I have watched the pharmaceutical industry evolve for more than a decade, and I can say with confidence that no shift has been as decisive as the rise of biologics. We are no longer merely improving drugs; we are redesigning how medicine works at its core. Biologics have moved from being niche, high-complexity therapies to becoming the backbone of modern treatment strategies.
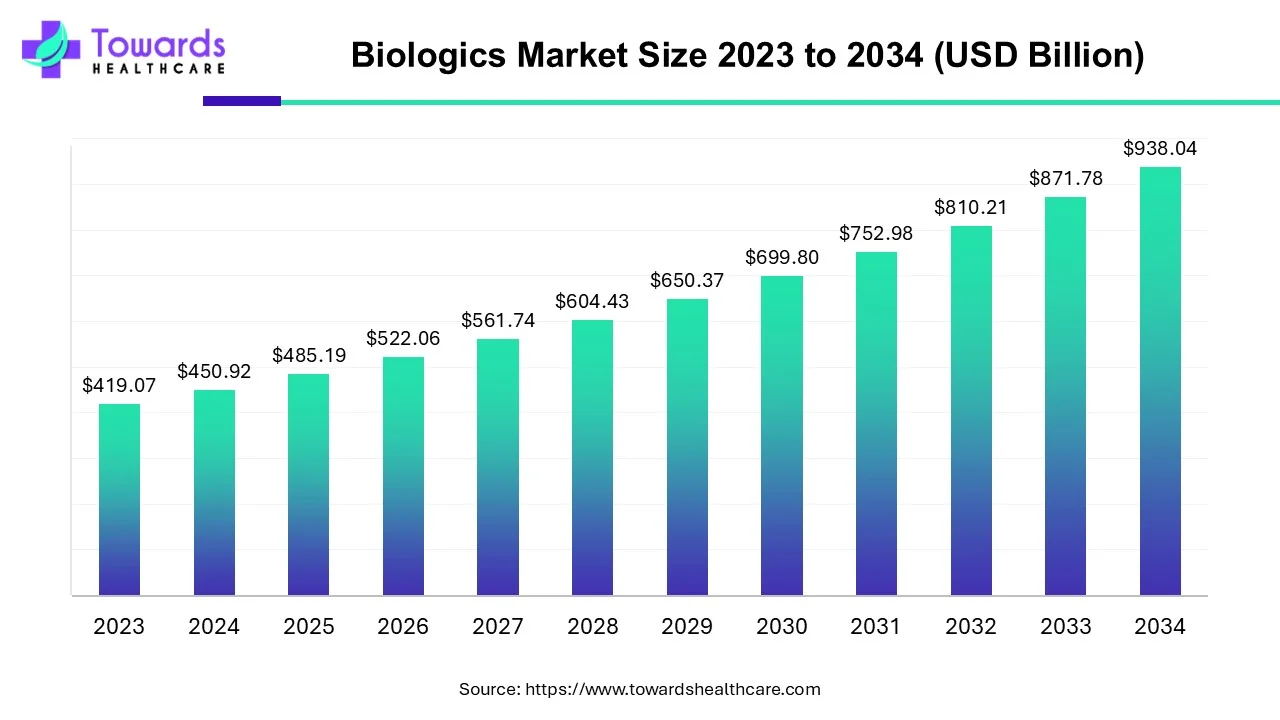
By 2026, the global biologics market is expected to surpass USD 522 billion, and projections suggest it could approach USD 1 trillion by 2035, growing at a steady and resilient pace. This growth does not come from hype. It comes from real clinical need, scientific maturity, and the hard reality that conventional small-molecule drugs often fall short against complex, chronic diseases.
Download Free Sample: https://www.towardshealthcare.com/download-sample/5108
What makes this moment remarkable is that biologics are no longer viewed as experimental breakthroughs. They now represent standard-of-care solutions across oncology, autoimmune disorders, hematology, and rare diseases. Healthcare systems, regulators, manufacturers, and investors have aligned around the same conclusion: the future of medicine is biological.
From Chemicals to Cells: A Fundamental Shift in Treatment Logic
Traditional drugs typically rely on chemical synthesis and broad mechanisms of action. They often affect multiple pathways, which leads to side effects and limited precision. Biologics operate on a different philosophy. Scientists design them from living systems or their components, including proteins, cells, and nucleic acids. These therapies interact with specific molecular targets, often mimicking natural processes inside the human body. That precision explains why monoclonal antibodies, gene-based therapies, and RNA-targeting treatments now command attention.
This shift from chemical intervention to biological modulation changes not just treatment outcomes but the entire value chain of healthcare. Development cycles now revolve around genetic data, protein engineering, and cell-line optimization. Manufacturing facilities resemble high-tech bioprocessing labs rather than traditional chemical plants. Regulatory pathways have also evolved, recognizing the complexity and variability inherent in biological systems.
Cancer Care Redefined by Precision
Oncology continues to dominate the biologics landscape, and the reason is straightforward. Cancer is not one disease; it is a spectrum of molecular abnormalities. Biologics, particularly monoclonal antibodies and immunotherapies, match this complexity with targeted action. Instead of indiscriminately killing dividing cells, these treatments identify specific antigens or immune checkpoints. They enhance survival rates while reducing the collateral damage that once defined chemotherapy.
The global cancer burden remains immense, with millions of new cases each year. That demand fuels continuous innovation in antibody engineering, bispecific formats, and antibody-drug conjugates. At the same time, RNA-based therapies and personalized vaccines are moving from experimental stages into structured pipelines. Oncology has become the proving ground for nearly every major biologics platform, and success here often opens doors in other therapeutic areas.
Chronic Diseases Keep the Growth Engine Running
Beyond cancer, chronic noncommunicable diseases create a persistent and expanding need for advanced therapies. Cardiovascular disorders, autoimmune conditions, diabetes-related complications, and chronic respiratory diseases affect a vast portion of the global population. These conditions require long-term management, and biologics offer mechanisms that align well with sustained therapy. They modulate immune responses, block inflammatory pathways, or replace deficient proteins in ways that conventional drugs cannot easily replicate.
As patient awareness rises and diagnostic tools improve, demand for targeted treatments increases. Patients now ask for therapies that not only control symptoms but address underlying mechanisms. Biologics meet that expectation, and this alignment between clinical need and technological capability drives durable market expansion.
Hematology and Rare Diseases Step Into the Spotlight
While oncology remains dominant, hematological disorders represent one of the fastest-growing segments. Genetic blood disorders, hemophilia, and certain anemias benefit from biologics that correct or compensate for molecular defects. Advances in recombinant proteins and gene-targeting approaches make it possible to move closer to root-cause treatment rather than symptomatic relief.
Rare diseases also gain attention. Although each condition affects a small population, collectively they represent a substantial unmet need. Biologics, especially gene therapies and RNA-based modalities, provide platforms that can be adapted for ultra-specific targets. Regulatory agencies increasingly support orphan indications, creating an environment where innovation and patient impact align with commercial viability.
The Manufacturing Evolution: Control Versus Collaboration
Manufacturing biologics remains one of the industry’s most complex undertakings. Companies historically preferred in-house production to maintain tight control over quality, intellectual property, and supply continuity. Advanced bioreactors, cell culture optimization, and purification technologies have become strategic assets. For large biopharma players, owning these capabilities provides competitive advantage and supply security.
At the same time, outsourcing has accelerated. Smaller and mid-sized biotech firms often lack the capital and infrastructure to build full-scale facilities. They rely on contract development and manufacturing organizations that offer specialized expertise in cell-line development, process scale-up, and regulatory compliance. This partnership model spreads risk and speeds time to market. Over the next decade, we will likely see a hybrid ecosystem where strategic in-house capacity coexists with a sophisticated global network of CDMOs.
Microbial Roots and Mammalian Sophistication
From a production standpoint, source systems define performance and scalability. Microbial systems, such as bacteria and yeast, have historically dominated because they grow quickly and cost-effectively. They support the production of recombinant proteins like insulin and certain growth factors. However, mammalian expression systems continue to gain ground. These systems more accurately reproduce human-like protein structures and post-translational modifications, which are crucial for complex antibodies and fusion proteins.
Advances in cell-line engineering and culture technologies reduce previous limitations in yield and cost. As pipelines increasingly feature sophisticated molecules, demand for mammalian systems will continue to expand. The balance between microbial efficiency and mammalian fidelity shapes the future of bioprocessing strategies.
AI Enters the Biologics Equation
Artificial intelligence has moved from buzzword to operational tool. In biologics discovery, AI algorithms analyze massive biological datasets to identify promising targets and predict binding interactions. This capability compresses early-stage timelines that once consumed years. Machine learning also optimizes protein design, stability prediction, and immunogenicity risk assessment.
Clinical development benefits as well. AI-driven patient matching improves recruitment and stratification, increasing the likelihood of trial success. During manufacturing, predictive analytics monitor process parameters in real time, reducing batch failures and improving yield consistency. AI does not replace scientists, but it amplifies their capacity to interpret complex biological information at scale.
Biosimilars Reshape the Competitive Field
As patents expire on blockbuster biologics, biosimilars introduce both opportunity and tension. These therapies offer comparable efficacy at lower prices, expanding patient access and easing pressure on healthcare budgets. For payers and governments, biosimilars represent a crucial lever for sustainability.
For originator companies, the landscape becomes more competitive. They must differentiate through next-generation formulations, combination strategies, or superior delivery systems. Speed, cost efficiency, and regulatory navigation become decisive factors. Over the coming decade, dozens of high-revenue biologics will lose exclusivity, intensifying the race among biosimilar developers.
Regional Powerhouses Drive Global Momentum
North America continues to lead, supported by strong research ecosystems, favorable reimbursement structures, and dense clusters of biotech innovation. Cities known for life sciences research serve as hubs where academia, startups, and pharmaceutical giants converge. Regulatory clarity and investment capital sustain this leadership.
Asia-Pacific, however, shows some of the fastest growth. Countries such as China, India, and Japan invest heavily in biomanufacturing capacity and biotechnology research. Large-scale facilities, expanding domestic markets, and supportive policy frameworks accelerate regional development. Europe maintains strength through scientific heritage and regulatory rigor, while emerging markets gradually build infrastructure and expertise.
The Economics of Complexity
Biologics remain expensive to develop and manufacture. Production involves living systems, sensitive processes, and stringent quality controls. Cold-chain logistics add another layer of cost. Yet healthcare systems increasingly accept these expenses when therapies deliver significant clinical benefit. Value-based pricing models and outcome-driven reimbursement schemes may become more common as stakeholders seek balance between innovation and affordability.
Collaboration Defines the New Normal
The biologics ecosystem thrives on partnerships. Large pharmaceutical companies collaborate with biotech innovators to access novel platforms. Venture capital supports early-stage science. Academic institutions contribute fundamental discoveries. Contract research and manufacturing organizations extend operational reach. No single entity can master every dimension of biologics development, and the industry has adapted to this interdependence.
Looking Ahead with Scientific Confidence
As we move deeper into 2026 and beyond, the trajectory remains clear. Biologics will not replace all small-molecule drugs, but they will dominate areas where precision matters most. RNA therapeutics, gene-editing tools, and advanced antibody formats will broaden therapeutic reach. Manufacturing technologies will become more automated and data-driven. Regulatory frameworks will continue to adapt, balancing safety with the urgency of unmet medical needs.
From my vantage point, the most striking aspect of this evolution is not just the scale of market growth. It is the shift in mindset. We no longer view disease as something to bluntly suppress. We aim to understand, target, and modulate biological systems with accuracy. That philosophy, supported by technology and global investment, ensures that biologics will remain central to healthcare’s next chapter.
The biologics era is not approaching. It is already here, and it is redefining medicine in ways that will shape patient care for decades.
Access our exclusive, data-rich dashboard dedicated to the biotechnology industry – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout Biologics Market Report Now at: https://www.towardshealthcare.com/checkout/5108
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest