As pharmaceutical innovation accelerates, bioanalytical testing has become one of the most critical pillars of drug development. Without it, researchers would struggle to prove how medicines interact with the human body.
Why Bioanalytical Testing Matters More Than Ever
The pharmaceutical industry has entered an era of precision medicine, targeted therapies, and biologics. Each new drug candidate requires detailed testing to ensure safety, stability, and performance.
Bioanalytical testing helps researchers answer fundamental questions:
-
How quickly does a drug absorb into the bloodstream?
-
How does the body metabolize the compound?
-
Does the drug reach the intended target site?
These insights guide scientists from early discovery stages to large clinical trials. As the number of drug candidates grows worldwide, demand for specialized testing services continues to rise.
Small Molecules Still Lead the Market
Despite the excitement surrounding biologics, small-molecule drugs remain the backbone of the pharmaceutical industry.
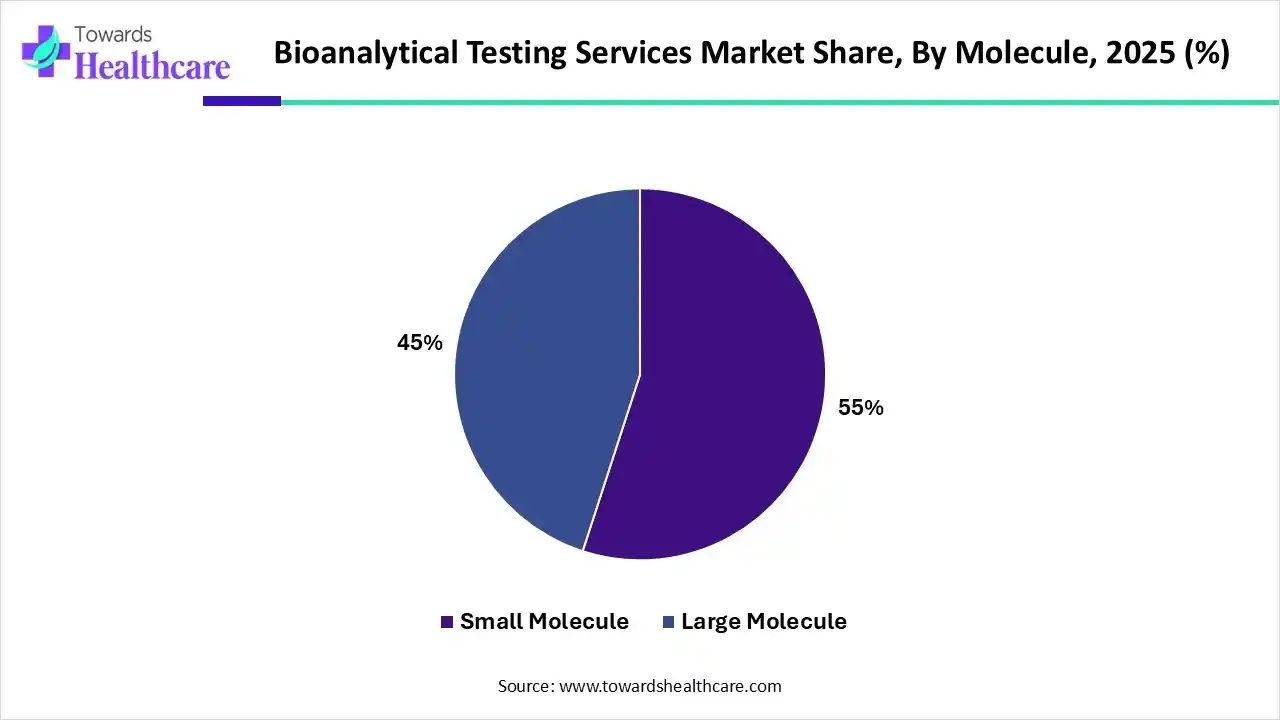
In 2025, the small molecule segment accounted for roughly 55% of the bioanalytical testing services market, reflecting their widespread use across therapeutic areas.
Download the free sample and get the complete insights and forecasts report on this market @ https://www.towardshealthcare.com/download-sample/6422
Small molecules dominate for several practical reasons. Researchers can design and synthesize them using well-established chemical pathways. Their structures are simpler compared to complex biologics, which makes drug discovery faster and more cost-efficient.
However, simplicity does not eliminate the need for rigorous analysis.
Bioanalytical laboratories perform extensive testing on small molecules to evaluate drug metabolism and pharmacokinetics (DMPK) profiles. Scientists study these compounds across multiple stages, including discovery research, preclinical studies, toxicology assessments, and clinical trials.
This continuous evaluation ensures that potential drugs behave predictably in the human body.
The Rise of Large Molecule Bioanalysis
While small molecules dominate today, large-molecule drugs are reshaping the future of medicine.
Biologics such as monoclonal antibodies, peptides, and recombinant proteins provide highly targeted treatment options. These therapies often minimize systemic side effects by acting directly on specific disease pathways.
However, their complexity introduces new challenges.
Large molecules require sophisticated laboratory equipment and specialized analytical methods. Researchers must track protein stability, immune responses, and biological interactions that do not occur with traditional small molecules.
As more biotechnology companies enter the drug development race, laboratories increasingly invest in advanced technologies to support large-molecule bioanalysis.
This shift is expected to drive rapid growth in this segment during the coming years.
Bioavailability Testing Takes Center Stage
Among the various testing methods used in drug development, bioavailability studies hold a particularly important role.
In 2025, the bioavailability segment captured nearly 40% of the market share, reflecting its importance in understanding drug absorption and distribution.
Bioavailability measures how quickly and how much of a drug reaches the bloodstream after administration. If a drug fails to reach adequate concentrations, it cannot deliver the desired therapeutic effect.
During development, bioavailability studies help researchers:
-
Identify weaknesses in drug formulation
-
Modify drug delivery mechanisms
-
Improve absorption efficiency
By refining these parameters early, scientists can develop medicines that work more effectively in real-world clinical settings.
Bioequivalence Studies Gain Momentum
The pharmaceutical landscape is also witnessing a surge in generic drugs and biosimilars. As patents for major medications expire, companies race to introduce equivalent alternatives.
This trend has increased the demand for bioequivalence testing.
Bioequivalence studies compare a generic drug to its original branded counterpart. Scientists analyze whether both versions release the active ingredient into the bloodstream at the same rate and extent.
If the results match within regulatory standards, authorities approve the generic drug for market use.
With biosimilar approvals steadily increasing worldwide, bioequivalence studies are expected to experience significant growth in the coming years.
ADME Studies: The Early Warning System
One of the biggest challenges in pharmaceutical research is drug failure during clinical trials.
A major contributor to these failures lies in poor pharmacokinetic properties. This is where ADME studies—absorption, distribution, metabolism, and excretion—play a critical role.
ADME analysis helps researchers predict how a drug will behave inside the body long before it reaches clinical testing.
Scientists use multiple approaches, including:
-
Computer simulations (in silico)
-
Laboratory experiments (in vitro)
-
Animal studies (in vivo)
These studies act as an early warning system. If a compound shows poor ADME characteristics, researchers can modify its chemical structure before investing heavily in clinical trials.
Sample Analysis: The Core of Bioanalytical Testing
Behind every drug study lies a massive amount of laboratory data.
In 2025, sample analysis accounted for about 50% of the workflow within bioanalytical testing services, making it the largest operational segment.
During this stage, scientists analyze biological samples such as blood, plasma, or tissue. They measure drug concentrations and identify metabolic by-products.
Advanced instruments like mass spectrometry and chromatography enable laboratories to detect even trace amounts of compounds.
Automation technologies and artificial intelligence tools are also transforming this process. These innovations reduce manual errors and allow researchers to analyze larger datasets with greater accuracy.
The Bottleneck: Sample Preparation
Before scientists can analyze samples, they must prepare them carefully.
Sample collection and preparation represent one of the most technically challenging steps in bioanalytical workflows.
Biological matrices contain complex substances such as proteins, lipids, and enzymes. These components can interfere with analytical results if not handled properly.
Therefore, laboratories follow strict procedures for collecting, transporting, and storing biological samples. Proper preparation ensures that analytes remain stable and results remain reliable.
As research volumes grow, improving efficiency in this stage has become a major priority for many laboratories.
Method Development: Ensuring Accuracy in Every Test
Another critical stage in bioanalytical testing involves method development and validation.
Scientists design analytical methods to measure drug concentration, purity, potency, and stability. These methods must meet strict regulatory standards before they can be used in official studies.
Validation confirms that the method produces accurate, reproducible results under different laboratory conditions.
This process helps researchers choose the most reliable analytical techniques while ensuring that regulatory authorities trust the generated data.
Pharma Companies Drive Market Demand
Pharmaceutical and biopharmaceutical companies remain the primary users of bioanalytical testing services.
In 2025, this segment accounted for approximately 60% of total market activity.
Large pharmaceutical companies often maintain internal testing facilities equipped with advanced instruments. These in-house laboratories allow them to conduct experiments quickly and maintain tighter control over research timelines.
However, the growing number of drug candidates has increased the workload dramatically.
As a result, many companies outsource specific testing activities to specialized laboratories.
CROs and CDMOs Step Into the Spotlight
Contract research organizations (CROs) and contract development and manufacturing organizations (CDMOs) play an increasingly important role in the pharmaceutical ecosystem.
These organizations provide specialized laboratory services under strict regulatory guidelines. Their expertise allows pharmaceutical companies to accelerate drug development without investing heavily in new infrastructure.
CROs typically focus on research services such as:
-
Bioanalytical testing
-
Method development and validation
-
Clinical sample analysis
Meanwhile, CDMOs support drug development and manufacturing processes, ensuring stability, safety, and production quality.
Together, these partners form an integrated network that supports the entire pharmaceutical innovation pipeline.
Access our exclusive, data-rich dashboard dedicated to the healthcare market – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout the Market Report Now at: https://www.towardshealthcare.com/checkout/6422
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium