
A Market Quietly Transforming Clinical Research
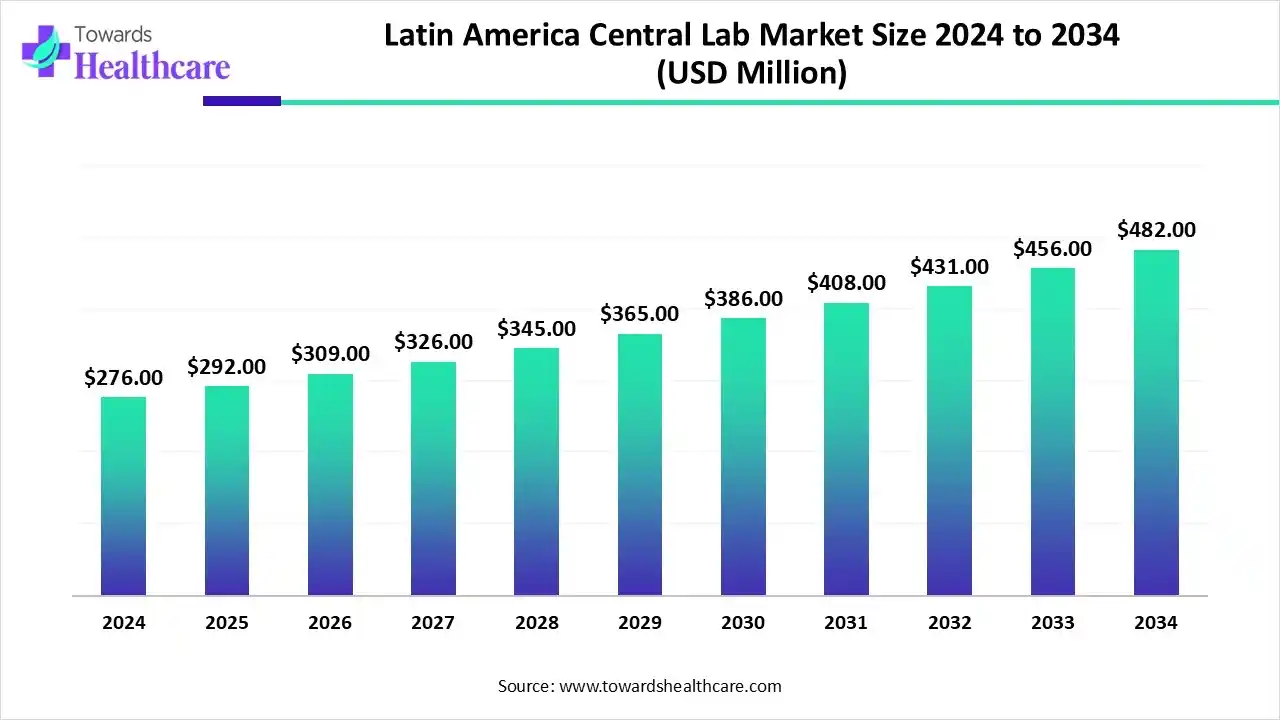
Latin America’s central laboratory market is no longer operating in the shadows of North America and Europe. What was once considered a cost-efficient alternative has steadily evolved into a strategic hub for clinical trials, advanced diagnostics, and laboratory standardization. With the market valued at US$ 276 million in 2024 and expected to reach nearly US$ 482 million by 2034, the region is demonstrating consistent, well-structured growth rather than short-term expansion driven by temporary demand cycles.
Download Free Sample Now and Get the Complete Report Easily at: https://www.towardshealthcare.com/download-sample/6327
This growth reflects a deeper shift in how pharmaceutical companies, biotechnology firms, and contract research organizations view Latin America. The region is no longer chosen solely for affordability. It is increasingly selected for its operational maturity, regulatory progress, patient diversity, and growing scientific capabilities. Central laboratories now play a pivotal role in enabling this transformation by delivering standardized testing, reliable data, and integrated clinical trial support across multiple countries.
Why Central Labs Matter More Than Ever in Latin America
Central laboratories form the backbone of modern clinical trials. They ensure consistency in testing protocols, manage complex datasets, and support regulatory compliance across borders. In Latin America, this role has become even more critical due to the region’s geographic diversity and varying national healthcare systems. Sponsors rely on central labs to harmonize results from Brazil, Mexico, Argentina, and emerging trial destinations, ensuring data integrity from first patient enrollment to final submission.
The rising number of multinational clinical trials in Latin America has amplified demand for centralized testing services. Pharmaceutical companies increasingly prefer a single laboratory partner that can manage logistics, testing, and reporting across the region. This approach reduces variability, shortens trial timelines, and improves regulatory confidence. As a result, central labs are no longer peripheral service providers; they are strategic collaborators in drug development.
Brazil’s Leadership Shapes the Regional Narrative
Brazil dominates the Latin American central lab market, and this leadership is not accidental. The country combines advanced healthcare infrastructure, a large patient population, skilled laboratory professionals, and supportive government policies. Recent regulatory reforms have significantly reduced clinical trial approval timelines, making Brazil one of the most efficient trial destinations globally.
The inauguration of advanced research initiatives such as the Orion Project in São Paulo signals Brazil’s long-term commitment to biomedical research. By integrating pathogen research with cutting-edge infrastructure like particle accelerators, the country is positioning itself at the forefront of laboratory science. Central labs operating in Brazil benefit directly from this ecosystem, gaining access to advanced technologies, trained talent, and international collaborations.
A Region Built for Clinical Trial Growth
Latin America offers a unique clinical trial environment. The region’s population includes a high proportion of treatment-naïve patients, which allows sponsors to observe drug efficacy with fewer confounding variables. Recruitment timelines are often faster than in saturated Western markets, reducing development costs and accelerating trial completion.
Central labs enable this advantage by managing large sample volumes and ensuring rapid turnaround times. As trial complexity increases, particularly in oncology and rare diseases, the demand for advanced testing capabilities continues to rise. Central laboratories in Latin America have responded by expanding molecular diagnostics, biomarker analysis, and bioanalytical services to meet global standards.
Biomarker Assays Drive Precision Medicine Forward
Biomarker assays emerged as the largest service segment in 2024, accounting for nearly two-fifths of market revenue. This dominance reflects the global shift toward precision medicine and targeted therapies. Biomarkers guide patient selection, monitor treatment response, and improve trial success rates, making them indispensable in modern drug development.
Latin American central labs have significantly expanded biomarker testing capabilities, particularly in oncology and cardiovascular research. These services support both clinical trials and routine diagnostics, bridging the gap between research and patient care. The increasing availability of biomarker assays in the region signals a maturing laboratory ecosystem capable of supporting complex therapeutic programs.
Molecular and Genetic Testing Signals the Next Growth Wave
While biomarker assays currently dominate, molecular and genetic testing represents the fastest-growing service area. Advances in polymerase chain reaction technologies and next-generation sequencing have transformed disease detection and personalized treatment planning. Latin America has rapidly adopted these technologies, driven by rising demand for early diagnosis and precision oncology.
Central labs play a critical role in scaling molecular testing across the region. By centralizing high-complexity analyses, laboratories reduce costs and ensure quality consistency. As genetic screening becomes more integrated into routine healthcare and clinical trials, molecular diagnostics will continue to reshape the region’s laboratory landscape.
Clinical Chemistry Remains the Operational Backbone
Despite the rise of advanced diagnostics, clinical chemistry analyzers remain the most widely used laboratory technology. These systems support high-throughput testing for routine biochemical parameters, making them essential for hospitals and healthcare systems. Their dominance reflects the continued need for reliable, cost-effective diagnostic testing across Latin America.
Automation has significantly improved efficiency and accuracy in clinical chemistry, enabling central labs to process large volumes of samples with minimal error. This capability supports both public health programs and private healthcare providers, reinforcing the foundational role of clinical chemistry in the regional market.
Hospitals Anchor Demand While CROs Accelerate Growth
Hospitals and healthcare systems represented the largest end-user segment in 2024, driven by high patient volumes and broad diagnostic needs. Central labs support hospitals by providing specialized testing, advanced analytics, and standardized reporting that internal laboratories may not be equipped to handle.
At the same time, contract research organizations are emerging as the fastest-growing customer segment. As pharmaceutical companies outsource larger portions of clinical development, CROs rely heavily on central labs for specialized testing and data management. This trend underscores the increasing integration of laboratory services into outsourced research models.
Blood Samples Dominate, but Non-Invasive Testing Gains Ground
Blood, plasma, and serum samples accounted for the majority of testing volume in 2024, reflecting their versatility and diagnostic value. These samples remain essential for biochemical, immunological, and molecular analyses across therapeutic areas.
However, non-invasive sample types such as saliva and swabs are gaining momentum. Their ease of collection improves patient compliance and expands testing accessibility, particularly in large-scale screening programs. Central labs are adapting workflows to accommodate these sample types, signaling a broader shift toward patient-centric diagnostics.
Infectious Diseases Sustain Core Demand
Infectious diseases continue to represent the largest therapeutic application for central labs in Latin America. The region’s exposure to diseases such as dengue, Zika, and tuberculosis creates sustained demand for diagnostic testing and surveillance. Central laboratories play a crucial role in outbreak management, providing timely data to public health authorities.
At the same time, rare diseases and genetic screening are emerging as high-growth areas. Increased awareness, improved diagnostic technologies, and expanding research initiatives are driving adoption. Central labs enable these programs by offering advanced genetic analyses that were previously unavailable or inaccessible in the region.
Outsourcing Redefines the Operating Model
Outsourced and third-party central labs accounted for the largest share of the market in 2024. This dominance reflects the growing preference for specialized providers capable of handling complex testing requirements at scale. Governments, hospitals, and research organizations increasingly rely on external laboratories to meet diagnostic demand without investing heavily in infrastructure.
Hybrid operating models are gaining traction as well. These models combine local laboratory networks with international partners, offering flexibility and access to advanced technologies. This approach allows central labs to maintain regional expertise while meeting global quality standards.
Mexico and Argentina Strengthen Regional Balance
While Brazil leads the market, Mexico and Argentina are strengthening their positions through targeted investments and policy initiatives. Mexico has focused on expanding molecular and genomic testing capabilities, integrating advanced diagnostics into public health programs and research initiatives.
Argentina has prioritized accreditation and infrastructure expansion, enhancing its capacity for infectious disease monitoring and molecular diagnostics. These efforts improve diagnostic coverage and reinforce the country’s role as a regional laboratory hub.
Artificial Intelligence Redefines Laboratory Efficiency
Artificial intelligence is reshaping central laboratory operations across Latin America. Automated data analysis, predictive analytics, and real-time monitoring improve accuracy and reduce turnaround times. AI-driven platforms enable laboratories to manage complex datasets efficiently, supporting faster decision-making in clinical trials.
The adoption of AI also reduces operational costs by minimizing manual errors and optimizing workflows. As digital infrastructure improves across the region, AI integration will become a defining feature of competitive central labs.
Sustainability Moves from Concept to Practice
Sustainability is no longer a peripheral concern for central laboratories. Energy-efficient equipment, waste reduction initiatives, and digital data management are becoming standard practices. Laboratories recognize that sustainable operations not only reduce environmental impact but also improve long-term cost efficiency.
The adoption of eco-friendly reagents and paperless workflows reflects a broader commitment to responsible growth. These initiatives align with global environmental standards and enhance the reputation of Latin American labs among international partners.
Investment Confidence Signals Long-Term Stability
The presence of multinational pharmaceutical companies, leading CROs, and private equity investors underscores confidence in the Latin American central lab market. Investments focus on infrastructure expansion, technology upgrades, and workforce development. This capital inflow supports sustainable growth rather than speculative expansion.
A growing startup ecosystem further strengthens the market. Innovative diagnostic companies and digital health platforms are emerging across the region, often in collaboration with established labs and research institutions. These startups introduce new technologies and business models, enhancing market dynamism.
A Market Aligned with Global Growth Trends
Latin America’s central lab market mirrors global growth patterns while offering distinct regional advantages. The global market is projected to grow steadily over the next decade, driven by rising R&D investments and increasing clinical trial complexity. Latin America’s alignment with these trends positions it as an integral part of the global laboratory ecosystem.
Central labs in the region increasingly participate in multinational trials, contributing data that meets international regulatory standards. This integration reinforces Latin America’s credibility as a reliable research destination.
Looking Ahead with Measured Confidence
The Latin America central lab market is not defined by explosive growth but by consistent, strategic expansion. Its trajectory reflects careful investment, regulatory progress, and technological adoption. Central laboratories have evolved from service providers into strategic enablers of clinical research and diagnostics.
As clinical trials become more complex and precision medicine gains momentum, the role of central labs will continue to expand. Latin America’s combination of cost efficiency, patient diversity, and improving infrastructure positions it as a critical contributor to global healthcare innovation.
The question is no longer whether Latin America belongs in the global central lab landscape. The real question is how far its influence will extend as science, technology, and collaboration continue to converge across borders.
Important Links
Access our exclusive, data-rich dashboard dedicated to the healthcare market – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout Latin America Central Lab Market Report Now at: https://www.towardshealthcare.com/checkout/6327
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest




