Clinical trials have always been the backbone of pharmaceutical innovation, providing the evidence needed to bring new treatments to patients safely and effectively. However, the complexity of these trials, the enormous volume of data they generate, and the growing expectations from regulators, patients, and healthcare providers have pushed the industry to rethink how trials are designed, executed, and monitored. In recent years, two forces have emerged as game-changers in the clinical trial ecosystem: Artificial Intelligence (AI) and full-service Contract Research Organizations (CROs). Together, they are redefining timelines, improving efficiencies, and accelerating the future of clinical development.
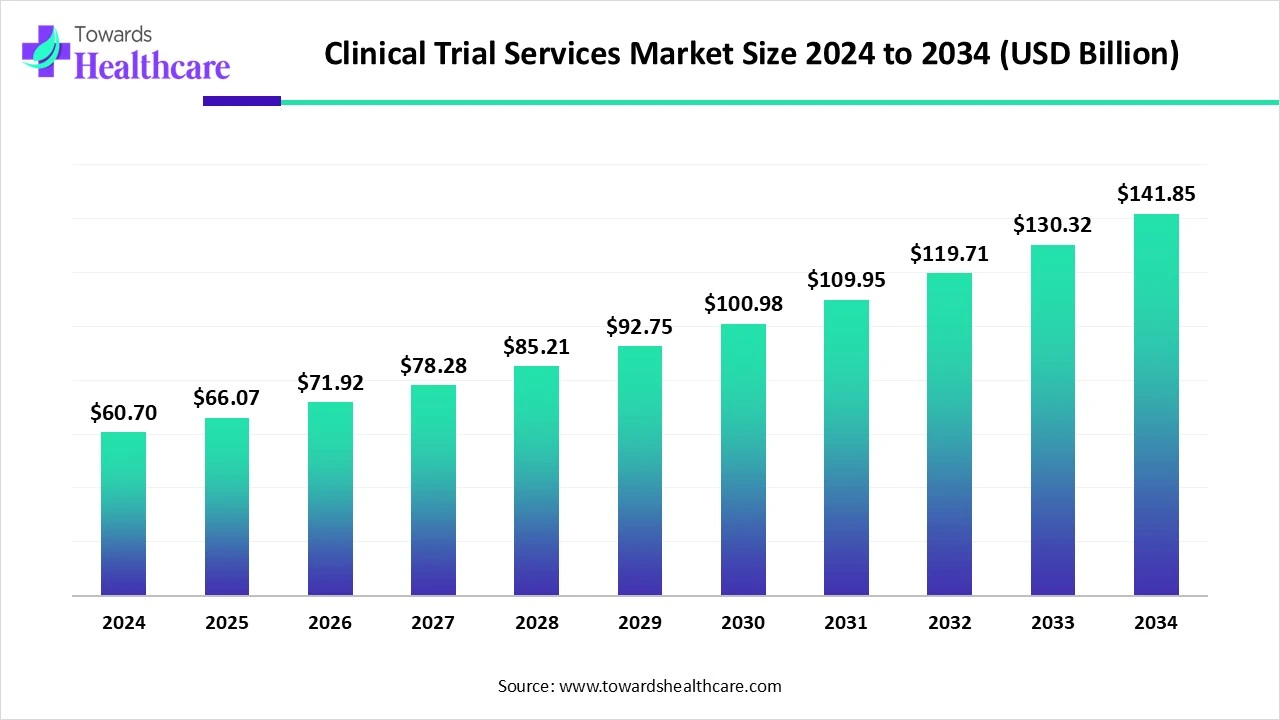
The global clinical trial services market reflects this transformation vividly. In 2025, the market was valued at approximately USD 66.07 billion and has already grown to an estimated USD 71.92 billion in 2026. Analysts project that this market could more than double by 2035, reaching nearly USD 154.28 billion, with a robust compound annual growth rate of 8.85% between 2026 and 2035. Such growth underscores the increasing reliance of pharmaceutical sponsors, biotechnology companies, and research institutions on specialized trial services to navigate the complexities of modern drug development.
Download Free Sample: https://www.towardshealthcare.com/download-sample/6031
The Market Pulse: What’s Driving the Growth?
The growth of clinical trial services is far from incidental. It is fueled by multiple intertwined trends, including the surge in research and development activities, the rising incidence of complex diseases such as cancer and rare disorders, and the proliferation of decentralized trial models. AI, in particular, is being leveraged to enhance the operational efficiency and reliability of trials. It allows CROs to analyze vast datasets, optimize patient recruitment, identify optimal trial sites, and even predict potential risks before they escalate. This capability is increasingly critical as trials become more complex, multi-centric, and global.
Full-service CROs, or end-to-end clinical development providers, are emerging as the preferred partners for sponsors who want a unified approach to trial management. These organizations offer comprehensive solutions that span protocol design, site management, patient recruitment, clinical monitoring, data management, regulatory affairs, pharmacovigilance, and study close-out. The holistic nature of these services helps sponsors maintain consistency, meet compliance requirements, and accelerate the overall timeline of clinical development. In 2024, the full-service segment accounted for nearly 46% of the market share, reflecting its dominance and the growing demand for integrated solutions.
Meanwhile, functional service provider (FSP) or resourcing models are rapidly gaining ground, driven by their flexibility and cost-effectiveness. These models allow sponsors to access specialized expertise on-demand, from biostatisticians to clinical research associates, without committing to a full-service contract. The FSP model is projected to be the fastest-growing segment during the next decade, offering a scalable alternative for increasingly complex and adaptive trial designs.
Phase II/III Trials: The Heart of Clinical Development
Clinical trials are stratified into different phases, each with unique challenges and requirements. Phase II and III trials are particularly significant, representing the largest patient cohorts and often involving multi-regional study sites. These phases are resource-intensive, both in terms of logistics and data management, which explains why they held nearly 58% of the market share in 2024. The high complexity of these trials, spanning protocol adherence, patient monitoring, regulatory compliance, and outcome measurement, necessitates the use of specialized trial services. AI-enabled CROs can process vast amounts of clinical data in real-time, ensuring accurate monitoring, early detection of errors, and predictive insights for trial adjustments.
While phase II/III dominates in scale, phase IV trials, which encompass real-world evidence (RWE) and post-marketing surveillance, are projected to be the fastest-growing segment. These studies focus on understanding patient quality of life, long-term treatment adherence, and post-approval safety. Decentralized trial methodologies, remote patient monitoring, and real-time data capture are critical in these studies, and AI is playing an essential role in making these processes seamless and reliable.
Therapeutic Areas Driving Innovation
Oncology has emerged as the largest therapeutic area for clinical trial services, holding nearly 39% of the market share in 2024. Cancer trials are inherently complex due to heterogeneous patient populations, high variability in treatment response, and the need for sophisticated biomarker-driven designs. Full-service CROs with integrated data platforms can navigate these challenges efficiently, reducing delays and improving trial reliability.
At the same time, rare and orphan diseases are expected to see the fastest growth during the forecast period. Limited patient populations, coupled with high unmet medical needs, require precision in patient identification, recruitment, and monitoring. AI applications, from electronic health record analysis to predictive recruitment modeling, are critical to the success of these trials. Full-service CROs are uniquely positioned to meet these needs by providing end-to-end services that integrate scientific, regulatory, and operational expertise.
AI as a Force Multiplier
Artificial Intelligence is no longer a futuristic concept for clinical trials; it is already embedded in many trial processes. Its applications are diverse: AI can scan global trial databases to identify optimal clinical sites, analyze patient eligibility from electronic health records, optimize protocol design using predictive modeling, and even monitor data in real-time to detect errors before they become critical. By automating routine tasks and providing actionable insights, AI reduces both timelines and operational costs, making clinical trials more scalable and reliable.
Beyond operational efficiency, AI contributes to better patient outcomes by ensuring trials are more patient-centric. By identifying suitable patients faster and predicting likely dropout risks, AI helps sponsors design protocols that are both scientifically rigorous and feasible for real-world implementation. Combined with full-service CRO capabilities, this technology enables a level of trial precision and adaptability that was previously unimaginable.
Global Dynamics: North America Leads, Asia Pacific Surges
Regionally, North America remains the largest market, accounting for approximately 41% of the global clinical trial services market in 2024. This dominance is driven by a mature CRO ecosystem, robust sponsor base, and extensive R&D investments. The U.S., in particular, benefits from a concentration of leading academic institutions, pharmaceutical giants, and biotechnology firms, all of which contribute to a high demand for clinical trial services. Canada, while smaller in scale, is seeing rapid adoption due to growing drug development activities and the integration of AI and big data analytics in clinical research.
Asia Pacific is emerging as the fastest-growing market. Expanding healthcare infrastructure, increasing government support, and rising investments in clinical research are driving growth. Countries such as China, India, and Japan are becoming preferred destinations for multi-regional trials, owing to their large patient populations and cost-effective clinical sites. In addition, advancements in digital health technologies and telemedicine are facilitating the adoption of decentralized trial models in these regions.
Challenges in Clinical Trial Services
Despite the positive trends, the clinical trial services market faces notable challenges. Increasing protocol complexity can significantly extend trial duration, drive up costs, and complicate patient recruitment. Regulatory requirements vary across regions, adding another layer of complexity. While AI and full-service CROs offer solutions, the human factor—training, compliance, and oversight—remains critical. Trial errors, data discrepancies, and patient dropouts are ongoing risks that must be managed proactively.
Another challenge is integrating diverse datasets from multiple sources. Clinical trials generate massive amounts of data, ranging from laboratory results to patient-reported outcomes. Unified data platforms are becoming essential to manage, reconcile, and analyze this information in real-time. The adoption of such platforms is growing, but standardization across regions and regulatory frameworks remains an area requiring ongoing innovation.
Sustainability and Strategic Investments
Sustainability in clinical trials is increasingly about efficiency, reliability, and patient-centric approaches rather than environmental metrics alone. Companies are investing heavily to optimize trial processes, reduce redundancies, and leverage technology to enhance outcomes. In July 2025, Veeda Lifesciences invested in Mango Sciences to integrate AI capabilities into its trial services, enhancing speed, quality, and efficiency. In March 2025, a $300 million joint venture between SPRIM Global Investments and One Investment Management aimed to expand financing solutions for trials across phases 1 to 3. Such investments highlight the strategic focus on accelerating clinical development and improving patient access to innovative therapies.
Full-Service Outsourcing: The Key to Seamless Trials
The full-service outsourcing model dominates the clinical trial services landscape, holding a 52% share in 2024. Sponsors benefit from end-to-end solutions that integrate protocol design, data management, patient recruitment, clinical monitoring, and regulatory compliance. This model reduces the risk of fragmented trial execution and allows for consistent, high-quality data collection across multiple sites. The alternative, functional outsourcing, is gaining traction due to its flexible and cost-efficient approach, particularly for sponsors seeking specialized expertise or adapting to trial-specific challenges.
Looking Ahead: The Future of Clinical Trial Services
The future of clinical trial services will be defined by a convergence of technology, strategic partnerships, and patient-centric design. AI will continue to evolve, moving from predictive analytics to prescriptive and adaptive solutions that can dynamically adjust trial parameters in real-time. Full-service CROs will expand their capabilities, integrating decentralized trial models, real-world data, and precision medicine approaches.
Therapeutic areas such as oncology, rare diseases, and immunology will continue to drive demand, while regions like Asia Pacific will emerge as hubs for multi-regional trials. Regulatory frameworks are likely to adapt to new technologies, enabling faster approvals and more flexible trial designs. The combination of AI-driven insights, full-service CRO expertise, and patient-centered methodologies will redefine how clinical trials are conducted, accelerating the path from laboratory discovery to patient access.
Conclusion: A New Era of Clinical Trials
The clinical trial services market stands at a pivotal moment. The integration of AI and full-service CROs is not just improving efficiency; it is transforming the very nature of clinical research. By optimizing trial design, accelerating patient recruitment, ensuring regulatory compliance, and enabling real-time monitoring, these innovations are setting new standards for speed, reliability, and scalability. The result is a more agile, responsive, and patient-centric ecosystem, capable of delivering life-saving therapies faster and more efficiently than ever before.
The coming decade promises remarkable growth and transformation in clinical trial services. Companies that embrace AI, leverage full-service solutions, and focus on innovation will lead the charge in reshaping the clinical research landscape. For patients, sponsors, and the healthcare ecosystem as a whole, this is more than a technological shift—it is the beginning of a new era where clinical trials are faster, smarter, and more impactful.
Access our exclusive, data-rich dashboard dedicated to the life science industry – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout Clinical Trials Services Market Report Now at: https://www.towardshealthcare.com/checkout/6031
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest




