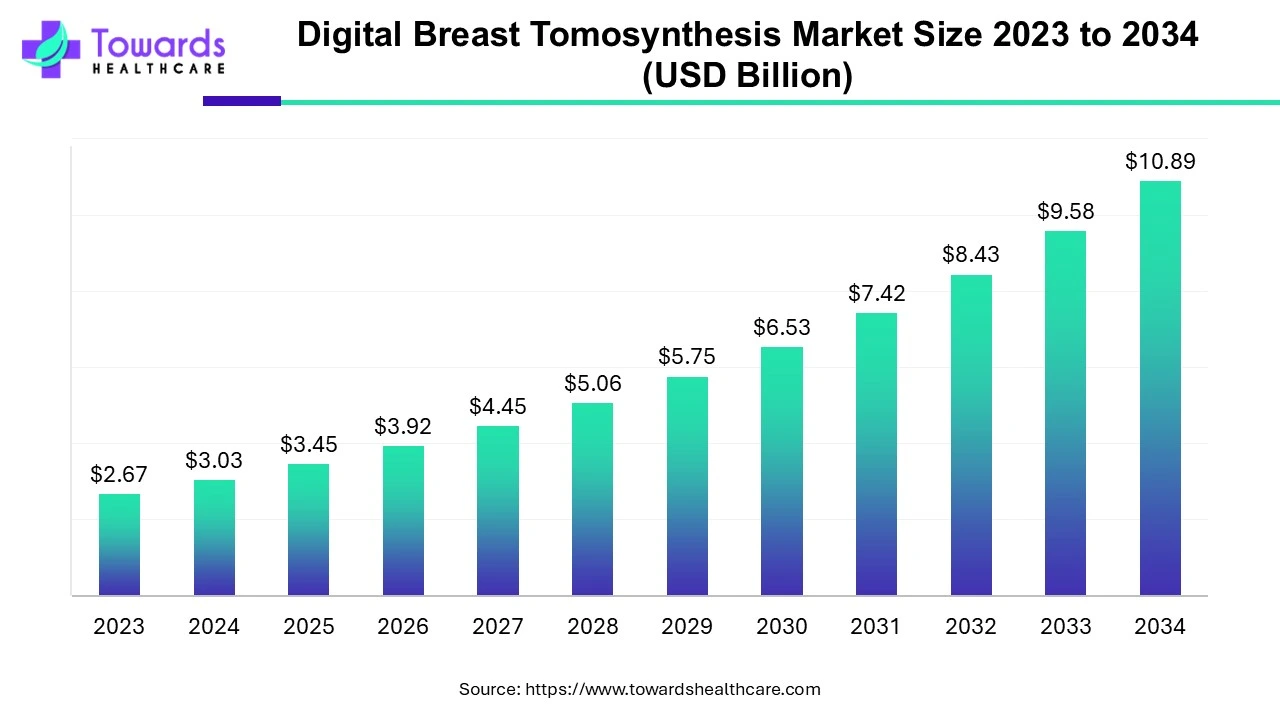
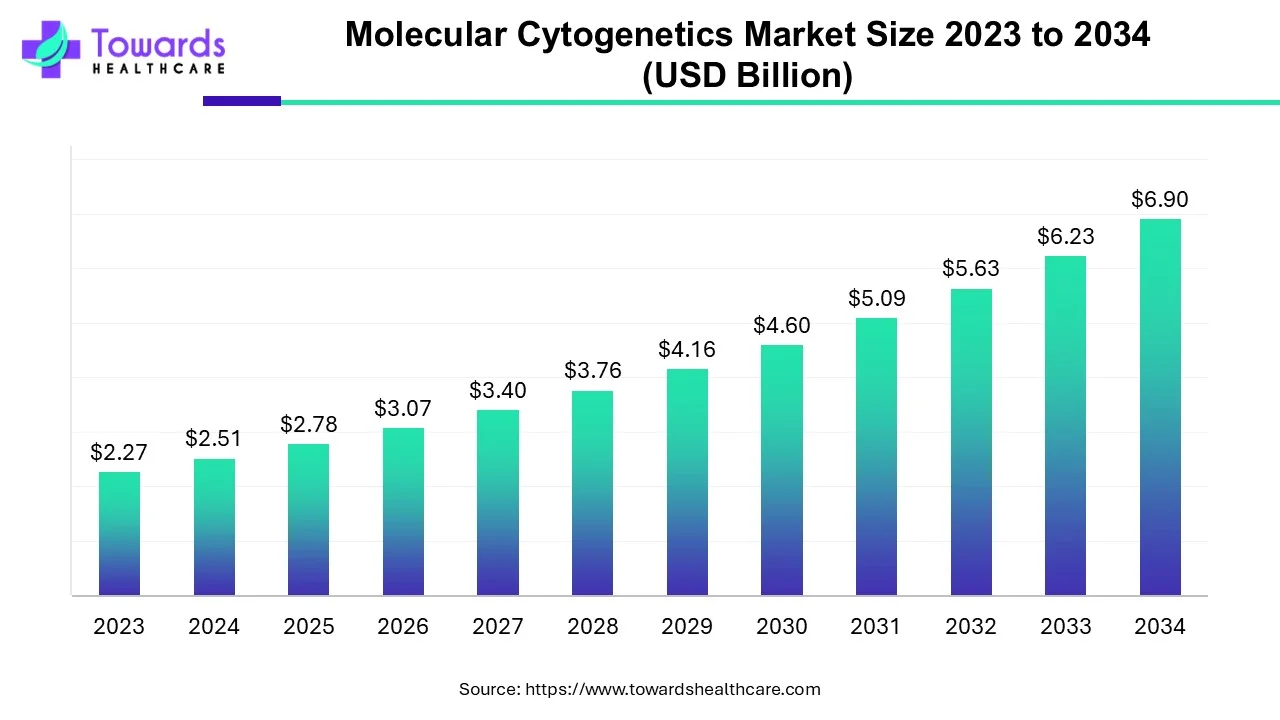
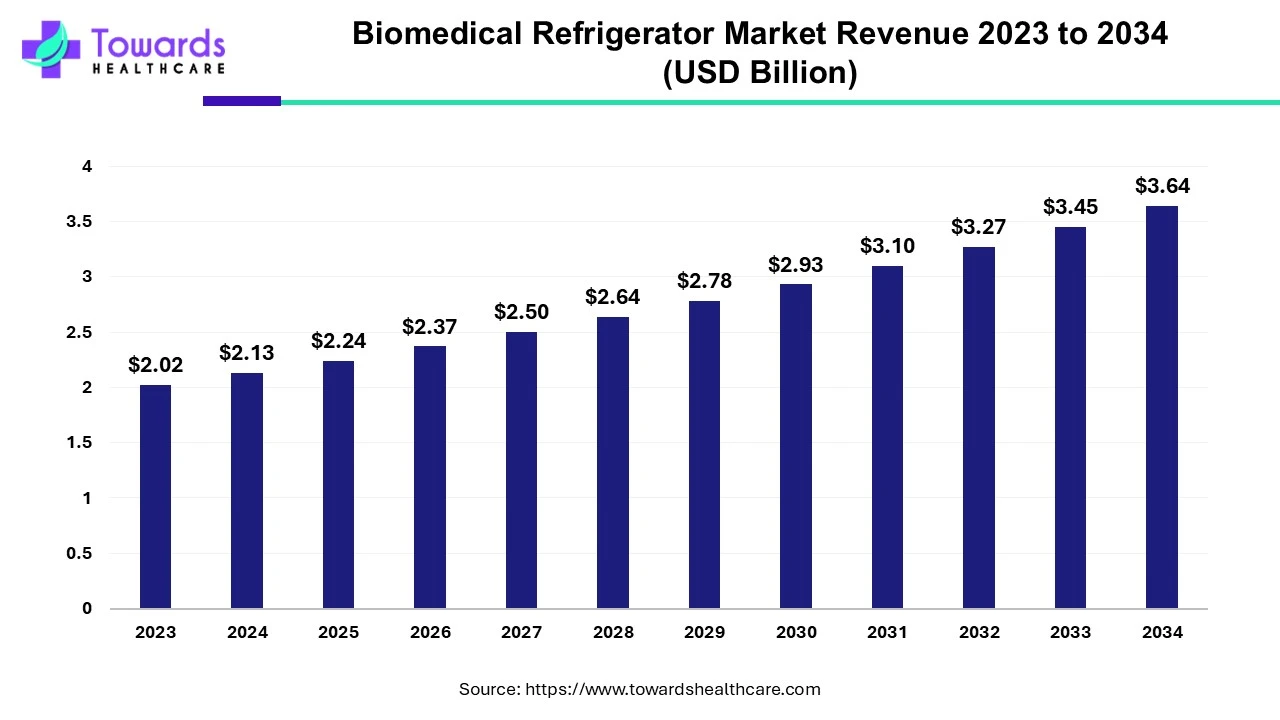
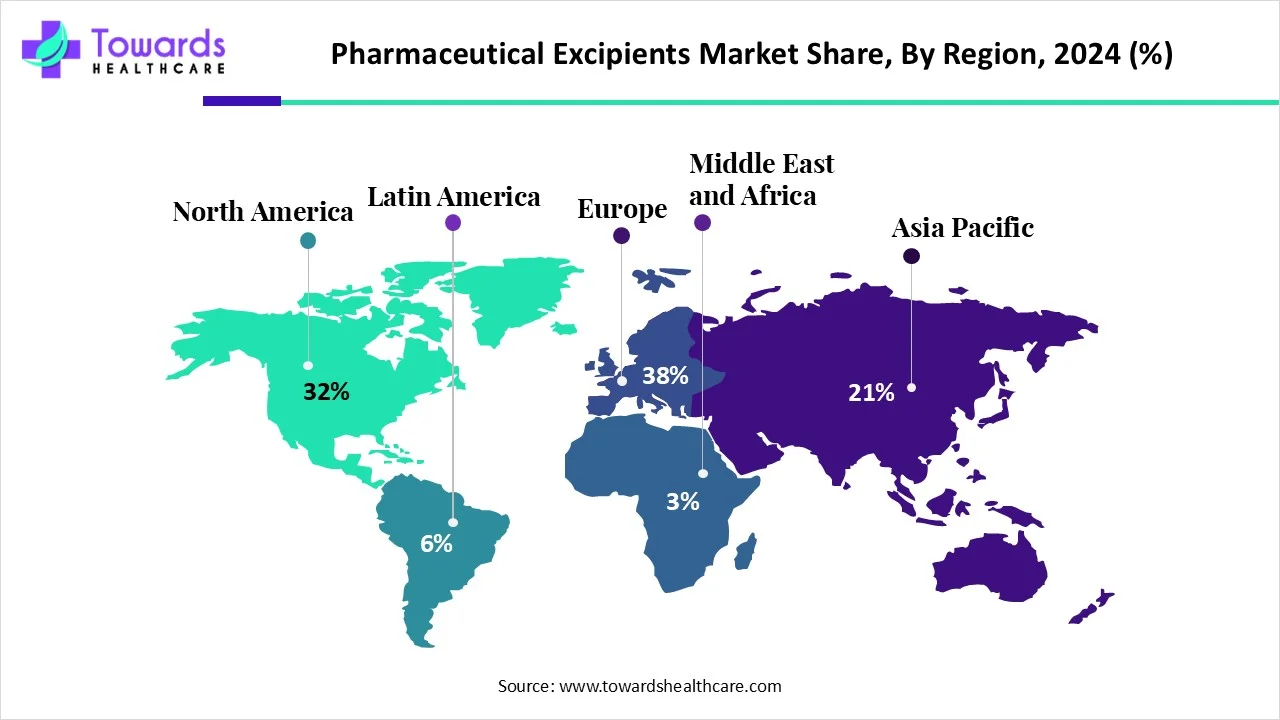
Pharmaceutical companies are changing the way they develop and manufacture drugs. More than 70% of projects today rely on Contract Development and Manufacturing Organizations (CDMOs) for critical support.
CDMOs are no longer just service providers; they are partners in innovation. They handle everything from early-stage development to large-scale manufacturing, allowing pharma companies to focus on what they do best: discovering life-saving drugs.

Our experts provide detailed stats, market segments, competitor insights and many more to guide your business decisions; Checkout first, BUY Later
Why Pharma Is Outsourcing Like Never Before
Outsourcing to CDMOs is not just about cost-cutting. Companies are increasingly seeking speed, flexibility, and technical expertise.
With drug development timelines stretching longer every year, companies cannot afford delays. CDMOs step in with ready-to-use infrastructure, skilled teams, and regulatory know-how.
This collaboration reduces bottlenecks and ensures drugs reach patients faster. It is no exaggeration to say that CDMOs are becoming the silent backbone of modern pharma.
The Numbers Speak for Themselves
- 70% of pharma projects now use CDMO services to accelerate development and reduce risk.
- 50% faster time-to-market is reported when CDMOs are involved early in the development cycle.
- Biologics and advanced therapies account for 65% of outsourced projects, highlighting the increasing complexity of modern drugs.
These numbers reflect a deeper trend: outsourcing is no longer optional; it’s strategic.
From Vendor to Strategic Partner
In the past, CDMOs were treated like vendors; tasked with executing predefined orders. Today, they are collaborators from day one.
Companies involve CDMOs in early research, formulation, and process optimization. This early engagement ensures scalability, improves regulatory compliance, and minimizes costly errors.
The result? A partnership where both sides share knowledge, risks, and successes.
Why Advanced Therapies Are Fueling Growth
Biologics, cell therapies, and gene therapies demand specialized facilities and expertise. Many pharma firms don’t have the resources to handle these in-house.
CDMOs provide end-to-end solutions:
- Controlled manufacturing environments for sensitive biologics
- Regulatory expertise to navigate global approvals
- Large-scale production capacity to meet growing demand
As these advanced therapies grow, CDMOs are evolving alongside them, ensuring the industry keeps pace with innovation.
Global Trends Driving the Outsourcing Boom
Different regions are shaping CDMO growth in unique ways:
- North America leads with biotech innovation and established regulatory systems.
- Europe contributes through mature manufacturing hubs and regulatory expertise.
- Asia-Pacific is growing rapidly, with countries like India and China providing cost-efficient services and expanding technical capabilities.
This regional diversity is making the CDMO ecosystem more competitive and robust than ever.
Technology Is Transforming CDMOs
Digital tools, automation, and artificial intelligence are revolutionizing how CDMOs operate.
They help:
- Improve precision and reduce errors
- Monitor quality in real-time
- Accelerate regulatory compliance
Companies that invest in tech-savvy CDMOs gain a clear competitive edge. They aren’t just producing drugs—they are creating smarter, faster, and safer pathways to patients.
Want to explore before committing? Our sample is personalized to your business needs.
Get it for free now
The Future: Collaboration Over Ownership
The trend is clear: pharma companies will increasingly rely on CDMOs as strategic partners rather than temporary contractors.
Long-term partnerships, joint investments, and shared responsibilities will become the norm. Sustainability will also matter more—eco-friendly processes, green manufacturing, and ethical sourcing are shaping the next generation of CDMO services.
Closing Thought
CDMO services are powering over 70% of pharma outsourcing projects, and their role will only grow.
From accelerating development timelines to supporting complex biologics, CDMOs are proving that they are more than a service; they are the backbone of modern pharmaceutical innovation.
They may operate behind the scenes, but without them, much of today’s progress in drug development would simply not be possible.
Access our exclusive, data-rich dashboard dedicated to the healthcare market – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
About Us
Healthcare Web Wire is a part of Towards Healthcare, which is a leading global provider of technological solutions, clinical research services, and advanced analytics, with a strong emphasis on life science research. Dedicated to advancing innovation in the life sciences sector, we build strategic partnerships that generate actionable insights and transformative breakthroughs. As a global strategy consulting firm, we empower life science leaders to gain a competitive edge, drive research excellence, and accelerate sustainable growth.
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium