1,700+ Clinical Trials Powering the CAR T-Cell Therapy Market!
For decades, cancer treatment followed a familiar script. Surgery removed tumors when possible, chemotherapy attacked rapidly dividing cells, and radiation attempted to contain what could not be cut away. These approaches saved lives, yet they often came at a heavy cost to patients’ quality of life and long-term health. Against this backdrop, CAR T-cell therapy did not arrive with loud promises. It entered clinical research as an experimental idea rooted in immunology: what if the body’s own immune cells could be reprogrammed to hunt cancer with precision?
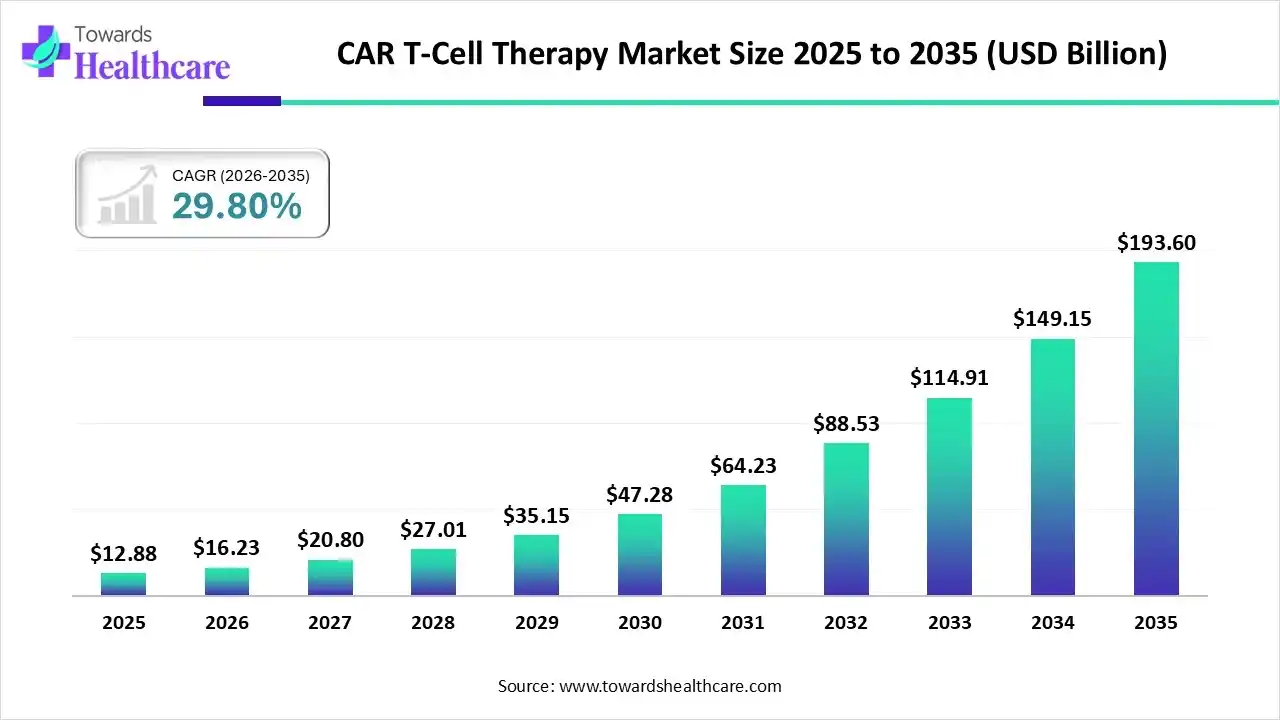
Today, that idea is no longer theoretical. CAR T-cell therapy has become one of the most disruptive advances in modern oncology, reshaping expectations around personalized medicine, long-term remission, and immune-based cancer control. The market numbers reflect this transformation. From a valuation of US$ 12.88 billion in 2025 to a projected US$ 193.6 billion by 2035, the growth trajectory tells a story of accelerating confidence from clinicians, regulators, investors, and patients alike. Yet beyond the numbers lies a deeper narrative of scientific persistence, clinical learning, and global collaboration.
Download Free Sample Now and Get the Complete Report Easily at: https://www.towardshealthcare.com/download-sample/5028
Table of Contents
ToggleWhy CAR T-Cell Therapy Commands Attention Now
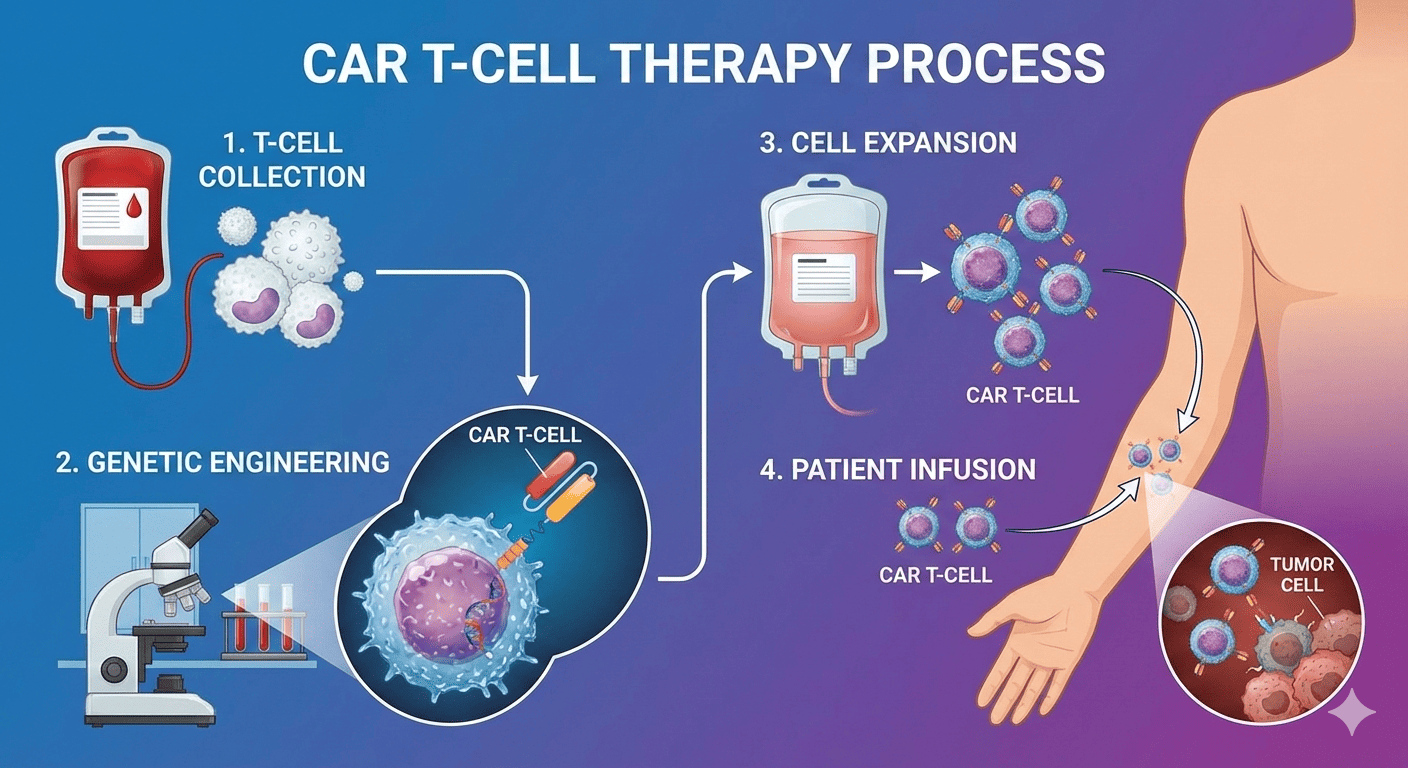
CAR T-cell therapy stands apart because it does not treat cancer as a generic disease. Instead, it treats cancer as a highly individual biological failure that demands a personalized response. The process begins by collecting a patient’s own T cells, genetically engineering them to express chimeric antigen receptors, and reinfusing them to recognize and destroy cancer cells with remarkable specificity. This approach turns the immune system into a living drug, capable of adapting, persisting, and responding if cancer attempts to return.
What makes this moment particularly important is not just that CAR T-cell therapies work, but that they have demonstrated durable responses in patients who had exhausted nearly every other option. In relapsed or refractory blood cancers such as lymphoma, acute lymphocytic leukemia, and multiple myeloma, CAR T-cell therapy has achieved outcomes that were once considered improbable. These results have forced a reassessment of how aggressive cancers should be treated and when advanced therapies should be introduced.
The Market’s Rapid Expansion Signals More Than Commercial Success
A compound annual growth rate nearing 30 percent is rare in healthcare, especially for therapies that involve complex manufacturing and specialized clinical delivery. This expansion signals something deeper than commercial enthusiasm. It reflects a structural shift in oncology toward regenerative and cell-based medicine. As small molecules and traditional biologics reach their limits in addressing complex cancers, CAR T-cell therapy fills a critical therapeutic gap.
North America’s early leadership in this market is no coincidence. Advanced healthcare infrastructure, reimbursement frameworks, and a concentration of biopharmaceutical innovators allowed the region to move first. The United States, in particular, established itself as an early center of excellence through regulatory clarity and large-scale clinical trial activity. With hundreds of accredited hospitals capable of delivering CAR T-cell therapy, the country created an ecosystem that could support both innovation and access.
At the same time, the global balance is shifting. Asia-Pacific is no longer a follower in this field. China’s rapid expansion in clinical trials has altered the global research landscape, while India’s push toward indigenous CAR T-cell development reflects a broader ambition to democratize access to advanced therapies. These regional dynamics suggest that the future of CAR T-cell therapy will not be dominated by a single geography, but shaped by multiple innovation hubs working in parallel.
Clinical Trials Reveal a Technology Still in Motion
One of the most telling indicators of the CAR T-cell therapy market’s maturity is the sheer volume of ongoing clinical trials. With more than 1,700 registered studies worldwide, the field remains highly dynamic. Early trials focused almost exclusively on hematological malignancies, where CAR T-cells could easily access cancer cells circulating in blood or bone marrow. Over time, the scope expanded.
Researchers now actively explore CAR T-cell applications in solid tumors, autoimmune diseases, and even adverse drug reactions. These efforts reflect growing confidence in the platform’s adaptability, even as they expose its limitations. Solid tumors present a hostile microenvironment that can suppress immune activity, creating barriers that CAR T-cells must overcome. While success in this area remains uneven, progress continues through improved CAR designs, combination therapies, and smarter patient selection.
The evolution of trial diversity also underscores a shift in scientific ambition. CAR T-cell therapy is no longer viewed solely as a last-resort oncology treatment. It is increasingly seen as a foundational technology with applications that may extend well beyond cancer.
Drug Innovation Drives Competitive Differentiation
Not all CAR T-cell therapies are created equal. Differences in target antigens, manufacturing processes, and safety profiles shape clinical adoption and market share. Axicabtagene ciloleucel’s early dominance reflects its strong performance in large B-cell lymphoma, a disease with significant unmet need. Its success validated the commercial viability of CAR T-cell therapy and encouraged broader investment.
At the same time, therapies such as tisagenlecleucel highlight how safety and patient demographics influence market growth. Its use in pediatric and young adult populations with acute lymphocytic leukemia demonstrates how CAR T-cell therapy can be adapted to vulnerable groups. These distinctions matter because they influence physician confidence, reimbursement decisions, and long-term adoption trends.
As new products enter the market, competition increasingly revolves around manufacturing efficiency, reduced toxicity, and broader indications. Companies that can simplify production or shorten vein-to-vein time gain a strategic advantage in a market where speed and reliability directly affect patient outcomes.
Hospitals and Specialized Centers Shape Real-World Adoption
The dominance of hospitals as primary end users reflects the therapy’s complexity. CAR T-cell treatment demands specialized infrastructure, trained multidisciplinary teams, and robust patient monitoring capabilities. Hospitals that invest in these capabilities often become regional referral centers, reinforcing their leadership position.
However, the fastest growth now emerges from dedicated cancer treatment centers. These facilities offer focused expertise, streamlined workflows, and patient-centric environments tailored to advanced oncology care. As CAR T-cell therapy becomes more standardized, such centers may play a growing role in expanding access, particularly in regions where large academic hospitals remain scarce.
This shift also reflects patient expectations. As awareness of CAR T-cell therapy grows, patients increasingly seek out specialized centers with proven experience, reinforcing demand for high-quality, dedicated treatment environments.
Artificial Intelligence Enters the CAR T-Cell Equation
Artificial intelligence has quietly become one of the most influential forces shaping the next phase of CAR T-cell therapy. From patient selection to manufacturing optimization, AI-driven tools promise to address some of the therapy’s most persistent challenges. Predictive analytics can help identify which patients are most likely to benefit, reducing unnecessary risk and cost. Machine learning models can optimize CAR design, improving target specificity while minimizing toxicity.
In manufacturing, automation powered by AI enhances consistency and scalability, addressing one of the biggest barriers to broader adoption. As CAR T-cell therapies move from artisanal production toward industrialized processes, AI becomes less of an enhancement and more of a necessity.
Beyond production, AI-enabled monitoring through wearables and digital platforms allows clinicians to track patient responses in real time. This capability supports earlier intervention when side effects arise and strengthens long-term follow-up, which remains critical in assessing durability and safety.
Regulatory Confidence Signals Long-Term Commitment
Regulatory approvals serve as milestones that shape market confidence. With multiple CAR T-cell therapies already approved and many more in late-stage development, regulators have demonstrated a willingness to engage with complex, novel treatment paradigms. This evolving regulatory maturity reduces uncertainty and encourages continued investment.
Governments also play a critical role through funding and policy support. Public investment in CAR T-cell research reflects recognition that these therapies align with broader healthcare goals, including personalized medicine, long-term disease control, and reduced reliance on chronic treatment regimens. Such support is particularly visible in countries seeking to build domestic manufacturing capacity and reduce dependence on imported therapies.
Challenges Remain, but the Trajectory Is Clear
Despite its promise, CAR T-cell therapy is not without limitations. High costs, manufacturing complexity, and serious side effects such as cytokine release syndrome remain significant hurdles. Access disparities persist, particularly in low- and middle-income regions. Durability varies among patients, and relapse remains a concern.
Yet the industry’s response to these challenges reveals why long-term optimism remains justified. New CAR designs aim to reduce toxicity and improve persistence. Allogeneic approaches seek to create off-the-shelf therapies that lower costs and speed delivery. Combination strategies attempt to overcome resistance and extend remission.
Each limitation has become a research priority rather than a roadblock, reflecting a field that continues to learn and adapt.
A Market That Reflects a Broader Healthcare Shift
The rise of CAR T-cell therapy mirrors a broader transformation in healthcare. Medicine increasingly moves away from one-size-fits-all treatments toward highly individualized solutions. Patients, clinicians, and policymakers now measure value not only by immediate outcomes, but by long-term impact on survival and quality of life.
In this context, the CAR T-cell therapy market represents more than a fast-growing segment. It represents a proof of concept for immune engineering as a therapeutic strategy. Its success influences how future cell and gene therapies are developed, regulated, and delivered.
Looking Ahead with Measured Confidence
From an experienced industry perspective, the most striking aspect of the CAR T-cell therapy market is not its speed, but its resilience. Few technologies withstand such intense scientific scrutiny, regulatory complexity, and clinical risk while continuing to expand. CAR T-cell therapy has done exactly that.
Over the next decade, the market will likely see further diversification, improved affordability, and expanded indications. Success in solid tumors would mark a turning point, unlocking an even larger patient population. At the same time, advances in manufacturing and AI integration will determine how widely and equitably these therapies can be delivered.
CAR T-cell therapy began as an ambitious experiment. Today, it stands as one of the most compelling examples of how science, technology, and clinical insight can converge to redefine what is possible in cancer care. The market’s explosive growth is not hype. It is a reflection of a therapy that has already changed lives and shows every sign of shaping the future of oncology for years to come.
Access our exclusive, data-rich dashboard dedicated to the Therapeutic area sector – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Important Links
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout CAR T-Cell Therapy Market Report Now at: https://www.towardshealthcare.com/checkout/5028
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest




