A Quiet Revolution Reshaping How the World Tests New Medicines
For decades, clinical trials followed a rigid formula. Patients traveled to hospitals or research centers, adhered to strict visit schedules, and adjusted their lives around study protocols. That model delivered scientific rigor, but it also excluded millions of people who could not participate due to distance, disability, time constraints, or socioeconomic barriers. Over the past few years, that reality has begun to change. Decentralized clinical trials, commonly referred to as DCTs, are not merely a technological upgrade to traditional research. They represent a structural transformation in how evidence is generated, how patients engage with science, and how regulators evaluate innovation.
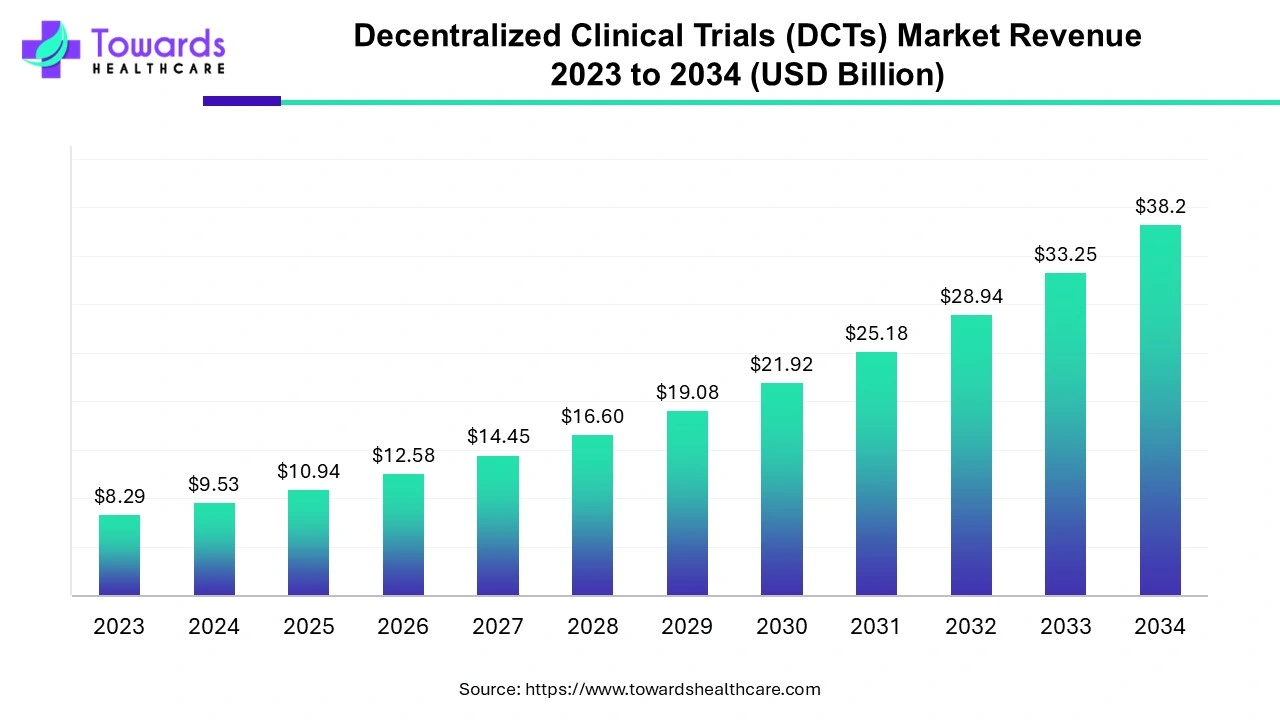
The global decentralized clinical trials market, valued at US$ 8.29 billion in 2023 and projected to reach US$ 38.2 billion by 2034, reflects more than financial growth. It signals a shift in mindset. Sponsors, regulators, healthcare providers, and patients increasingly view decentralization as a practical, ethical, and scientifically robust alternative to site-centric research. With a projected compound annual growth rate of 14.9 percent between 2024 and 2034, DCTs are moving from experimental pilots to mainstream clinical development strategies.
Download Free Sample Now and Get the Complete Report Easily at: https://www.towardshealthcare.com/download-sample/5264
From Hospital Corridors to Living Rooms
At its core, a decentralized clinical trial reduces or eliminates the need for participants to be physically present at a hospital or research site. Instead, trials rely on a combination of digital tools and localized services to conduct study activities. Wearable sensors track vital signs in real time. Mobile applications collect patient-reported outcomes. Electronic consent platforms replace paper-based forms. Telemedicine enables investigators to interact with participants remotely. Home healthcare professionals administer treatments or collect samples at the patient’s doorstep.
This approach does not remove rigor from clinical research. Instead, it redistributes it. Data collection becomes continuous rather than episodic. Patient behavior reflects real-world conditions rather than artificial clinical environments. For many indications, particularly chronic and long-term diseases, this shift improves both data quality and participant experience.
Decentralization also addresses one of the most persistent challenges in clinical research: recruitment and retention. Traditional trials often struggle to enroll diverse populations. Patients who live far from urban centers, elderly individuals, people with mobility limitations, and caregivers balancing work and family obligations frequently opt out. DCTs bring trials to these populations rather than expecting them to come to the trial.
Checkout Decentralized Clinical Trials (DCTs) Market Report Now at: https://www.towardshealthcare.com/checkout/5264
Why the Market Accelerated So Quickly?
The growth of decentralized clinical trials did not occur in isolation. It emerged at the intersection of technological readiness, regulatory openness, and systemic pressure on healthcare systems. The COVID-19 pandemic acted as a catalyst, forcing sponsors and regulators to adopt remote methods almost overnight. What began as an emergency response soon proved to be a viable long-term solution.
As healthcare systems recovered, many organizations chose not to revert entirely to pre-pandemic practices. Instead, they refined hybrid and fully decentralized models. Sponsors recognized that remote monitoring reduced trial delays. Regulators observed that patient safety could be maintained outside traditional sites. Patients experienced greater convenience and autonomy. These aligned incentives created momentum that continues to build.
North America emerged as the early leader in this transformation. Strong digital infrastructure, mature pharmaceutical and biotechnology sectors, and proactive regulatory bodies positioned the region at the forefront of adoption. The United States, in particular, played a defining role as the Food and Drug Administration publicly acknowledged the potential advantages of decentralized trials and issued guidance to support their implementation across drugs, biologics, and medical devices.
Regulators Step Into the Future
One of the most significant developments shaping the DCT landscape has been regulatory evolution. For years, uncertainty around compliance, data integrity, and patient safety slowed adoption. That uncertainty has steadily diminished.
Recent guidance updates from the FDA have clarified expectations around remote informed consent, technology validation, risk-based oversight, and the use of real-world data. These updates do not lower regulatory standards. Instead, they modernize them. Sponsors now have clearer pathways to design decentralized trials that meet regulatory requirements while remaining flexible and cost-efficient.
Canada followed a similar trajectory. Its Regulatory Innovation Agenda emphasizes modernization of clinical trial laws as a national priority. The pandemic accelerated the adoption of decentralized methods, and the country continues to integrate these approaches into its broader research ecosystem.
In Asia, regulatory momentum is gaining speed. Japan has introduced guidelines for electronic informed consent and the use of remotely collected data for safety and efficacy evaluations. Discussions in 2025 highlighted the need for clinical research hospitals to become DCT-capable, signaling a long-term commitment to decentralized infrastructure. China’s growing engagement with digital and decentralized trials further reinforces Asia Pacific’s position as the fastest-growing regional market.
Asia Pacific’s Rapid Rise
While North America currently dominates the global decentralized clinical trials market, Asia Pacific is expected to grow at the fastest rate over the forecast period. Several factors drive this expansion. The region faces a rising burden of chronic diseases such as diabetes, cardiovascular disorders, cancer, and infectious diseases. Healthcare systems seek scalable research models that can keep pace with this demand.
Asia Pacific also offers large and diverse patient populations, making it an attractive destination for global clinical research. Decentralized models allow sponsors to reach these populations more effectively while reducing infrastructure constraints. As digital health adoption increases across countries such as China, Japan, India, and South Korea, the operational feasibility of DCTs continues to improve.
China’s engagement with decentralized trials reflects a broader national focus on digital health innovation. The publication of expert consensus documents on decentralized and digitalized clinical trials underscores growing institutional support. In the Chinese context, digitalized trials and decentralized trials are often used interchangeably, highlighting how deeply technology is embedded in the research narrative.
Europe’s Steady Expansion
Europe occupies a distinct position in the decentralized clinical trials landscape. Rather than rapid disruption, the region demonstrates steady, policy-supported growth. Increasing investment in research and development across diagnostics and therapeutics drives demand for more flexible trial models. Countries such as the United Kingdom, Germany, and France actively support innovation through regulatory guidance and public funding.
In the United Kingdom, industries developing new treatment approaches increasingly rely on decentralized methods to accelerate testing and approval. Germany’s focus on innovation, combined with government support, further strengthens adoption. Across Europe, regulatory alignment and cross-border collaboration play a critical role in enabling decentralized trials at scale.
Interventional Trials Take the Lead
Among the different types of decentralized clinical trials, interventional studies dominate the market and are expected to grow at the fastest rate. These trials form the backbone of medical progress. They provide the evidence required to determine whether new therapies outperform existing standards of care.
Decentralization enhances the feasibility of interventional trials by reducing logistical complexity. Remote monitoring allows investigators to track safety and efficacy more frequently. Home-based interventions improve adherence. Patients remain engaged for longer periods because participation interferes less with daily life.
Regulators continue to rely on interventional trial data for approval decisions, making this segment central to the future of decentralized research. As sponsors gain confidence in remote methodologies, interventional DCTs are likely to expand across therapeutic areas.
Oncology Drives Adoption Through Necessity
Cancer research occupies a unique place within the decentralized clinical trials market. Oncology dominated application segments in 2023, reflecting both scientific urgency and practical need. Cancer remains one of the most pressing global health challenges, and the demand for innovative treatments continues to grow.
Approximately 85 percent of cancer care occurs within the community rather than specialized research centers. Decentralized trials align with this reality by bringing research closer to where patients receive care. Local healthcare professionals can participate in trial activities, improving access and equity. Patients benefit from reduced travel and greater continuity between standard care and research participation.
Decentralization also supports longitudinal monitoring, which is essential in oncology. Wearable devices and digital reporting tools capture real-world outcomes that traditional site visits may miss. This depth of data enhances understanding of treatment effectiveness and tolerability.
Cardiovascular Research Finds New Momentum
Cardiovascular disease remains the leading cause of death worldwide, and the need for continuous innovation persists. Decentralized clinical trials offer particular advantages in this domain. Traditional cardiovascular trials often require large sample sizes, long durations, and substantial funding. These requirements can limit feasibility.
Decentralized approaches address these challenges by improving recruitment, retention, and data efficiency. Home-based monitoring captures daily fluctuations in heart rate, activity, and other vital parameters. Remote engagement reduces patient burden, enabling participation over extended periods.
As healthcare systems increasingly emphasize home-based care, cardiovascular DCTs align with broader trends in chronic disease management. This alignment positions the cardiovascular segment for significant growth during the forecast period.
Technology Redefines the Trial Experience
Artificial intelligence and machine learning are reshaping decentralized clinical trials in profound ways. Advanced analytics process vast volumes of data generated by wearables, sensors, and electronic patient-reported outcomes. These tools enable real-time insights, predictive modeling, and adaptive trial designs.
AI-driven monitoring improves patient safety by identifying anomalies early. Machine learning algorithms help optimize trial protocols, potentially reducing sample sizes and shortening timelines. These efficiencies lower costs while maintaining scientific validity.
Partnerships between technology providers and clinical research platforms illustrate this evolution. Collaborations that integrate cloud infrastructure, AI capabilities, and patient-centric design signal a future in which trials become more responsive and intelligent. As computing power and algorithmic sophistication continue to advance, AI will play an increasingly central role in decentralized research.
The Persistent Challenge of Data Privacy
Despite its promise, decentralized clinical research faces complex operational challenges. Data privacy and compliance remain among the most critical. Remote data collection introduces new risks related to authentication, security, and jurisdictional variability. Privacy laws differ across countries and even across regions within the same country.
Moving data collection from controlled clinical environments into patients’ homes increases exposure to potential breaches. Sponsors must implement robust security frameworks, clear consent processes, and transparent data governance models. Regulatory guidance increasingly emphasizes these requirements, but implementation remains demanding.
Balancing innovation with trust is essential. Patients must feel confident that their data is secure and used responsibly. As decentralized trials expand globally, harmonizing privacy standards will become a priority for regulators and industry stakeholders alike.
Industry Players Shape the Ecosystem
The decentralized clinical trials market includes a diverse set of participants, from global contract research organizations to specialized technology providers. Companies such as IQVIA, Labcorp, ICON, Parexel, and Thermo Fisher Scientific bring operational scale and regulatory expertise. Technology-focused firms such as Medable, Science 37, Oracle, and Medidata contribute digital platforms that enable decentralization.
Recent product launches and partnerships highlight ongoing innovation. Integrated platforms that combine electronic outcomes assessment, consent management, telemedicine, and sensor integration simplify trial execution. Site networks designed specifically for decentralized research help standardize quality and adoption.
These developments reflect an ecosystem approach rather than isolated solutions. Successful decentralized trials require coordination among sponsors, sites, technology vendors, regulators, and patients. Companies that understand this interconnectedness are shaping the future of clinical research.
A Market That Reflects a Philosophical Shift
Decentralized clinical trials represent more than a market opportunity. They embody a philosophical shift toward patient-centric research. By meeting participants where they are, DCTs challenge long-standing assumptions about how trials must operate.
This shift does not eliminate the need for traditional sites or expert oversight. Instead, it expands the toolkit available to researchers. Hybrid models that combine site-based and decentralized elements are likely to become the norm. Fully decentralized trials will continue to grow as technology and regulation evolve.
The projected expansion of the global DCT market through 2034 underscores this trajectory. Growth is driven not only by investment and innovation but also by necessity. Healthcare systems face increasing pressure to deliver faster, more inclusive, and more efficient research outcomes. Decentralization offers a credible path forward.
Access our exclusive, data-rich dashboard dedicated to the healthcare market – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Quick Links:
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout Decentralized Clinical Trials (DCTs) Market Report Now at: https://www.towardshealthcare.com/checkout/5264
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest




