Across laboratories, research hospitals, and biotech centers in the United States, a new wave of medical innovation is quietly reshaping how diseases may be treated in the future. Scientists are no longer focused only on managing symptoms. Instead, they are exploring ways to repair genes, modify cells, and potentially cure diseases that were once considered untreatable.
This transformation is unfolding through the rapid expansion of cell and gene therapy clinical trials. These trials evaluate treatments that use living cells or genetic material to target disease at its biological source. As research accelerates, the United States has emerged as one of the most active environments for testing these advanced therapies.
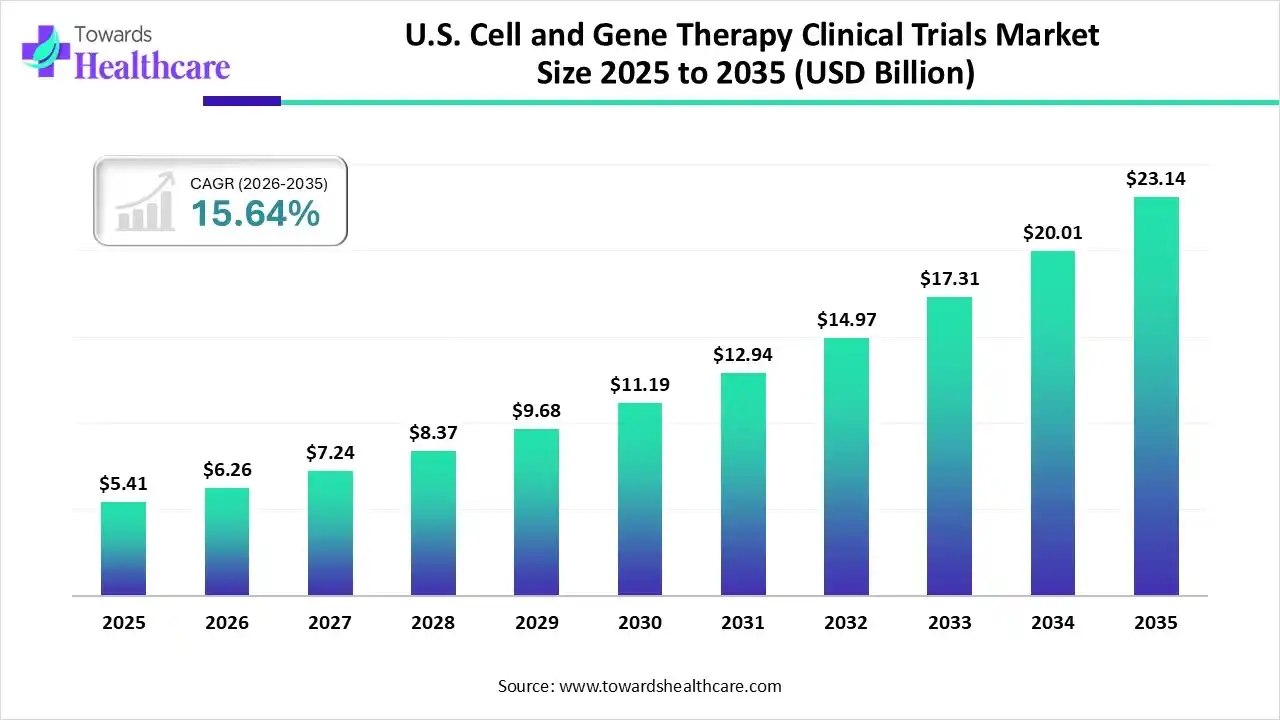
The U.S. cell and gene therapy clinical trials market was valued at USD 5.41 billion in 2025 and is projected to grow to USD 6.26 billion in 2026. Analysts expect the market to reach USD 23.14 billion by 2035, reflecting the growing interest in next-generation medicine and the rising number of clinical programs entering development.
𝐃𝐨𝐰𝐧𝐥𝐨𝐚𝐝 𝐅𝐫𝐞𝐞 𝐒𝐚𝐦𝐩𝐥𝐞: https://www.towardshealthcare.com/download-sample/6607
Why Cell and Gene Therapies Are Gaining Momentum
Traditional medicines typically treat disease symptoms or slow progression. Cell and gene therapies take a different approach. They attempt to correct the underlying cause of disease at the molecular or cellular level.
Researchers are developing therapies that:
-
Replace or repair defective genes
-
Reprogram immune cells to attack cancer
-
Regenerate damaged tissues
-
Deliver therapeutic proteins directly into cells
This approach has generated strong momentum across the healthcare ecosystem. Pharmaceutical companies, biotech startups, research institutions, and venture capital investors are all increasing their involvement in the field.
Several factors are driving this growth.
Rising Demand for Advanced Treatments
Cancer and rare genetic disorders continue to create major healthcare challenges. Many of these conditions have limited treatment options. Cell and gene therapies offer a new pathway for addressing these diseases with targeted and long-lasting effects.
A Strong Biotechnology Ecosystem
The United States hosts some of the world’s most advanced biotechnology clusters. Collaboration between academic researchers, clinical institutions, and biotech companies allows discoveries to move quickly from laboratories into clinical trials.
Supportive Regulatory Environment
Regulatory pathways in the United States are evolving to support innovation in advanced therapies. Programs designed to accelerate treatment development are helping companies bring promising therapies into clinical testing more efficiently.
Clinical Trials Are Expanding at Every Stage
Clinical trials remain the backbone of medical innovation. They determine whether new therapies are safe, effective, and ready for broader use.
In the cell and gene therapy space, trials typically move through several phases of testing. Each phase answers critical questions about how the therapy works.
Phase III Trials Lead the Market
Late-stage Phase III trials currently account for the largest share of the U.S. clinical trial landscape. These trials involve large patient populations and evaluate treatment effectiveness before regulatory approval.
Because they require extensive patient monitoring, long study periods, and complex protocols, Phase III trials often demand significant financial and operational resources. As more therapies move toward commercialization, the number of these large-scale trials continues to grow.
Early-Stage Research Is Expanding Rapidly
While Phase III trials dominate today, Phase I trials are expected to grow the fastest in the coming years. These early studies focus on evaluating safety and determining appropriate dosage levels.
The increase in Phase I trials reflects the expanding pipeline of new therapies entering development. Biotech startups and research institutions are launching innovative programs aimed at treating rare diseases, genetic disorders, and previously untreatable conditions.
Oncology Continues to Drive Clinical Innovation
Cancer remains one of the primary areas of research in cell and gene therapy trials. Scientists are exploring ways to harness the immune system to identify and destroy cancer cells more effectively.
One of the most promising developments involves CAR-T cell therapies, which reprogram a patient’s immune cells to attack tumors. These therapies have already shown significant results in certain blood cancers, encouraging researchers to explore broader applications.
The scale of the cancer challenge also drives research activity. Millions of new cancer cases are diagnosed every year in the United States, creating urgent demand for better treatment strategies.
Because of this need, oncology has become the leading indication in cell and gene therapy clinical trials.
Cardiology Emerges as a New Frontier
While cancer research dominates the current landscape, cardiovascular disease is emerging as a new focus area for gene and cell therapy trials.
Heart disease remains a leading cause of death worldwide, and many advanced cardiac conditions have limited treatment options. Researchers are now investigating gene therapies that target inherited heart disorders and regenerative cell therapies that could help repair damaged heart tissue.
These innovations aim to improve heart function, slow disease progression, and potentially restore damaged cardiovascular systems. As research advances, cardiology could become one of the fastest-growing segments in the clinical trial pipeline.
Artificial Intelligence Is Changing How Trials Operate
Artificial intelligence is also beginning to transform how clinical trials are designed and managed.
Clinical trials traditionally require significant time and resources to recruit patients, analyze data, and monitor safety. AI technologies can streamline many of these processes and help researchers make faster, more informed decisions.
AI tools can assist researchers in several ways:
-
Identifying eligible patients by analyzing medical records and genetic data
-
Predicting treatment responses through advanced data modeling
-
Monitoring safety signals in real time during trials
-
Optimizing trial design to improve efficiency and reduce costs
By improving trial operations, AI may help accelerate the development of cell and gene therapies while lowering barriers for smaller biotech companies entering the field.
Access our exclusive, data-rich dashboard dedicated to the therapeutic area sector – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout this Market Report Now at: https://www.towardshealthcare.com/checkout/6607
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest