GLP-1 Small Molecule Market in North America, Asia Pacific & Europe: The Next Metabolic Revolution?
The global healthcare industry is witnessing a defining moment. The GLP-1 small molecule market is not merely expanding; it is reshaping the way the world approaches diabetes, obesity, and metabolic disorders. Between 2026 and 2035, this market is expected to accumulate hundreds of millions in revenue, driven by scientific innovation, strategic investments, and rising global demand.
What once began as a targeted therapeutic approach for Type 2 diabetes is now transforming into a multi-dimensional treatment strategy with applications spanning cardiometabolic health, obesity management, and even neurodegenerative and cardiovascular conditions. From North America’s regulatory momentum to Asia Pacific’s expanding patient base and Europe’s preference for oral formulations, the GLP-1 small molecule landscape is entering a new era of accelerated evolution.
Download Free Sample: https://www.towardshealthcare.com/download-sample/6550
Table of Contents
ToggleThe Science Behind the Momentum: Why GLP-1 Small Molecules Matter
Glucagon-like peptide-1 (GLP-1) therapies have already proven their value in managing blood glucose and supporting weight loss. However, traditional GLP-1 treatments primarily rely on injectable peptide-based drugs. Small molecule GLP-1 therapies introduce a new possibility: oral dosing, greater chemical stability, easier manufacturing, and potentially broader patient adoption.
Small molecules offer advantages in scalability and distribution. They often demonstrate improved storage stability and reduced dependency on cold chain logistics. These factors collectively increase accessibility, especially in emerging markets.
As global obesity rates surge and Type 2 diabetes cases rise sharply, healthcare systems are actively searching for more practical and scalable therapeutic solutions. GLP-1 small molecules stand at the intersection of innovation and necessity.
The Global Metabolic Burden Is Redefining Market Priorities
Urbanization, dietary changes, and sedentary lifestyles continue to accelerate metabolic disorders worldwide. Countries across Asia, North America, and Europe report rising prevalence of Type 2 diabetes and obesity.
In India and China alone, millions of new cases emerge each year. Healthcare systems face mounting pressure to provide affordable, effective, and scalable treatment options. At the same time, patients increasingly prefer therapies that integrate seamlessly into daily life. Oral GLP-1 small molecules align precisely with these evolving needs.
The market’s projected growth reflects not only commercial opportunity but also a pressing global health requirement.
North America: Regulatory Agility and High Disease Prevalence Drive Leadership
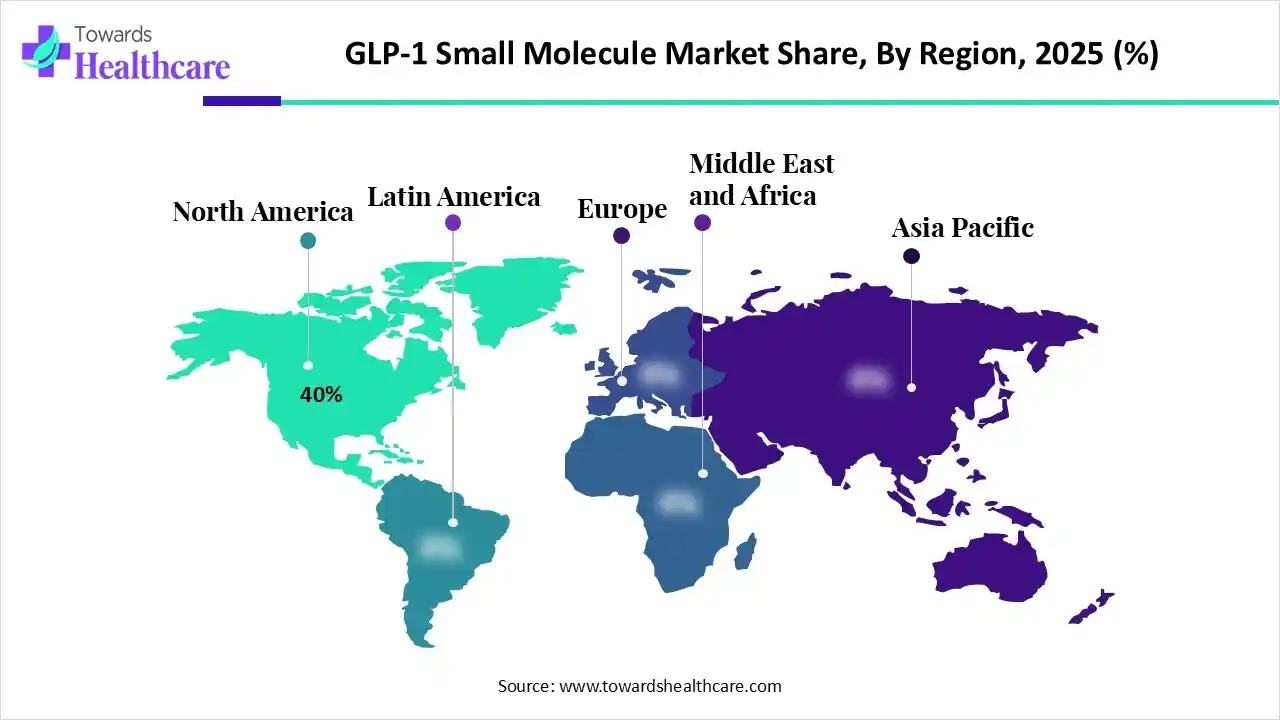
North America captured nearly 40% of the GLP-1 small molecule market share in 2025. The region’s dominance stems from a convergence of factors: high prevalence of obesity and Type 2 diabetes, strong healthcare infrastructure, favorable insurance coverage, and accelerated regulatory approvals.
The United States plays a central role in this growth trajectory. Rising metabolic disease rates have significantly expanded the patient base. Moreover, patients increasingly seek pharmacological alternatives to bariatric surgery, boosting demand for advanced drug therapies.
Regulatory momentum has also strengthened the market. The active involvement of the U.S. Food and Drug Administration has streamlined approval processes for innovative metabolic treatments. Faster review cycles encourage pharmaceutical companies to invest heavily in research and development.
Manufacturing Investments Signal Long-Term Confidence
In September 2025, Eli Lilly and Company announced plans to establish a $6.5 billion manufacturing facility at Generation Park in Houston. The project underscores the company’s commitment to expanding its small molecule pipeline across cardiometabolic health, oncology, immunology, and neuroscience.
Such investments reflect strong industry confidence in small molecule therapies. Large-scale manufacturing infrastructure ensures supply continuity, scalability, and long-term market sustainability.
Evolving Care Models Accelerate Adoption
Telehealth platforms and digital health services have further strengthened GLP-1 adoption in the United States. During supply constraints and healthcare access disruptions, telemedicine played a key role in maintaining treatment continuity.
Collaborations also shape the market landscape. In September 2025, Wellgistics Health, Inc. partnered with TheracosBio, manufacturer of Brenzavvy (bexagliflozin), to enhance affordability and accessibility of diabetes medications across the U.S. healthcare system. While not a GLP-1 product itself, such partnerships reflect broader industry strategies to optimize metabolic disease management ecosystems.
North America’s leadership arises not only from demand but from coordinated regulatory, manufacturing, and digital health frameworks.
Asia Pacific: Rapid Expansion Fueled by Scale and Innovation
Asia Pacific is anticipated to expand at a CAGR of approximately 24–28% during the forecast period. No other region combines such a vast patient population with accelerating pharmaceutical innovation.
India and China face enormous metabolic health challenges. Rapid urbanization, changing food habits, and sedentary lifestyles have dramatically increased obesity and diabetes rates. Governments and private players alike recognize the urgency of scalable treatment solutions.
Local Pharmaceutical Powerhouses Step Forward
Indian and Chinese pharmaceutical companies are actively investing in both generic and novel GLP-1 formulations to meet domestic demand while maintaining cost efficiency.
Companies such as Biocon Ltd, Tonghua Dongbao Pharmaceutical, and Sciwind Biosciences Co. are strengthening regional capabilities in metabolic drug development.
China, in particular, demonstrates significant innovation momentum. Sciwind Biosciences has advanced Ecnoglutide, a GLP-1 peptide therapy that delivered robust Phase 3 results, with patients experiencing average weight reductions of 9% to 13% after 40 weeks. Although peptide-based, such breakthroughs signal China’s growing expertise in GLP-1 research, which can accelerate small molecule advancements as well.
Cost Sensitivity Encourages Small Molecule Adoption
Small molecule therapies often offer manufacturing efficiencies compared to peptide injectables. In cost-sensitive markets such as India and China, this advantage becomes critical.
Healthcare systems aim to balance affordability with therapeutic effectiveness. Small molecules provide a promising path forward by enabling broader distribution and potentially lower production costs.
Asia Pacific’s growth trajectory reflects both necessity and opportunity. The region’s expanding middle class, improving healthcare infrastructure, and domestic innovation pipelines create fertile ground for sustained expansion.
Europe: Oral Preference and R&D Drive Structural Growth
Europe stands poised for significant growth in the GLP-1 small molecule market during 2026–2035. Patients across European countries increasingly prefer oral medications over injectable therapies, particularly for long-term metabolic management.
Adherence plays a crucial role in chronic disease control. Oral dosing improves patient compliance and simplifies treatment routines, especially among elderly populations.
Expanding Production Capacity Strengthens Supply Chains
In November 2025, Eli Lilly and Company announced plans to invest $3 billion in a new production facility in Katwijk, located within Leiden Bio Science Park. The facility aims to enhance oral medicine production capacity and reinforce global supply chains.
This investment signals Europe’s strategic importance in scaling oral small molecule manufacturing.
Germany Emerges as a Strategic Manufacturing Hub
Germany is predicted to grow at a notable CAGR through 2035. Strong research infrastructure and advanced pharmaceutical manufacturing capabilities position the country as a regional leader.
Companies such as CordenPharma are expanding peptide manufacturing facilities in Switzerland to meet rising demand from U.S. and European markets. While focused on peptides, these infrastructure expansions contribute to the broader GLP-1 ecosystem and foster technological spillover into small molecule development.
European growth reflects a combination of patient preference, regulatory clarity, and advanced pharmaceutical ecosystems.
Beyond Diabetes: Expanding Therapeutic Horizons
The GLP-1 small molecule market is no longer confined to glycemic control and weight management. Researchers increasingly explore applications in:
-
Cardiovascular protection
-
Neurodegenerative conditions
-
Metabolic syndrome
-
Inflammatory disorders
Clinical evidence suggests GLP-1 receptor activation may deliver cardioprotective benefits. Pharmaceutical companies are investing heavily in trials to expand labeled indications.
Regulatory agencies such as the European Medicines Agency continue to evaluate new applications and combination therapies, reinforcing structured oversight and accelerating approvals when clinical outcomes justify expansion.
Diversification across therapeutic areas reduces market concentration risk and enhances long-term sustainability.
Competitive Landscape: Strategic Shifts Shape the Next Decade
The competitive environment within the GLP-1 small molecule market continues to intensify. Large pharmaceutical corporations invest in vertically integrated manufacturing capabilities. Mid-sized biotech firms focus on innovative mechanisms and dual-target formulations.
Dual-target GLP-1 small molecules—designed to influence additional metabolic pathways—represent a particularly promising area of development. These therapies may deliver enhanced weight loss and glycemic control while reducing side effects.
Strategic collaborations, licensing agreements, and regional partnerships define the market’s structure. Companies prioritize pipeline diversification and global distribution networks to maintain competitive advantage.
Supply Chains, Scalability, and the Role of Infrastructure
Manufacturing scale will determine long-term market leadership. Small molecules offer logistical advantages compared to injectable biologics, but scaling production still requires advanced facilities and regulatory compliance.
Recent billion-dollar facility announcements across North America and Europe demonstrate how companies aim to secure supply resilience. Asia Pacific’s growing domestic manufacturing capacity also reduces dependency on imports.
Supply chain diversification enhances risk mitigation and ensures consistent availability amid fluctuating global demand.
The Patient-Centric Shift: Convenience Drives Growth
Patients increasingly demand treatments that fit seamlessly into daily routines. Oral small molecule therapies eliminate injection discomfort and reduce dependency on clinical administration.
Improved convenience leads to stronger adherence. Stronger adherence leads to better outcomes. Better outcomes reinforce market growth.
Healthcare providers also prefer therapies that reduce hospital visits and long-term complications. GLP-1 small molecules align with value-based care models emphasizing prevention and sustained metabolic control.
Investment Trends and Capital Flow
Venture capital and institutional investments continue to flow into metabolic research. Investors recognize the long-term demand trajectory driven by demographic and lifestyle trends.
Between 2026 and 2035, the market is projected to accumulate hundreds of millions in revenue. This projection reflects not speculative hype but measurable demographic and clinical realities.
Pharmaceutical companies are rebalancing portfolios toward metabolic therapies, often reallocating capital from lower-growth therapeutic areas.
Challenges That May Influence the Growth Curve
Despite strong momentum, the GLP-1 small molecule market faces certain constraints:
-
Pricing pressures in cost-sensitive regions
-
Regulatory complexity in multi-indication approvals
-
Competition from established injectable GLP-1 therapies
-
Safety monitoring in long-term usage
Companies must address these factors strategically to sustain growth.
Robust clinical data, transparent pricing strategies, and patient education campaigns will shape adoption rates.
The Decade Ahead: Strategic Outlook 2026–2035
From 2026 onward, the GLP-1 small molecule market will likely evolve along three strategic dimensions:
-
Therapeutic Diversification – Expansion beyond diabetes into cardiovascular and neurological domains.
-
Regional Manufacturing Expansion – Increased infrastructure investments across North America, Europe, and Asia Pacific.
-
Cost Optimization – Development of generics and competitive formulations to improve accessibility in emerging markets.
Asia Pacific’s rapid CAGR reflects structural demand. North America’s dominance demonstrates regulatory and infrastructural maturity. Europe’s growth highlights patient-centric therapeutic evolution.
Together, these regions define a globally interconnected yet regionally distinct growth landscape.
Conclusion: A Structural Shift in Metabolic Healthcare
The GLP-1 small molecule market in North America, Asia Pacific, and Europe represents more than a commercial expansion—it marks a structural shift in chronic disease management.
Rising metabolic disorders demand scalable, effective, and patient-friendly solutions. Small molecule GLP-1 therapies meet these requirements by combining scientific innovation with manufacturing practicality.
As companies invest billions in infrastructure and R&D, regulatory agencies accelerate approvals, and patients increasingly prefer oral therapies, the next decade promises transformative growth.
The GLP-1 small molecule market does not simply respond to demand—it actively reshapes the global strategy for managing metabolic disease.
Access our exclusive, data-rich dashboard dedicated to the therapeutic area sector– built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout GLP-1 Small Molecule Market Report Now at: https://www.towardshealthcare.com/checkout/6550
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest