinterest