Anti-Hypertensive Drugs Market Companies with Offerings
| Vendor | Headquarters | Key Offerings / Highlights |
| Pfizer Inc. | U.S. | Leading anti-hypertensive drugs and combination therapies; active in R&D for new hypertension treatments. |
| AstraZeneca Plc | UK | Develops ACE inhibitors, ARBs, and combination drugs for hypertension and cardiovascular diseases. |
| Johnson & Johnson | U.S. | Offers anti-hypertensive therapies and cardiovascular care products; invests in patient adherence technologies. |
| Merck & Co., Inc. | U.S. | ACE inhibitors and ARBs in its cardiovascular portfolio; focuses on global hypertension management. |
| Boehringer Ingelheim GmbH | Germany | Provides angiotensin receptor blockers (ARBs) and fixed-dose combination therapies for hypertension. |
| Takeda Pharmaceutical Company Limited | Japan | Anti-hypertensive drug development, including ARBs and combination therapies; expanding market access. |
| Bayer AG | Germany | ACE inhibitors, ARBs, and novel formulations for hypertension; emphasizes patient-centric therapy solutions. |
| Daiichi Sankyo Company, Limited | Japan | Focuses on ARBs and multi-drug combinations for effective blood pressure control. |
| Teva Pharmaceutical Industries Ltd. | Israel | Generic anti-hypertensive drugs portfolio; helps improve affordability and accessibility worldwide. |
Request a free customized Anti-Hypertensive Drugs Market report with valuable stats and market breakdowns; Download Now @ https://www.towardshealthcare.com/download-sample/6416
Anti-hypertensive Drugs Market Value Chain Analysis
R&D
The R&D process for a new anti-hypertensive drug includes discovery and development, preclinical research, clinical trials, regulatory review, and post-market safety monitoring.
Key Players: Merck & Co. Inc., Sanofi S.A., Novartis AG, Pfizer Inc., Johnson & Johnson, etc.
Clinical Trials and Regulatory Approvals
The U.S. FDA approved Aprocitentan, WIDAPLIK, and Javadin. The agency also supports clinical trials and emerging therapies.
Key Players: Merck & Co. Inc., Sanofi S.A., Novartis AG, Pfizer Inc., Johnson & Johnson, etc.
Patient Support and Services
They include digital health integration, multidisciplinary care teams, medication adherence strategies, and improved patient education.
Key Players: Sanofi S.A., Novartis AG, Pfizer Inc., Johnson & Johnson, Merck & Co. Inc.
Top Companies in the Anti-Hypertensive Drugs Market
Novartis AG
Corporate Information
- Headquarters: Basel, Switzerland
- Year Founded: 1996
Business Overview
Novartis AG is a global pharmaceutical company that researches, develops, manufactures, and markets innovative medicines, generics/biosimilars (primarily through Sandoz), and consumer health products. In the area of cardiovascular and metabolic diseases (including hypertension), Novartis has a strong presence, offering multiple antihypertensive agents and fixed-dose combinations, and highlighting its commitment to cardiovascular, renal, and metabolic therapeutic areas.
Business Segments / Divisions
- Novartis has historically operated with divisions such as Innovative Medicines (pharmaceuticals), Sandoz (generics/biosimilars), and Consumer Health.
- As of its recent restructuring, Novartis is increasingly focused on innovative medicines (pharma) and has spun off the generics/biosimilar unit (Sandoz) to allow greater strategic focus.
- Within its Innovative Medicines business, Novartis covers therapeutic areas like cardiovascular/renal/metabolic, immunology, oncology, neuroscience, ophthalmology, etc.
Geographic Presence
Novartis operates globally, with sales and production facilities spanning the Americas, Europe, AsiaPacific, Latin America, the Middle East & Africa. The company maintains manufacturing and R&D sites in Switzerland, the U.S., Austria, Slovenia, China, France, Italy, and elsewhere.
Key Offerings
- Diovan® (valsartan, an ARB) and Diovan HCT® (valsartan + hydrochlorothiazide), longstanding agents in hypertension management.
- Exforge® (valsartan + amlodipine) and Exforge HCT® (valsartan + amlodipine + hydrochlorothiazide) – combination therapies designed for patients requiring more than one agent.
- Valturna® (aliskiren + valsartan) – singlepill combination targeting the reninangiotensin system at two points.
- Amturnide® / Rasitrio® – triplecombination pills (aliskiren + amlodipine + HCTZ) for patients uncontrolled on two drugs.
- Entresto® (sacubitril/valsartan) has received indication expansions for hypertension in certain markets (e.g., China) beyond its heart failure indication.
- These offerings demonstrate Novartis’s strategic emphasis on combinations (singlepill and triplepill) to improve adherence and control in hypertension.
SWOT Analysis
- Strengths:
- Strong global brand and deep legacy in cardiovascular disease management (e.g., ARBs, combination therapies).
- Extensive global presence with manufacturing and R&D capabilities across major regions, enabling scale and reach.
- Focus on innovative fixed-dose and multi-agent therapies in hypertension – aligning with market trends toward combination therapy for improved adherence.
- Weaknesses:
- Patent expirations and generic competition in some core cardiovascular/hypertension products can erode margins and market share.
- Complexity and regulatory risks associated with multi-agent combination therapies (e.g., safety, adherence, approval).
- Opportunities:
- Rising global prevalence of hypertension (especially in emerging markets) creates a large demand for novel therapies and adherence-improving formats.
- Growing emphasis on single-pill/triple-pill combinations aligns with Novartis’s portfolio strength and can differentiate them in the market.
- Expansion into digital health, remote monitoring/adherence support as adjuncts to pharmacotherapy – a trend in hypertension management.
- Threats:
- Intense generic competition, especially in high volume hypertension drugs; regulatory and pricing pressures in many markets.
- Healthcare cost containment policies and reimbursement challenges may pressure pricing of novel combination therapies.
- Safety/regulatory concerns for novel mechanisms or combination therapies (for example, renininhibitor safety issues).
Recent News
In August 2025, Novartis initiated a Phase 2b, multicenter, randomized, double-blind, placebo-controlled study to evaluate the efficacy, safety, and pharmacodynamics of QCZ484 in patients with mild to moderate hypertension. The study is expected to test multiple subcutaneous doses of QCZ484 administered every six months over a 12-month period.
Sanofi S.A.
Corporate Information
- Headquarters: Paris, France (46 Avenue de la Grande Armée, 75017 Paris)
- Year Founded: The company traces its origins to 1973.
Business Overview
Sanofi is a global pharmaceutical and healthcare company engaged in research, development, manufacturing, and marketing of prescription medicines, vaccines, and consumer health products. In the context of the anti-hypertensive drugs market, Sanofi holds a legacy role via established cardiovascular / hypertension therapies (for example, its ARB irbesartan under brand names such as Aprovel) and single-pill combinations.
Business Segments / Divisions
Sanofi organizes its business into several major segments including:
- Pharmaceuticals (innovative medicines)
- Vaccines (via Sanofi Pasteur)
- Consumer Healthcare
Rare Diseases / Specialty Care
Within Pharmaceuticals, therapeutic areas include cardiovascular (which covers hypertension), diabetes, immunology, oncology, rare diseases & more.
Geographic Presence
Sanofi operates in over 100 countries across all major regions (Americas, Europe, Asia-Pacific, Latin America, Middle East & Africa) and has manufacturing and R&D facilities worldwide. Its Western Europe market remains significant, and it is expanding in emerging markets.
Key Offerings
- Aprovel® (irbesartan): an angiotensin II receptor blocker (ARB) indicated for essential hypertension and renal protection in hypertensive type2 diabetic patients.
- CoAprovel® (irbesartan + hydrochlorothiazide): a fixeddose combination for hypertension not adequately controlled by irbesartan alone.
- Aprovasc® (irbesartan + amlodipine): a singlepill fixed combination of an ARB + a calcium channel blocker, marketed by Sanofi in collaboration (e.g., in Korea) for improved adherence and bloodpressure control.
SWOT Analysis
- Strengths:
- Strong global brand with deep legacy in hypertension/ cardiovascular therapeutics (e.g., irbesartan series).
- Broad global footprint and diversified business (pharmaceuticals + vaccines + consumer health) providing resilience.
- Experience in fixeddose combinations in hypertension, which aligns with market trends of singlepill therapy.
- Weaknesses:
- Some legacy hypertension products may face generic competition, reducing margins and requiring pipeline renewal.
- Hypertension is a highly competitive market with many players; innovation in bloodpressure-lowering drugs may be less differentiated.
- Cardiovascular/hypertension may not be as strategic a focus as specialty or immunology areas compared with peers, possibly limiting resource prioritization.
- Opportunities:
- Rising global prevalence of hypertension (especially in emerging markets) gives a large addressable market.
- Growth potential in single-pill/combo therapies and digital health adherence tools in hypertension management.
- Leverage its experience to develop novel agents (or combinations) aimed at resistant hypertension or comorbid metabolic/renal disease.
- Threats:
- Intense generic competition in antihypertensive drugs (ARB, ACE inhibitors) and pressure on pricing in many markets.
- Reimbursement & costcontainment pressures globally, especially in cardiovascular drugs, which are often lower margin than specialty therapies.
- Regulatory, safety, or trialoutcome risks associated with new antihypertensive combinations or mechanisms in the cardiovascular domain.
Market Growth
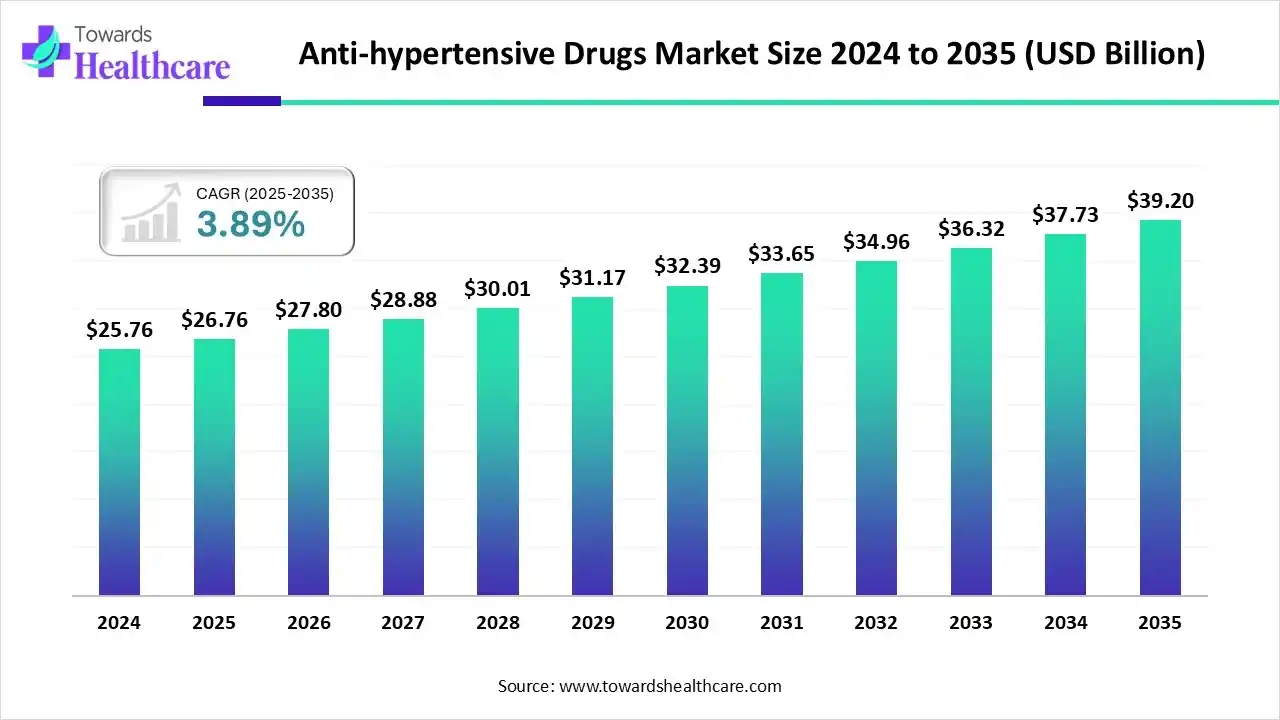
The global anti-hypertensive drugs market size is estimated at US$ 26.76 billion in 2025, grew to US$ 27.8 billion in 2026, and is projected to reach around US$ 39.2 billion by 2035. The market is expanding at a CAGR of 3.89% from 2026 to 2034.
Recent Developments
- In August 2025, AstraZeneca’s experimental drug Baxdrostat showed promise in treating uncontrolled or resistant high blood pressure. If approved, it could be one of the first new treatment approaches in decades. Trial results were presented at the European Society of Cardiology Congress in Madrid and published in the New England Journal of Medicine.
- In June 2025, George Medicines announced FDA approval of WIDAPLIK™ (telmisartan, amlodipine, indapamide), the first triple-combination pill approved for initial hypertension therapy in adults. Available in three doses, WIDAPLIK offers a triple-mechanism approach to effectively lower blood pressure with proven safety and tolerability.
Access our exclusive, data-rich dashboard dedicated to the pharmaceuticals industry – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout the Market Report Now at: https://www.towardshealthcare.com/checkout/6416
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium