From Support Function to Strategic Partner: The New Role of Cell and Gene Therapy CROs
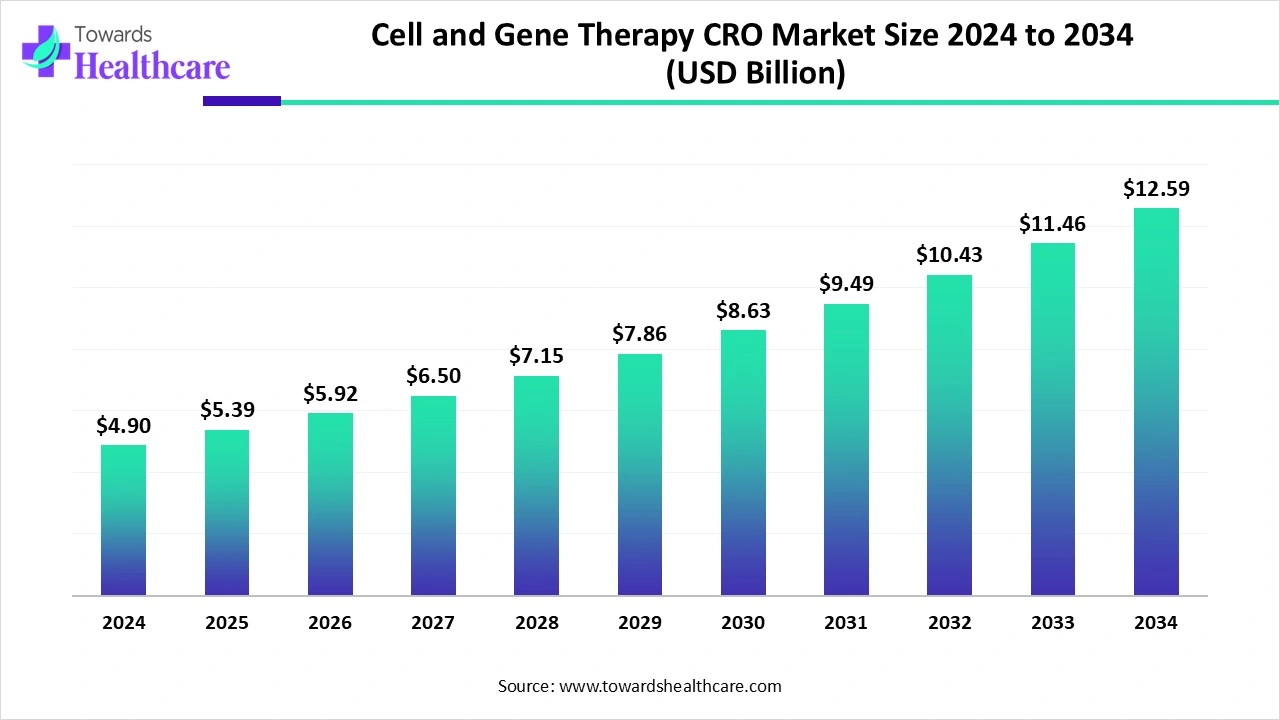
The cell and gene therapy CRO (contract research organization) market has moved far beyond its early experimental phase. What was once a niche service supporting a handful of academic trials has evolved into a sophisticated global ecosystem that underpins some of the most complex and high-stakes therapies in modern medicine. In 2024, the global market reached an estimated value of USD 4.90 billion, and the momentum shows no sign of slowing. By 2034, the market is expected to approach USD 12.59 billion, reflecting steady growth at a CAGR of nearly 10 percent over the coming decade.
𝐃𝐨𝐰𝐧𝐥𝐨𝐚𝐝 𝐅𝐫𝐞𝐞 𝐒𝐚𝐦𝐩𝐥𝐞:
https://www.towardshealthcare.com/download-sample/6095
This growth is not driven by hype. It is a direct response to mounting scientific complexity, escalating development costs, and increasingly demanding regulatory frameworks. Cell and gene therapies are not traditional drugs. They are living, evolving interventions that challenge every assumption the industry once held about clinical development, manufacturing, logistics, and patient management. As a result, sponsors are turning to specialized CROs not as vendors, but as strategic partners essential to success.
Why Outsourcing Has Become a Strategic Necessity
The rise of cell and gene therapy has exposed the limitations of traditional in-house development models. These therapies require expertise that spans viral vector analytics, gene editing technologies, autologous and allogeneic cell handling, long-term patient follow-up, and highly specialized regulatory pathways. Building all of these capabilities internally is neither cost-effective nor realistic for most organizations.
CROs fill this gap by offering integrated, therapy-specific solutions that accelerate development while maintaining compliance and quality. From IND-enabling studies and GLP toxicology to complex clinical operations and chain-of-identity logistics, specialized CROs now manage some of the most critical elements of the development lifecycle. Their role has shifted from execution to orchestration, coordinating science, operations, data, and regulatory strategy under one operational framework.
North America Sets the Pace, But the World Is Catching Up
North America continues to dominate the cell and gene therapy CRO market, largely due to its concentration of research-intensive biotech companies, robust funding environment, and mature clinical infrastructure. The region accounts for a substantial share of global CGT pipelines, and the United States remains the epicenter of innovation, clinical trials, and regulatory activity.
The U.S. market alone is projected to grow from USD 2.56 billion in 2026 to over USD 6 billion by 2035. This growth reflects sustained investment in advanced therapies, strong government support, and a healthcare system increasingly willing to adopt high-impact, one-time treatments for severe and rare diseases.
However, the most striking shift is happening in Asia Pacific. The region is emerging as the fastest-growing market, driven by proactive government policies, expanding clinical trial capacity, and a vast patient population. Countries such as China, Japan, South Korea, and India are no longer peripheral players. China, in particular, now hosts a significant share of global CAR-T clinical trials, signaling a structural shift in where advanced therapies are developed and tested.
Clinical Operations Remain the Backbone of the Market
Clinical operations and site management continue to dominate the service landscape, accounting for a significant portion of CRO revenues in 2024. This dominance is logical. Cell and gene therapy trials are operationally intensive, often involving small patient populations, single-site or hybrid trial designs, and highly individualized treatment workflows.
Managing these trials requires precise coordination across patient recruitment, regulatory approvals, data integrity, and ethical oversight. Errors are costly, not only financially but clinically. CROs with deep experience in advanced therapy trials provide the operational discipline needed to navigate these challenges, ensuring that studies progress efficiently without compromising patient safety or data quality.
At the same time, bioanalytics and assay development are emerging as the fastest-growing service segment. As therapies become more complex, the demand for sophisticated bioanalytical methods has surged. Sponsors increasingly rely on CROs that can deliver integrated bioanalytical support across preclinical, translational, and clinical phases, reducing handoffs and minimizing data inconsistencies.
Gene-Modified Cell Therapies Lead, While In-Vivo Approaches Accelerate
Gene-modified cell therapies remain the dominant therapy modality supported by CROs, reflecting the maturity and clinical success of approaches such as CAR-T and other ex-vivo gene-modified platforms. These therapies have demonstrated transformative potential in oncology and rare genetic diseases, driving sustained investment and clinical activity.
However, the fastest growth is occurring in in-vivo gene therapies. By delivering therapeutic genes directly into the patient’s body, these approaches aim to simplify treatment, reduce manufacturing complexity, and expand patient access. Advances in vector engineering and non-viral delivery systems have significantly improved safety and efficacy profiles, making in-vivo therapies one of the most closely watched segments in the CGT landscape.
For CROs, this shift demands continuous adaptation. Supporting in-vivo therapies requires different analytical tools, regulatory strategies, and clinical designs than ex-vivo platforms. Organizations that can flexibly support both modalities are positioning themselves for long-term relevance.
Biotech Innovators Drive Today’s Market, Big Pharma Shapes Tomorrow
Biotech and venture-backed innovators currently dominate CRO engagement in the cell and gene therapy space. These companies often operate with lean internal teams and aggressive timelines, making outsourcing essential. Many are pursuing first-in-class or best-in-class therapies for indications with limited or no existing treatments, placing a premium on speed, expertise, and regulatory precision.
Large pharmaceutical companies, however, are emerging as the fastest-growing sponsor segment. As CGTs transition from experimental to commercial reality, big pharma is increasingly acquiring, partnering with, or internally developing advanced therapy programs. Their entry brings scale, capital, and global commercialization expertise, but it also raises expectations for CRO performance, data rigor, and global coordination.
This dynamic is reshaping CRO offerings. Providers must now balance the agility demanded by biotech innovators with the process discipline and global reach required by large pharmaceutical clients.
Early-Phase Trials Anchor the Market, Pivotal Studies Drive Growth
Phase I and Phase I–II trials continue to represent the largest share of CRO activity in the CGT market. These early-phase studies are critical for establishing safety, dosing, and preliminary efficacy, particularly given the long-lasting and sometimes irreversible nature of gene-based interventions.
At the same time, Phase II and pivotal trials are emerging as the fastest-growing segment. As more therapies progress beyond proof of concept, sponsors require CROs capable of supporting larger, more complex studies designed to meet regulatory approval standards. Preparing for pivotal trials in cell and gene therapy requires a deep understanding of therapy-specific risks, long-term follow-up requirements, and manufacturing consistency, areas where experienced CROs provide significant value.
AI Moves From Experiment to Infrastructure
Artificial intelligence is no longer an emerging concept in the cell and gene therapy CRO market. It is becoming foundational infrastructure. CROs and sponsors are increasingly using AI to analyze large genomic datasets, identify predictive biomarkers, optimize patient selection, and improve trial outcomes.
In personalized medicine, AI-driven models help tailor therapies to individual patients, particularly in complex treatments such as CAR-T. AI also supports manufacturing optimization, helping reduce variability, improve yield, and lower costs. As proof of feasibility grows, partnerships between CROs, pharmaceutical companies, and AI technology providers are accelerating, further embedding digital intelligence into the CGT development process.
Regulatory Complexity Remains the Market’s Greatest Test
Despite rapid growth, the cell and gene therapy CRO market faces persistent challenges. Manufacturing costs remain high, supply chains are fragile, and regulatory requirements vary significantly across regions. The need for long-term safety monitoring adds another layer of complexity, extending trial timelines and increasing operational burden.
Moreover, investment tends to concentrate on commercially attractive indications, leaving ultra-rare diseases underfunded. CROs often operate at the intersection of innovation and constraint, helping sponsors navigate these challenges while maintaining scientific and ethical integrity.
Government Initiatives Expand Access and Opportunity
Government programs are playing an increasingly important role in shaping the market. Initiatives designed to improve access to cell and gene therapies, particularly for rare and severe diseases, are expanding the addressable market and encouraging broader adoption. These programs also create opportunities for CROs to support large-scale, multi-stakeholder initiatives that extend beyond traditional clinical trial models.
As healthcare systems grapple with the cost and complexity of advanced therapies, CROs are becoming key contributors to solutions that balance innovation with sustainability.
A Value Chain Built on Precision and Trust
The value chain for cell and gene therapy CROs spans R&D, clinical trials, and patient support, each stage requiring precision and coordination. From cell harvesting and gene transduction to patient infusion and long-term monitoring, every step carries high stakes. CROs that succeed in this environment do so by building trust, not only with sponsors but with regulators, investigators, and patients.
Leading players in the market have invested heavily in specialized infrastructure, scientific talent, and digital systems to support this end-to-end complexity. At the same time, boutique CROs continue to thrive by offering deep expertise in specific niches, proving that scale and specialization can coexist.
Looking Ahead: A Market Defined by Execution
The next decade will define the cell and gene therapy CRO market. Growth is assured, but success will depend on execution rather than ambition. CROs must continue to evolve alongside the therapies they support, integrating new technologies, adapting to shifting regulatory landscapes, and maintaining an unwavering focus on patient safety.
For sponsors, the choice of CRO is no longer a procurement decision. It is a strategic commitment that can determine the success or failure of an entire development program. As cell and gene therapies move closer to becoming standard of care for many conditions, the CROs supporting them will play a quiet but decisive role in shaping the future of medicine.
In this sense, the market is not merely expanding. It is maturing into one of the most critical pillars of the global biopharmaceutical industry, poised to define how innovation reaches patients in the years to come.
Access our exclusive, data-rich dashboard dedicated to the healthcare market – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout Cell and Gene Therapy CRO Market Report Now at: https://www.towardshealthcare.com/checkout/6095
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest
Table of Contents
Toggle



