Can Cold Chain Logistics Safely Deliver the Future of Cell and Gene Therapy?

The cell and gene therapy cold chain logistics market has moved far beyond being a background support system in healthcare. It has become one of the most critical pillars of advanced medical treatment. In 2024, this market clearly showed that delivering these therapies is as important as developing them. Cell and gene therapies are highly sensitive, often living products, and even a small mistake in temperature control or timing can destroy their effectiveness. This reality has pushed cold chain logistics into the spotlight.
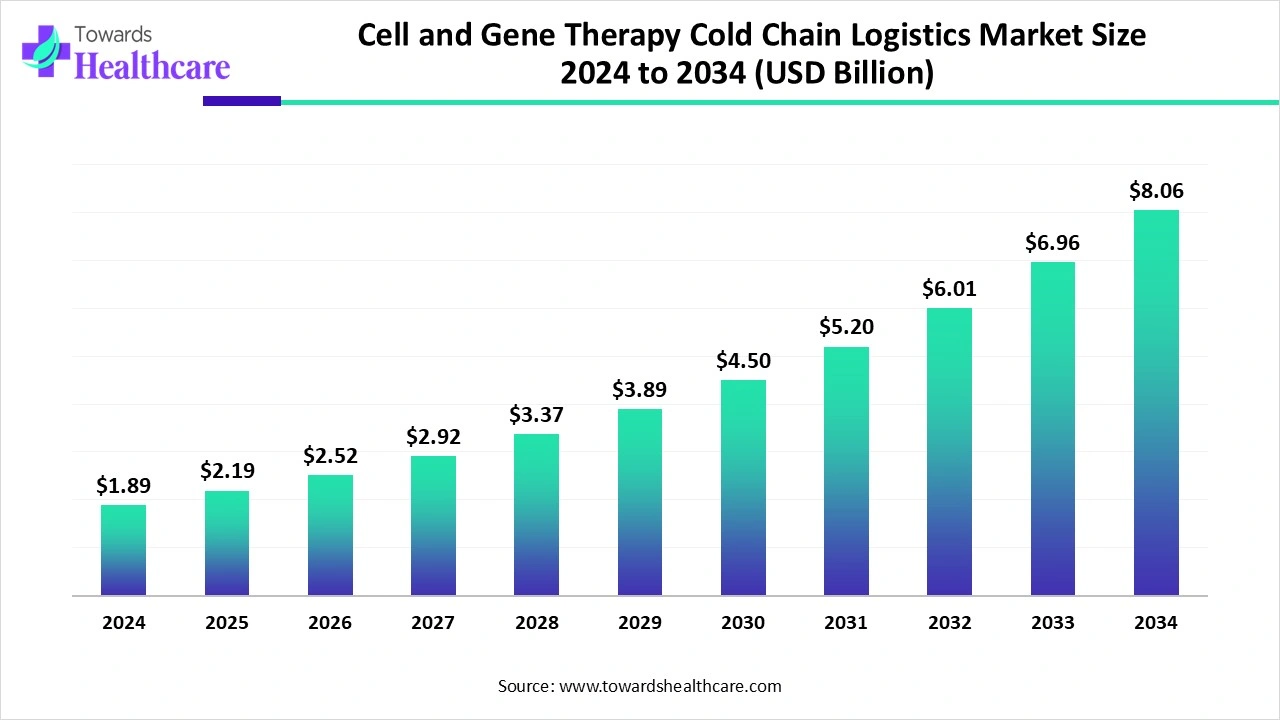
The global cell and gene therapy cold chain logistics market was valued at US$ 1.89 billion in 2024 and increased to US$ 2.19 billion in 2025. It is expected to grow steadily and reach approximately US$ 8.06 billion by 2034, registering a strong compound annual growth rate of 15.64% from 2025 to 2034.
After closely observing this market for over a decade, it is evident that traditional pharmaceutical logistics models are no longer sufficient. Cell and gene therapies demand precision, real-time visibility, and strong coordination between manufacturers, logistics providers, and healthcare facilities. The growth of this market is not driven by convenience or scale alone, but by the urgent need to protect therapies that can change or save lives.
Why Transportation Took the Lead in 2024
Transportation emerged as the largest service segment in the cell and gene therapy cold chain logistics market in 2024, holding around 48% of the total share. This leadership is not surprising. These therapies often operate within extremely tight time windows. Many treatments involve collecting patient samples, transporting them to manufacturing sites, and returning the final therapy for administration. Any delay can directly affect patient outcomes.
Over the years, transportation in this market has evolved into a highly controlled clinical process. It now involves validated routes, trained professionals, specialized containers, and continuous coordination. Speed alone is not enough. Logistics providers must ensure stable temperatures throughout the journey, even while crossing borders and regulatory checkpoints. As clinical trials expand globally, transportation has become the backbone that connects research, manufacturing, and patient care.
Monitoring and Tracking Move from Support to Necessity
While transportation led the market in size, monitoring and tracking solutions are growing at the fastest rate. This growth reflects a shift in mindset across the industry. Earlier, monitoring systems were mainly used to record temperature data for compliance. Today, they are essential tools for preventing losses before they happen.
Real-time monitoring allows stakeholders to detect delays, temperature deviations, or handling issues as they occur. This enables immediate action, such as rerouting shipments or replacing containers. As the value of cell and gene therapies continues to rise, companies are less willing to accept risk. Monitoring and tracking solutions provide confidence that product integrity is maintained throughout the journey.
Gene Therapies Drive the Largest Share of Demand
Gene therapies dominated the market by therapy type in 2024, accounting for nearly 46% of the total share. These therapies are extremely sensitive to environmental conditions and often require cryogenic temperatures to remain stable. Even minor temperature changes can damage the genetic material, making strict cold chain control essential.
The strong position of gene therapies is also linked to rapid growth in research and development. More gene therapies are entering clinical trials and early commercialization stages, increasing shipment volumes. At the same time, regulatory authorities closely monitor how these therapies are handled, adding further pressure on logistics systems to meet high compliance standards.
Cell Therapies Signal Strong Growth Ahead
Although gene therapies currently lead the market, cell therapies are expected to grow at the fastest pace in the coming years. This growth is driven by rising clinical trials, increasing use of immunotherapies, and the development of personalized treatments.
Cell therapies, especially autologous ones, create unique logistical challenges. Each shipment is linked to a specific patient, leaving no room for error or delay. As these therapies move closer to wider adoption, logistics providers must manage growing volumes while maintaining accuracy and control. This balance between scale and personalization will shape the next phase of the market.
Why Cryogenic Temperatures Dominate Cold Chain Needs
The cryogenic temperature segment, ranging from −150°C to −196°C, held the largest share of the market in 2024, with around 52%. This dominance exists because many cell and gene therapies require these temperatures to maintain their stability and functionality.
Cryogenic logistics involve specialized containers, liquid nitrogen systems, and trained personnel. Over time, these capabilities have become standard requirements rather than niche services. Regulatory expectations have also strengthened, especially for international shipments, making cryogenic transport a trusted and widely adopted solution.
Refrigerated Logistics Gain Acceptance Through Practical Benefits
While cryogenic solutions dominate, refrigerated logistics in the +2°C to +8°C range are expected to see the highest growth. This trend is closely linked to the rise of allogeneic therapies, which are generally less temperature-sensitive than autologous ones.
Refrigerated logistics are easier to handle, more cost-effective, and aligned with existing pharmaceutical infrastructure. As therapy developers improve formulations and stability profiles, refrigerated transport offers a practical balance between safety and efficiency. This shift shows how the market is becoming more flexible and therapy-specific rather than relying on one temperature standard.
Key Reasons Behind the Market Shift in the Middle of the Decade
The changes seen in the cell and gene therapy cold chain logistics market are driven by several connected factors that came together in 2024:
-
Growing clinical trials across multiple regions increased cross-border shipments
-
High-value, personalized therapies raised the cost of logistics failure
-
Stronger regulatory oversight demanded better documentation and traceability
-
Technology improvements enabled real-time monitoring and faster decision-making
These factors collectively pushed logistics providers to upgrade systems, skills, and processes.
Pharmaceutical and Biotechnology Companies Lead as End Users
Pharmaceutical and biotechnology companies remained the largest end-user segment in 2024, holding about 55% of the market. Their leadership reflects increased production, expanding pipelines, and higher research investment in cell and gene therapies.
These companies view cold chain logistics as an extension of their operations rather than an external service. They look for partners who understand therapy science, regulatory needs, and clinical timelines. As shipments increase in number and complexity, reliable logistics has become essential to maintaining development momentum.
Specialty Logistics Providers Gain Strong Momentum
Although pharmaceutical and biotechnology companies dominate in volume, specialty logistics providers are expected to grow the fastest. These providers focus specifically on temperature-sensitive and high-value therapies, offering customized solutions designed for cell and gene products.
Their strength lies in deep expertise, flexible operations, and the ability to manage risk across the entire supply chain. As therapy developers seek partners who can handle complexity without disruption, specialty providers are increasingly seen as strategic collaborators rather than service vendors.
Regulation and Risk Redefine Market Expectations
Regulatory requirements continue to shape how the cold chain logistics market operates. Authorities emphasize patient safety, data accuracy, and full traceability. Compliance is no longer a one-time task but an ongoing responsibility throughout the product journey.
Risk management has become central to logistics planning. Providers must prepare for delays, equipment failures, and unexpected disruptions. Those who can demonstrate reliability under pressure are gaining long-term trust and stronger market positions.
The Futue of Cold Chain Logistics
The future of the cell and gene therapy cold chain logistics market lies in better integration. Transportation, monitoring, regulatory compliance, and data analysis are increasingly connected into unified systems. This integration improves visibility, reduces errors, and supports faster decision-making.
However, technology alone is not enough. Human expertise remains critical. Understanding therapy behavior, anticipating challenges, and responding quickly to exceptions require experience built over years. As these therapies move closer to mainstream healthcare, logistics providers must combine advanced tools with skilled professionals.
A Market Built on Care, Not Noise
The growth of the cell and gene therapy cold chain logistics market is grounded in responsibility rather than hype. Each shipment represents a patient, years of research, and high expectations. In 2024, transportation, gene therapies, cryogenic solutions, and pharmaceutical companies defined the market’s foundation. At the same time, monitoring systems, cell therapies, refrigerated logistics, and specialty providers pointed toward the future.
Cold chain logistics is no longer just about moving products. It is about protecting hope, ensuring safety, and delivering the next generation of medicine with care and precision.
Access our exclusive, data-rich dashboard dedicated to the healthcare services market – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout this Market Report Now at: https://www.towardshealthcare.com/checkout/6098
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest
Table of Contents
Toggle



