I have watched diagnostics evolve from slow, culture-based microbiology and broad histopathology interpretations into a precision-driven, gene-level discipline that now shapes frontline medical decisions. Molecular diagnostics no longer sits quietly behind the scenes. It directs therapy, flags resistance, predicts disease risk, and increasingly determines how health systems allocate resources. The period from 2026 onward will not simply show market growth; it will reflect a structural shift in how healthcare defines “early,” “accurate,” and “actionable.”
The molecular diagnostics market size is forecasted to expand from USD 17.1 billion in 2025 to USD 28.66 billion by 2035, growing at a CAGR of 5.3% from 2026 to 2035.
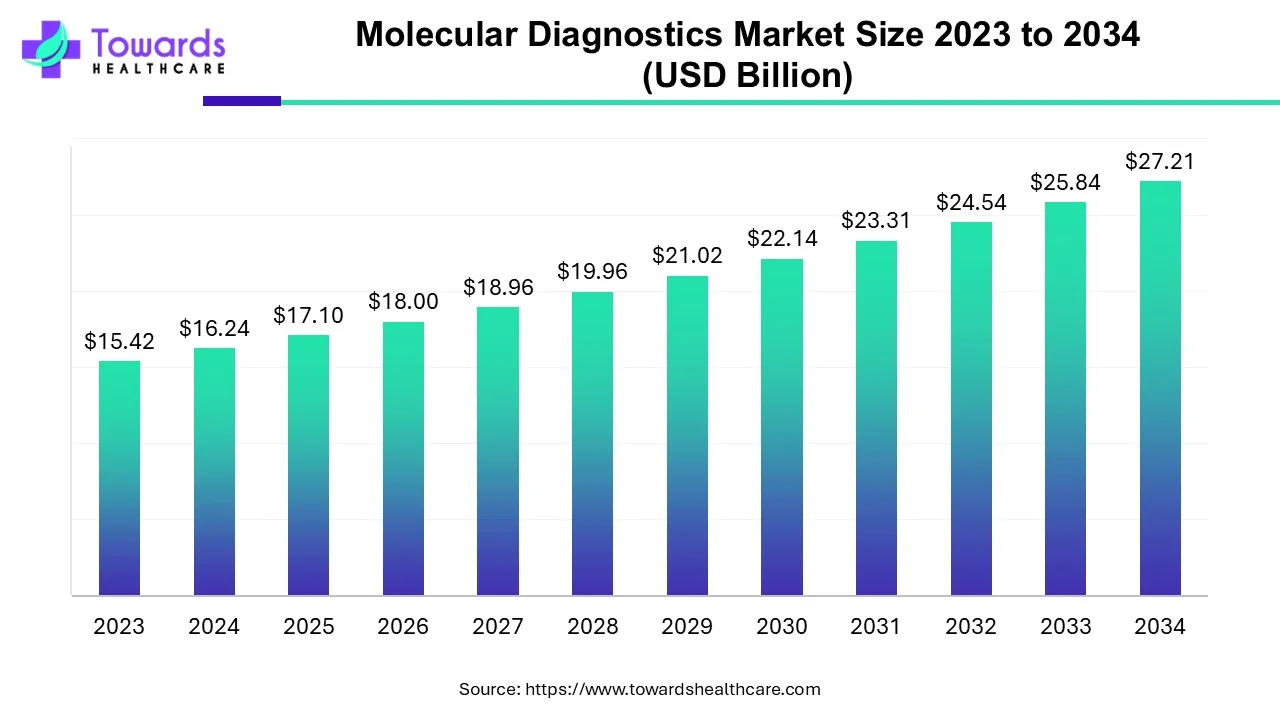
The molecular diagnostics market is entering a phase of steady but deeply transformative expansion. With projections showing growth from just over 17 billion dollars in the mid-2020s toward nearly 29 billion dollars within a decade, the numbers tell only part of the story. What truly matters is that molecular testing is moving from specialized labs into routine clinical pathways, urgent care settings, and even homes. This transition signals a fundamental change in medical culture: diagnosis is becoming faster, data-rich, and increasingly predictive rather than reactive.
From Detection to Decision-Making
Earlier in my career, laboratory tests often confirmed what clinicians already suspected. Today, molecular diagnostics frequently leads the conversation. By analyzing DNA, RNA, and specific protein signatures, clinicians can identify disease mechanisms before symptoms fully manifest. In oncology, this shift has been dramatic. Instead of labeling a tumor solely by organ of origin, we now characterize it by genetic mutations, expression patterns, and molecular pathways. That molecular fingerprint determines which drug to use, which therapy to avoid, and how aggressively to monitor recurrence.
The same dynamic now defines infectious disease management. Rapid nucleic acid amplification tests detect pathogens in hours rather than days and often reveal resistance markers at the same time. This dual capability changes treatment from empirical guesswork to targeted intervention. It also reduces inappropriate antibiotic use, a critical factor in the global fight against antimicrobial resistance. Molecular diagnostics does not merely detect microbes; it guides stewardship.
Early Diagnosis Is Redefining Survival Curves
Few areas illustrate the value of molecular diagnostics more clearly than cancer survival. When malignancies such as lung cancer are detected early, long-term survival rates increase dramatically. Molecular tools help drive this shift by identifying biomarkers and circulating tumor DNA long before imaging or symptoms reveal advanced disease. Liquid biopsies, once experimental, now represent a credible approach to monitoring tumor burden and detecting relapse with minimal invasiveness.
In infectious diseases, early molecular detection changes the trajectory of entire populations. Tuberculosis programs increasingly rely on rapid molecular assays as initial diagnostic tools, and HIV care uses viral load testing to monitor treatment success and emerging resistance. In neonates born to HIV-positive mothers, nucleic acid testing bypasses the interference of maternal antibodies and enables early diagnosis that can be life-saving. These are not incremental improvements; they are survival-critical advances.
Technology Has Outpaced Traditional Boundaries
Technological progress drives much of this transformation. Polymerase chain reaction remains foundational, but it now coexists with digital PCR, next-generation sequencing, and isothermal amplification platforms. Digital PCR pushes sensitivity and quantification to levels that were once unattainable, making it possible to detect rare mutations or minimal residual disease. Sequencing technologies, meanwhile, have shifted from large research facilities into clinical labs, enabling comprehensive genomic profiling at increasingly manageable costs.
Miniaturization represents another quiet revolution. Microfluidic systems shrink complex laboratory processes onto chips that support point-of-care testing. These systems reduce turnaround time and bring sophisticated molecular capabilities into decentralized settings, from rural clinics to emergency departments. When clinicians receive precise molecular results during a single patient visit, treatment decisions accelerate and outcomes improve.
CRISPR-based diagnostics, although still emerging, hint at the next leap forward. Their specificity and adaptability suggest a future where disease detection becomes even faster and more versatile. Combined with advances in bioinformatics and automation, these tools push molecular diagnostics toward higher throughput and broader accessibility.
The Pandemic Effect Still Echoes
The COVID-19 era reshaped global expectations around diagnostics. Health systems, governments, and the public saw how rapid molecular testing could alter the course of a crisis. Laboratories expanded capacity, supply chains adapted, and regulatory pathways accelerated innovation. That momentum has not disappeared. Instead, it has spilled into other disease areas, reinforcing the importance of preparedness and early detection.
Multiplex testing, which identifies multiple pathogens from a single sample, gained traction during the pandemic and continues to grow. This approach suits respiratory illnesses, gastrointestinal infections, and other syndromic presentations where symptoms overlap. The ability to distinguish between pathogens quickly reduces unnecessary treatments and improves infection control.
Point-of-Care and Home Testing Change Access
One of the most significant trends I see in 2026 is the shift of molecular diagnostics beyond central laboratories. Point-of-care platforms are becoming faster, more reliable, and easier to operate. This shift matters in emergency care, remote regions, and outbreak settings where time and infrastructure are limited. When a test delivers molecular accuracy at the bedside, it compresses the gap between diagnosis and treatment.
Home-based collection and self-testing also reshape the landscape. While central laboratories still play a crucial role in complex analyses, patient-facing diagnostics empower individuals and expand screening reach. As technology improves and regulatory frameworks mature, molecular tests will likely become a routine part of consumer health management, particularly for infectious diseases and certain genetic screenings.
Oncology and Infectious Diseases Lead, but the Field Is Broadening
Historically, oncology and infectious diseases dominated molecular diagnostics. They continue to do so, but new applications are emerging. Pharmacogenomics now helps predict how individuals respond to specific drugs, reducing adverse effects and improving efficacy. Genetic testing for inherited conditions informs preventive strategies long before disease develops. Neurological and cardiovascular fields are also exploring molecular markers that could transform risk assessment and early intervention.
Newborn screening programs increasingly incorporate molecular components to detect disorders that benefit from early treatment. These expansions demonstrate how molecular diagnostics is evolving from a specialty tool into a foundational component of preventive medicine.
Regional Dynamics Reflect Health Priorities
North America remains a leading region, supported by strong research ecosystems, established laboratory infrastructure, and substantial healthcare investment. However, growth momentum is clearly building in Asia Pacific. Rising awareness of antimicrobial resistance, expanding healthcare access, and government initiatives in disease surveillance drive adoption. Countries facing high burdens of tuberculosis and other infectious diseases increasingly deploy molecular tests as first-line tools.
Global funding patterns, especially in HIV and TB programs, reinforce this trend. Investments in diagnostics form a core part of disease control strategies, recognizing that accurate detection underpins effective treatment and prevention.
Mergers, Acquisitions, and Strategic Expansion
Corporate activity in recent years reflects confidence in the field’s long-term relevance. Companies across diagnostics, life sciences, and imaging have acquired molecular technology firms to strengthen portfolios. These moves aim not just at revenue growth but at integration of platforms, data capabilities, and clinical workflows. As diagnostic and therapeutic domains converge, molecular insights become central to precision medicine strategies.
This consolidation also indicates recognition that scale matters. Developing, validating, and distributing molecular technologies requires substantial resources, and partnerships help accelerate innovation and global reach.
Data Interpretation Becomes the Next Challenge
Despite remarkable progress, one reality remains clear: generating molecular data is now easier than interpreting it. We have accumulated vast genomic and transcriptomic information, but translating that knowledge into clear clinical guidance remains complex. Variants of uncertain significance, evolving resistance patterns, and multifactorial disease mechanisms challenge laboratories and clinicians alike.
Bioinformatics, artificial intelligence, and curated databases will play increasingly important roles. The value of molecular diagnostics will depend not only on detection sensitivity but also on the clarity and reliability of interpretation. As the field matures, multidisciplinary collaboration between laboratorians, clinicians, data scientists, and regulatory bodies becomes essential.
Affordability and Equity Remain Critical
While many molecular tests have become more cost-effective, disparities in access persist. High-end sequencing and specialized assays may remain concentrated in well-resourced centers. Bridging this gap requires simplified platforms, robust supply chains, and supportive reimbursement models. Innovations in point-of-care and microfluidic technologies offer hope, but policy and infrastructure decisions will heavily influence how equitably these tools spread.
The broader goal is clear: molecular diagnostics should not be a luxury. It should function as a standard element of healthcare, particularly for diseases where early detection significantly alters outcomes.
Looking Ahead with Experience and Caution
After more than a decade observing this field, I see both promise and responsibility. Molecular diagnostics has matured from a niche laboratory discipline into a strategic pillar of healthcare systems. It shapes clinical pathways, informs public health responses, and drives personalized medicine. The coming years will likely bring further integration of genomics, automation, and decentralized testing.
Yet progress must remain grounded in clinical relevance. Technology for its own sake does not improve care. Tests must answer meaningful questions, fit into workflows, and deliver results that clinicians and patients can act upon. When these conditions align, molecular diagnostics does more than identify disease. It changes the timing, precision, and effectiveness of treatment.
A Defining Era for Diagnostics
We are not simply witnessing market expansion; we are living through a redefinition of diagnosis itself. Molecular tools allow us to see disease at its biological roots, often before it manifests fully. They shorten the path from suspicion to certainty and from uncertainty to tailored care. As laboratories, clinicians, and health systems adapt, molecular diagnostics will increasingly shape how medicine anticipates and manages disease.
If the next decade fulfills its potential, diagnostics will no longer be viewed as a passive service. It will stand as an active, decision-driving force at the center of modern healthcare.
Access our exclusive, data-rich dashboard dedicated to the diagnostics market – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout Molecular Diagnostics Market Report Now at: https://www.towardshealthcare.com/checkout/5150
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest