The North America Life Sciences Sector is buzzing with innovation, investment, and transformative breakthroughs. From the rise of personalized medicine to cutting-edge genomic research, 2026 is shaping up to be a pivotal year for this industry.
With the North American life sciences market already hitting US$ 44.49 billion in 2026 and projected to soar to over US$ 124 billion by 2035, the growth story here is both compelling and complex. But what’s behind this momentum, and what trends are reshaping the industry? Let’s explore.
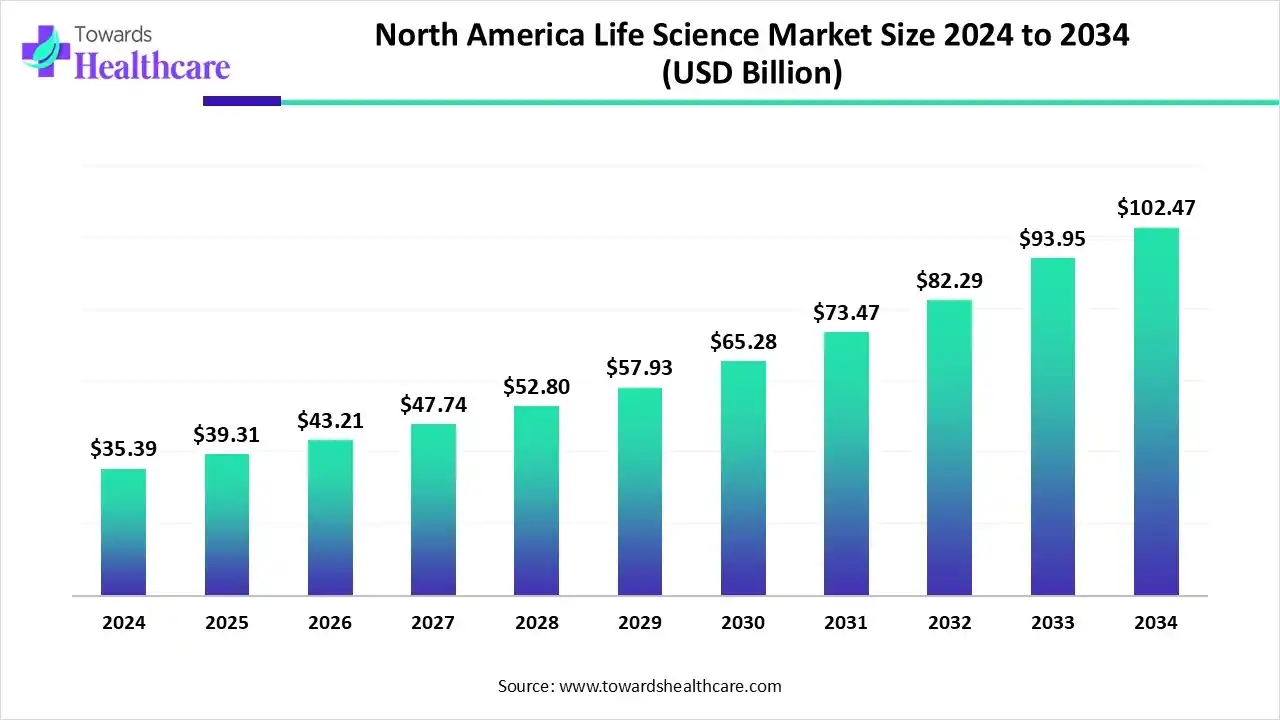
Download Free Sample: https://www.towardshealthcare.com/download-sample/6278
Betting on Innovation: Why Investments Are Soaring
Investments in North American life sciences have reached unprecedented levels. Companies are pouring resources into developing next-generation diagnostics, novel therapeutics, and AI-powered drug discovery platforms. The rising prevalence of chronic illnesses, coupled with the demand for personalized treatments, is pushing venture capital, government funding, and private investments into the sector.
Take, for instance, the BEACONS initiative launched in 2025. This multi-state genomic newborn screening program aims to integrate whole genome sequencing into state newborn screening systems. Backed by $14.4 million from the NIH Common Fund Venture Program, BEACONS exemplifies how government-backed initiatives are accelerating scientific discovery in real-world healthcare.
Similarly, Juniper Genomics, a Canadian startup, secured $4.6 million in seed funding in 2025 to commercialize an embryo screening test using whole genome and transcriptome sequencing. By analyzing millions of genetic markers, the company hopes to establish new standards for IVF genetic testing. These examples show that North American life sciences aren’t just evolving—they’re leaping forward.
Country-Level Moves: Biopharma Manufacturing on the Rise
North America isn’t just seeing innovation in labs—it’s witnessing major expansions in manufacturing capabilities. In September 2025, FUJIFILM Biotechnologies opened a massive biopharmaceutical manufacturing facility in Holly Springs. Touted as the largest cell culture CDMO facility in North America, this multi-billion-dollar investment signals a commitment to scaling drug production, ensuring that breakthroughs in R&D can reach patients efficiently.
This combination of research-driven innovation and industrial-scale manufacturing is essential for maintaining North America’s competitive edge in global life sciences. It’s one thing to discover new treatments; it’s another to make them accessible to patients in need.
R&D Revolution: Where Discovery Meets Technology
At the heart of North America’s life sciences growth is research and development. Companies are increasingly using technologies like gene editing, AI-driven drug discovery, and advanced genomics to tackle chronic diseases, cancer, and rare disorders.
Major players—such as AbbVie, Eli Lilly, Johnson & Johnson, Merck, Gilead, and Amgen—are leading the charge. These companies are pushing boundaries by exploring personalized medicine approaches, combining clinical data with AI models to predict treatment outcomes, and developing innovative therapeutics tailored to individual patients.
The R&D pipeline in North America is no longer linear. It’s dynamic, data-driven, and deeply patient-focused, emphasizing speed without compromising safety.
Clinical Trials and Approvals: Safeguarding Innovation
Innovation without regulation can be dangerous, and North America’s life sciences ecosystem reflects that. Clinical trials and regulatory approvals follow multi-phase processes to ensure safety, efficacy, and quality of new drugs and devices.
Companies like Pfizer, Moderna, AbbVie, and Johnson & Johnson are managing rigorous testing pipelines, often using adaptive trial designs that leverage real-time data for faster decision-making. This approach doesn’t just accelerate approvals—it improves the reliability of treatments reaching patients.
Interestingly, regulatory agencies are increasingly supporting collaborative frameworks, such as real-world evidence studies, which combine clinical trial data with patient outcomes observed outside controlled settings. This approach ensures therapies are not only safe but also practical and effective in everyday healthcare environments.
Patient Support and Services: Beyond the Lab
Life sciences isn’t just about drugs and devices—it’s about people. North American companies are expanding patient support programs to cover financial assistance, education, and access to treatments. For example:
-
Providing financial support for expensive therapies
-
Offering educational resources to improve health literacy
-
Facilitating easy access to medications, especially for chronic conditions
Companies like Amgen, Gilead, and Pfizer are focusing on patient-centric strategies that improve both outcomes and adherence to treatment plans. These initiatives highlight a growing awareness that innovation in life sciences must translate into real-world impact.
Spotlight on the Big Names: Who’s Shaping the Market
Here’s a quick look at some top players and their contributions:
Pfizer Inc. – Focused on innovative biopharmaceutical solutions for autoimmune diseases, cancer, and blood disorders.
Johnson & Johnson (Janssen) – Operating across pharmaceuticals, medical devices, and consumer health, with innovations in oncology, immunology, neuroscience, and cardiopulmonary care.
Merck & Co. (MSD) – A research-intensive company developing medicines, biologics, vaccines, and animal health products.
AbbVie Inc. – Concentrates on personalized medicine and genomics, providing solutions for cancer, eye care, immunology, and other conditions.
Amgen Inc. – Combines biotechnology with technology to tackle heart disease, cancer, rare diseases, osteoporosis, and inflammatory conditions.
These companies aren’t just profit-driven, they’re reshaping the healthcare landscape through innovation, collaboration, and patient-first approaches.
Global Context: North America in a Growing World
While North America leads in terms of investment, technology, and regulatory standards, the global life sciences market is also expanding rapidly. From US$ 88.2 billion in 2024 to an expected US$ 269.56 billion by 2034, the global sector mirrors North America’s trajectory but with a wider geographical reach.
Countries across Europe, Asia, and the Middle East are ramping up investments, adopting advanced R&D techniques, and creating supportive regulatory environments. This global expansion emphasizes a key point: innovation in life sciences is no longer localized—it’s interconnected and collaborative.
Market Drivers: What’s Fueling Growth?
Several factors are propelling North America’s life sciences market forward:
-
Rising Disease Burden – Chronic illnesses and rare diseases are increasing, creating a demand for advanced therapies.
-
Technological Innovation – AI, gene editing, and next-gen diagnostics are transforming drug development.
-
Policy and Funding Support – Government initiatives, venture capital, and private funding are boosting the sector.
-
Patient-Centric Approaches – Focus on affordability, accessibility, and education is improving treatment adherence.
-
Global Collaborations – Partnerships across borders accelerate R&D and expand market reach.
Emerging Trends: The Future Is Personalized
The North American life sciences sector is moving toward personalized and precision medicine. By analyzing genomic, transcriptomic, and proteomic data, companies can design therapies tailored to each patient’s unique biological profile.
Other emerging trends include:
-
Whole Genome Sequencing – Becoming a standard for early detection and preventive medicine.
-
AI-Driven Drug Discovery – Reducing R&D timelines and increasing predictive accuracy.
-
Next-Generation Biomanufacturing – Scaling production efficiently while maintaining quality.
-
Collaborative Research Models – Combining academia, startups, and big pharma for faster innovation.
These trends indicate a market that isn’t just growing in size—it’s evolving in sophistication.
Life Science Value Chain: From Lab to Patient
Understanding the North American life sciences value chain helps explain why the sector is thriving:
-
R&D – Focused on novel treatments using advanced technologies.
-
Clinical Trials & Regulatory Approvals – Ensuring safety, efficacy, and compliance.
-
Manufacturing & Supply – Scaling breakthroughs for wide distribution.
-
Patient Services – Supporting affordability, accessibility, and education.
This integrated value chain ensures that innovation translates into tangible health outcomes, making North America a global leader in life sciences.
Challenges Ahead: Balancing Speed with Safety
Despite the optimism, the sector faces challenges:
-
High R&D Costs – Developing new therapies can run into billions.
-
Regulatory Hurdles – Accelerating approvals while maintaining safety is a delicate balance.
-
Access and Affordability – Ensuring equitable access to treatments remains a concern.
-
Data Privacy – Handling genomic and health data securely is critical.
Navigating these challenges requires collaboration, innovation, and ethical stewardship—areas where North American companies are investing heavily.
Looking Ahead: What 2035 Could Look Like
By 2035, North America’s life sciences market is projected to reach US$ 124 billion, reflecting a CAGR of 12.08% from 2026 onward. This growth will likely be driven by:
-
Expanding Personalized Medicine – Individualized treatments for cancer, rare diseases, and chronic conditions.
-
AI and Genomics Integration – Smarter diagnostics, predictive analytics, and optimized therapies.
-
Global Collaboration – Partnerships across continents for research, trials, and manufacturing.
-
Patient-Centric Ecosystems – Beyond medicine, including digital health, financial support, and education.
The sector isn’t just growing—it’s transforming the way we approach healthcare, from prevention to treatment.
Conclusion: North America’s Life Sciences Are Writing the Future
North America’s life sciences market is more than a growth story—it’s a narrative of human progress. Fueled by investment, innovation, and patient-focused strategies, the sector is pioneering treatments, redefining clinical standards, and shaping a healthier future.
From genomic newborn screening programs to large-scale biomanufacturing facilities, every development signals a commitment to solving some of humanity’s most pressing health challenges.
As we move through 2026 and beyond, the life sciences industry in North America isn’t just keeping pace with global demand—it’s setting the pace, turning scientific discoveries into tangible benefits for millions of patients.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout North America Life Science Market Report Now at: https://www.towardshealthcare.com/checkout/6278
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest