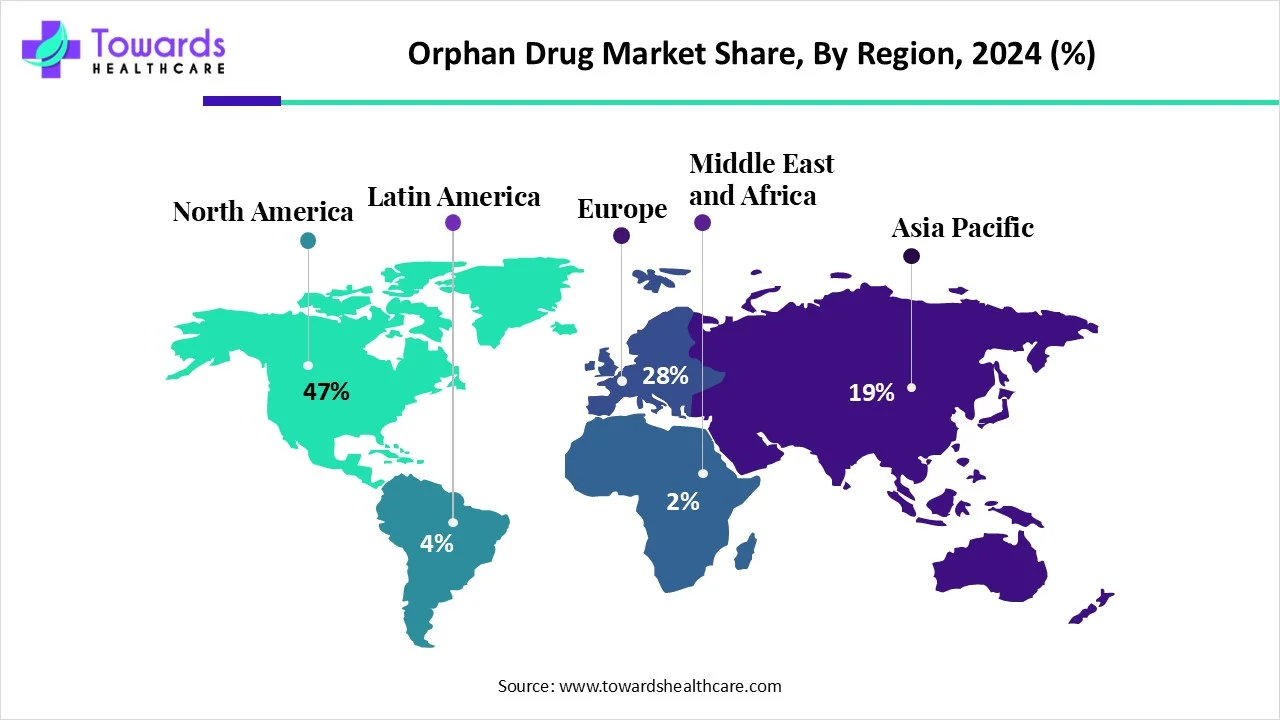
In 2025, North America captured nearly 47% of the global orphan drug market share, making it the dominant regional leader. The reasons behind this leadership are complex, involving policy innovation, strong healthcare infrastructure, and a growing understanding of rare diseases.
Download the free sample and get the complete insights and forecasts report on this market @ https://www.towardshealthcare.com/download-sample/5543
When Policy Meets Innovation
One of the biggest drivers of the orphan drug market in North America is regulatory support. Governments recognized early that pharmaceutical companies needed incentives to develop treatments for rare diseases.
In the United States, the Orphan Drug Act created a turning point. The legislation introduced benefits that significantly reduced the financial risk of developing treatments for rare conditions.
These incentives include:
-
Tax credits for clinical research
-
Government grants for drug development
-
Extended market exclusivity for approved treatments
Such policies have encouraged both large pharmaceutical companies and emerging biotechnology firms to invest in orphan drug development.
As a result, North America has become a hub for rare disease research, clinical trials, and new therapy approvals.
Why North America Dominates the Orphan Drug Market
North America’s leadership is not driven by a single factor. Instead, several structural advantages combine to create a strong ecosystem for orphan drug innovation.
First, the region has one of the most advanced healthcare infrastructures in the world. Hospitals, research institutes, and pharmaceutical laboratories operate in close collaboration, allowing faster development and testing of new treatments.
Second, awareness about rare diseases has increased significantly. Advocacy groups, patient organizations, and healthcare professionals are working together to identify rare conditions earlier and connect patients with treatment options.
Third, the region attracts major investment in biotechnology and pharmaceutical research. Venture capital firms, government grants, and academic partnerships provide a steady flow of funding that supports innovation.
Together, these elements create an environment where orphan drugs can move from research labs to patients much faster than in many other parts of the world.
The United States: The Engine of Orphan Drug Growth
Within North America, the United States plays the most influential role in shaping the orphan drug market.
The country combines scientific research, regulatory flexibility, and strong financial support for drug innovation. The U.S. Food and Drug Administration has also introduced faster approval pathways for rare disease treatments.
This approach helps companies bring therapies to market more quickly while still maintaining strict safety standards.
Another factor driving growth in the U.S. is the growing recognition of rare diseases. Millions of Americans are affected by conditions that previously lacked proper diagnosis or treatment.
Healthcare providers are now identifying these diseases earlier thanks to advances in genetic testing and diagnostic technology.
This expanding patient identification has increased the demand for orphan drugs across the healthcare system.
The Cost Challenge of Rare Disease Treatments
Despite their life-changing benefits, orphan drugs often come with a significant cost.
On average, many rare disease treatments cost around $32,000 per year, while some advanced therapies exceed $100,000 annually.
Several factors contribute to these high prices:
-
Small patient populations, which limit overall drug sales
-
High research and development costs for specialized therapies
-
Complex manufacturing processes, especially for biologic drugs
However, North America’s healthcare insurance systems often provide coverage for these treatments. Public and private insurance programs help patients access medicines that would otherwise be financially out of reach.
Patient assistance programs from pharmaceutical companies also play an important role in supporting individuals with rare conditions.
Canada’s Growing Role in Rare Disease Treatment
Canada is also strengthening its presence in the orphan drug landscape. Although the country represents a smaller market compared to the United States, it continues to expand its regulatory and healthcare support systems.
The Canadian government has introduced initiatives such as the Orphan Drug Program to encourage research and development for rare disease therapies.
Health Canada, the country’s regulatory authority, has also improved its approval processes for innovative treatments.
Canada’s well-structured healthcare system further supports access to specialized therapies, ensuring patients can receive treatment even when drug prices are high.
Collaboration between government agencies, research institutions, and pharmaceutical companies has also increased in recent years. These partnerships help accelerate clinical research and bring new therapies closer to patients.
Technology Is Changing Rare Disease Care
Another important factor shaping the orphan drug market in North America is digital health technology.
Telemedicine platforms are making it easier for patients with rare diseases to connect with specialists. In many cases, individuals with rare conditions must travel long distances to find doctors who understand their disease.
Digital healthcare tools are helping remove this barrier.
Remote consultations, digital monitoring systems, and online patient communities allow people to receive guidance and treatment recommendations without constant travel.
This shift not only improves patient convenience but also speeds up diagnosis and treatment initiation.
A Growing Focus on Patient Communities
Rare disease patients often face long diagnostic journeys. Many individuals spend years searching for answers before receiving a proper diagnosis.
North America has seen a rise in patient advocacy organizations that aim to change this reality.
These groups focus on:
-
Increasing awareness about rare conditions
-
Supporting research initiatives
-
Connecting patients with specialists and treatment options
By amplifying the voices of patients and families, these organizations have helped push rare disease research higher on healthcare policy agendas.
Their efforts also encourage pharmaceutical companies to invest in new therapies.
Access our exclusive, data-rich dashboard dedicated to the healthcare market – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout the Market Report Now at: https://www.towardshealthcare.com/checkout/5543
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium