Are GLP-1 Small Molecules the Next Metabolic Breakthrough?

GLP-1 Small Molecules
The global conversation around metabolic health is changing fast, and at the center of this shift sits a new class of therapies that few people discussed seriously a decade ago: GLP-1 small molecules. Once considered an ambitious alternative to injectable peptide drugs, these orally administered compounds are now reshaping how scientists, clinicians, regulators, and patients think about diabetes, obesity, and cardiometabolic disease management. What began as an experimental pursuit has evolved into one of the most strategically important battlegrounds in the pharmaceutical industry.
Over the last ten years, I have watched multiple therapeutic “next big things” rise and fall. Very few categories sustain momentum across science, regulation, investment, and patient demand simultaneously. GLP-1 small molecules are doing exactly that. Their emergence reflects not just scientific ingenuity, but also a deeper understanding of patient behavior, healthcare economics, and long-term disease burden.
Download Free Sample Now and Get the Complete Report Easily at: https://www.towardshealthcare.com/download-sample/6550
Table of Contents
ToggleWhy the World Needed an Oral GLP-1 Reset
Injectable GLP-1 receptor agonists changed the standard of care for type 2 diabetes and obesity. They delivered strong glycemic control, meaningful weight loss, and additional cardiovascular benefits that older therapies struggled to match. However, injections came with real-world frictions. Many patients delayed initiation, discontinued therapy early, or avoided treatment altogether due to needle aversion, storage requirements, gastrointestinal side effects, or access limitations.
Small molecule GLP-1 drugs address these friction points head-on. They offer oral dosing, room-temperature stability, simpler logistics, and the potential for lower manufacturing costs at scale. More importantly, they fit naturally into daily routines, which improves adherence and persistence. In chronic metabolic diseases, adherence is not a convenience metric; it is the difference between disease control and disease progression.
This shift toward oral GLP-1 therapies is not a cosmetic upgrade. It represents a structural correction to how metabolic diseases are treated globally, especially in regions where injectable therapies remain financially or logistically out of reach.
The Market Momentum Behind the Science
The GLP-1 small molecule market is entering a high-growth phase between 2026 and 2035, with forecasts pointing toward hundreds of millions in cumulative revenue. This growth is not speculative hype. It is anchored in epidemiological reality. Diabetes and obesity rates continue to rise across developed and developing economies, with Asia Pacific carrying one of the heaviest future burdens.
India and China, in particular, stand at the epicenter of this transformation. Rapid urbanization, sedentary lifestyles, and dietary shifts have driven an alarming increase in metabolic disorders. These markets demand scalable, affordable, and patient-friendly solutions. Oral GLP-1 small molecules align perfectly with those needs, making them a natural fit for large-population healthcare systems.
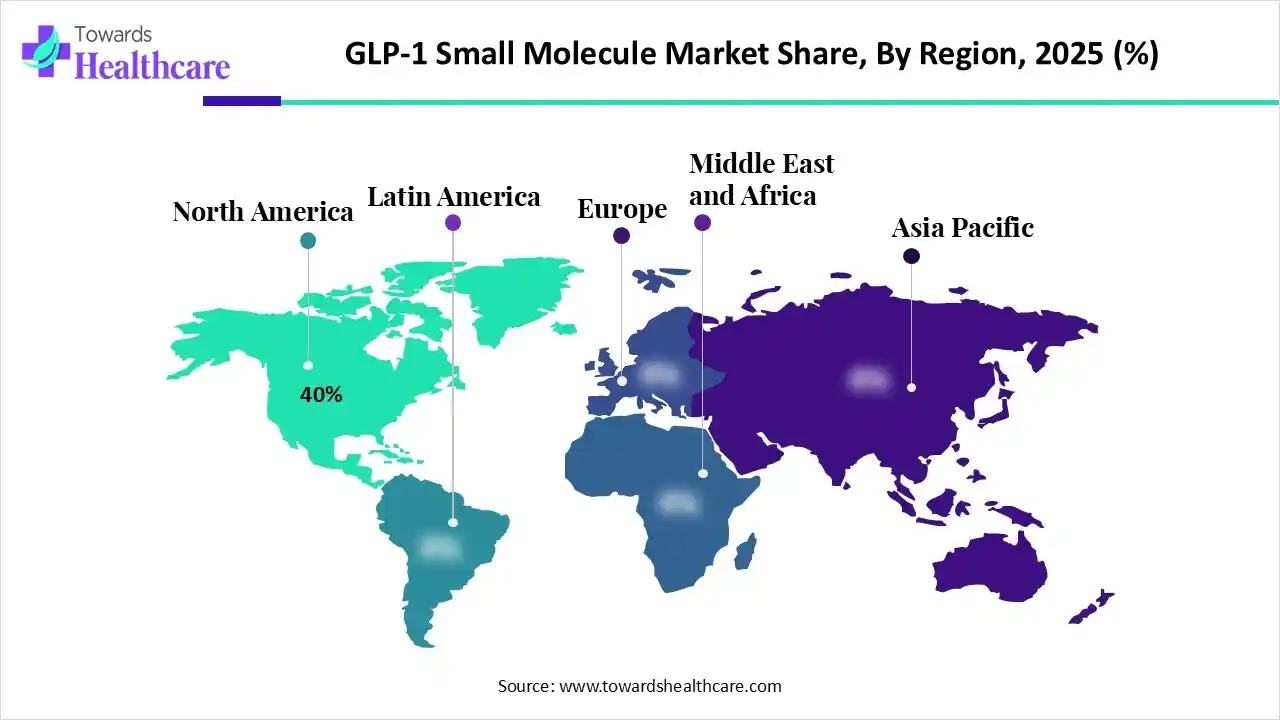
North America currently leads the market, capturing close to forty percent of global revenue in 2025. This dominance stems from early adoption, strong insurance coverage, aggressive R&D pipelines, and faster regulatory pathways. However, the fastest growth trajectory belongs to Asia Pacific, where annual growth rates are expected to significantly outpace mature markets over the next decade.
Beyond Diabetes: A Broader Therapeutic Horizon
One of the most underappreciated aspects of GLP-1 small molecules is how quickly their clinical relevance is expanding beyond diabetes and obesity. Early development focused narrowly on glycemic control. Today, researchers are exploring their role in cardiovascular risk reduction, fatty liver disease, kidney protection, and even neurodegenerative conditions.
This expansion reflects a deeper understanding of the GLP-1 receptor pathway as a systemic regulator rather than a glucose-specific switch. Full GLP-1 receptor agonism influences appetite signaling, insulin secretion, glucagon suppression, lipid metabolism, inflammation, and cellular survival pathways. When these effects are delivered through orally available small molecules, the therapeutic canvas becomes much broader.
Recent regulatory approvals and late-stage trials reinforce this direction. The industry is no longer asking whether GLP-1 therapies work beyond diabetes. It is asking how far their benefits can extend and which patient populations should be prioritized next.
Dual and Multi-Pathway Strategies Take Center Stage
Single-pathway solutions rarely dominate long-term metabolic care. The industry learned this lesson through insulin sensitizers, DPP-4 inhibitors, and early incretin therapies. GLP-1 small molecule development has absorbed that history and moved decisively toward dual and multi-pathway mechanisms.
Dual-target GLP-1 molecules that combine GLP-1 receptor agonism with GIP or glucagon receptor activity aim to deliver deeper weight loss, improved insulin sensitivity, and enhanced metabolic flexibility. These compounds are not incremental upgrades. They represent a strategic attempt to replicate, and potentially exceed, the metabolic benefits seen with injectable poly-agonist peptides while preserving oral convenience.
Multi-pathway small molecules take this concept further by biasing intracellular signaling toward pathways associated with efficacy while minimizing those linked to side effects. This level of receptor engineering was nearly impossible to achieve reliably a decade ago. Advances in computational chemistry, receptor modeling, and AI-assisted drug design have changed that equation.
How Artificial Intelligence Is Quietly Rewriting Drug Discovery
Artificial intelligence is no longer an auxiliary tool in GLP-1 small molecule development. It has become a core engine of innovation. Designing small molecules that can mimic the complex biological behavior of large peptide hormones is a non-trivial challenge. Traditional trial-and-error chemistry approaches struggled with receptor specificity, bioavailability, and safety margins.
AI-driven modeling has accelerated the identification of viable non-peptide agonists by predicting receptor binding conformations, signaling bias, and pharmacokinetic behavior before synthesis begins. Generative models now propose molecular structures that human chemists might never consider, shortening development timelines and reducing attrition rates.
AI also plays a growing role in formulation science. Nanoplatforms, polymer-based carriers, and smart hydrogels are being optimized using machine learning to improve stability, absorption, and controlled release. These innovations directly address one of the historical weaknesses of oral GLP-1 therapies: consistent bioavailability.
Oral Delivery Becomes the Market’s Anchor
Oral tablets currently dominate the GLP-1 small molecule market, capturing the majority of revenue in 2025. This dominance is logical. Tablets are familiar, scalable, and cost-efficient. They integrate seamlessly into existing pharmacy supply chains and do not require specialized handling.
What is more interesting is the momentum building behind alternative oral delivery formats. Encapsulated nanoparticles, liposomal systems, and pH-responsive hydrogels are gaining attention for their ability to protect active molecules through the gastrointestinal tract and release them at targeted sites. These technologies may not replace tablets, but they will expand the therapeutic toolkit, particularly for next-generation molecules with more complex absorption profiles.
Pharmacies, Platforms, and the Patient Experience
Retail pharmacies remain the primary distribution channel for GLP-1 small molecules, benefiting from their accessibility and the trusted role pharmacists play in chronic disease management. Even with oral therapies, patient education matters. Dosing schedules, side effect management, and lifestyle integration still require human guidance.
At the same time, online pharmacies and telehealth platforms are emerging as powerful growth engines. Digital consultations, home delivery, and integrated patient support systems reduce friction and expand access, especially for working populations and underserved regions. The rapid growth of this channel reflects broader changes in healthcare delivery rather than GLP-1-specific dynamics.
Regional Forces Shaping the Competitive Landscape
North America’s leadership is rooted in its innovation ecosystem. Large pharmaceutical companies continue to invest billions in manufacturing capacity, pipeline expansion, and regulatory readiness. The region also benefits from strong data infrastructure, which supports long-term outcomes research and payer adoption.
Asia Pacific’s growth story is different but equally compelling. Local pharmaceutical companies are investing aggressively in generics, biosimilars, and novel formulations tailored to regional needs. Cost sensitivity, scale, and speed matter more here than premium pricing. Governments and regulators are increasingly supportive of domestic innovation that reduces reliance on imported therapies.
Europe occupies a strategic middle ground. Patient preference for oral therapies, combined with strong regulatory oversight and advanced formulation expertise, positions the region as a key hub for next-generation GLP-1 development and manufacturing.
The Value Chain Evolves with the Molecule
GLP-1 small molecules simplify parts of the pharmaceutical value chain while complicating others. Compared to peptides, they reduce dependence on cold-chain logistics and sterile injectables manufacturing. This lowers barriers to entry and improves scalability.
However, they increase the importance of advanced medicinal chemistry, formulation science, and post-marketing surveillance. Small molecules may be easier to manufacture, but their long-term safety profiles require careful monitoring, especially as indications expand into non-diabetic populations.
Patient support services are also evolving. Successful GLP-1 therapies increasingly integrate lifestyle guidance, digital monitoring, and adherence programs. The molecule alone no longer defines success; the ecosystem around it does.
Strengths, Risks, and Strategic Tensions
GLP-1 small molecules offer undeniable advantages. They improve convenience, enhance adherence, reduce logistical complexity, and open doors to broader indications. Their potential role in neurological disorders, liver disease, kidney protection, and addiction treatment suggests that their impact will extend far beyond weight and glucose control.
At the same time, risks remain. Gastrointestinal side effects persist, and concerns around pancreatitis, thyroid health, and muscle mass loss demand long-term evidence. Regulatory scrutiny will intensify as usage expands into healthier populations seeking weight management rather than disease treatment.
Competition from injectable peptides will not disappear. In fact, peptide manufacturers continue to innovate aggressively, improving dosing frequency, tolerability, and efficacy. The market is not a zero-sum game, but strategic positioning will matter.
What the Next Decade Likely Holds
The most successful GLP-1 small molecule programs will be those that balance efficacy, safety, affordability, and real-world usability. Pure scientific brilliance will not be enough. Companies must understand patient psychology, healthcare system constraints, and regional market dynamics.
We are moving toward a future where metabolic diseases are managed earlier, more holistically, and with fewer barriers to access. GLP-1 small molecules fit naturally into that vision. They do not replace injectables entirely, but they fundamentally change the rules of engagement.
In my experience, transformative therapies are rarely loud at the beginning. They gain traction quietly, through consistent clinical results and patient acceptance. GLP-1 small molecules are at that inflection point now. The next ten years will determine whether they become a supporting act or the main narrative in metabolic care. Based on the current trajectory, they are positioning themselves to lead the story.
Access our exclusive, data-rich dashboard dedicated to the therapeutic area sector – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Important Links:
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout GLP-1 Small Molecule Market Report Now at:
Become a valued research partner with us - https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest




