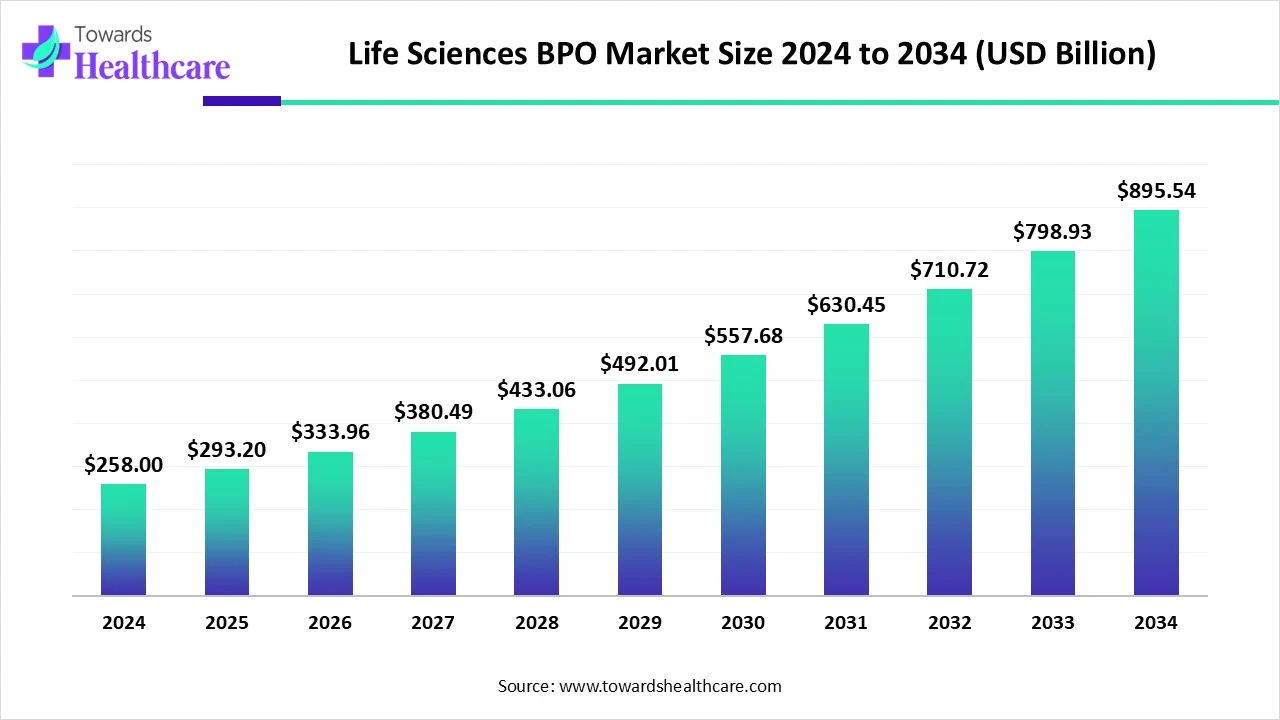
The global life sciences BPO market, valued at USD 258 billion in 2024 and projected to reach USD 895.54 billion by 2034 at a CAGR of 13.64%, is rapidly growing due to increasing R&D complexity, clinical trials, and AI-driven innovations.
Download the free sample and get the complete insights and forecasts report on this market @ https://www.towardshealthcare.com/download-sample/5869
2. Market Size
●2024: USD 258 billion – reflecting early adoption and reliance on BPO services.
●2025: USD 293.2 billion – initial growth supported by AI integration and new collaborations.
●2034 Projection: USD 895.54 billion – showing long-term exponential growth.
●CAGR: 13.64% (2025–2034) – among the highest across BPO industries due to increasing global demand.
●North America: Dominated 43% share in 2024 due to presence of mature pharma and biotech companies.
●Asia Pacific: Fastest-growing region due to increasing clinical trials, cost advantages, and skilled workforce.
Service Type Insights:
●CROs accounted for 41% share in 2024.
●CMOs/CDMOs expected to grow fastest during the forecast period.
Phase Type Insights:
●Preclinical & clinical services: 53% share in 2024.
●Commercial phase services: Fastest-growing due to post-marketing and pharmacovigilance needs.
End Users:
●Pharmaceutical companies: 46% share in 2024.
●Biotechnology companies: Fastest-growing segment due to limited internal capacity.
Regulatory Demand: High dependence on BPO for compliance, drug development, and operational efficiency.
3. Market Trends
AI Integration:
●AI platforms improve patient selection, trial outcome prediction, and data interpretation.
●Generative AI adoption enhances medical writing and regulatory compliance.
Global Collaborations:
●Hexaware & Abluva: AI safety and compliance in life sciences.
●Auriga Research & SFRI: AI-driven clinical research solutions for Africa, India, and the U.S.
Medical Writing Innovations:
●NEXT Medical Writing Automation (Indegene) merges GenAI with regulatory expertise for accurate and fast documentation.
Data Analytics Adoption:
●Axtria InsightsMAx enhances ROI and experimentation using AI-first data analytics platforms.
Regulatory & Compliance Focus:
●Companies outsource to ensure adherence to FDA, EMA, GDPR, and other global standards.
Operational Efficiency:
●Outsourcing non-core functions like pharmacovigilance, supply chain, IT management reduces costs and time-to-market.
Investment & Financial Strategies:
●Baker Tilly’s stewardshipNOW helps manage external funding efficiently and ethically.
Digital-First Platforms:
●Cloud-based tools (EDC, CTMS, eTMF, LIMS) streamline global trials and ensure data compliance.
Rising Clinical Trials:
●Increased trial complexity drives demand for specialized BPO services, reducing delays and risks.
Next-Gen Therapy Support:
●CMOs/CDMOs expand services to support gene/cell therapy development, reducing internal capacity requirements.
4. AI Impact / Role
Patient Selection Optimization: AI models identify ideal participants, reducing trial failure risk.
Trial Outcome Prediction: Algorithms forecast likelihood of success/failure in clinical phases.
Data Interpretation: AI efficiently processes complex datasets, identifying trends and anomalies.
Medical Writing Automation: GenAI generates regulatory-compliant documents faster.
Pharmacovigilance: AI aids in signal detection, adverse event reporting, and risk assessment.
Regulatory Compliance: Ensures adherence to global regulations and submission standards.
Operational Automation: Streamlines supply chain, logistics, and IT infrastructure management.
Global Trial Harmonization: AI aligns multi-country trials, ensuring consistency across regions.
Market Analytics & Commercial Insights: Provides pricing, reimbursement, and KOL engagement analysis.
Post-Marketing Surveillance: AI monitors real-world drug safety and product performance.
Regional Insights
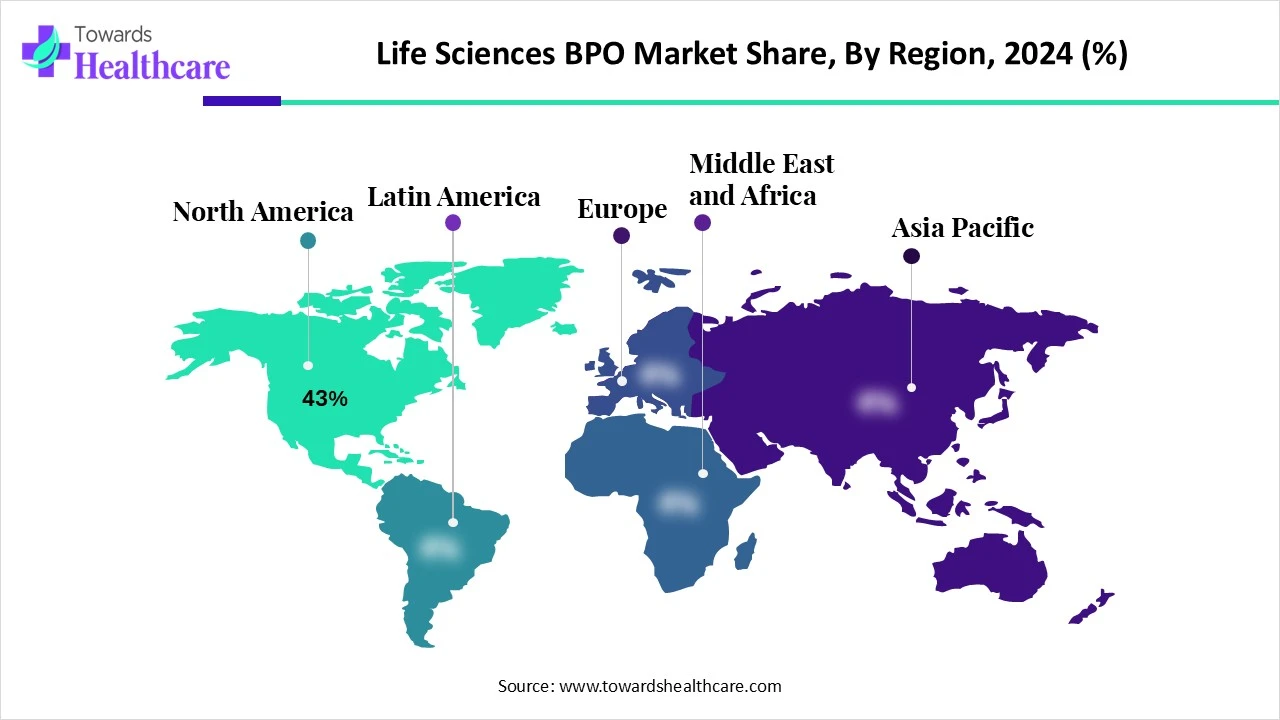
North America (43% Market Share, 2024)
●North America dominates the Life Sciences BPO market due to a mature pharma and biotech ecosystem, strong regulatory framework, and high adoption of advanced technologies.
United States
AI-Driven BPO Adoption:
●Pharma and biotech firms leverage AI-powered platforms for clinical trial management, data analytics, and regulatory submissions.
●Enhances efficiency and reduces manual errors in medical writing, pharmacovigilance, and data interpretation.
Acceleration of Diagnostics & Clinical Trials:
●Outsourcing preclinical and clinical trial management helps manage complex trials and shorten timelines.
●CROs and CDMOs support patient recruitment, protocol adherence, and site monitoring.
Regulatory Compliance Focus:
●BPO services help companies navigate FDA guidelines and maintain compliance with 21 CFR Part 11 and HIPAA.
●Reduces the risk of regulatory penalties and accelerates approvals.
Cost and Operational Efficiency:
●Outsourcing non-core functions such as supply chain management, pharmacovigilance, and IT infrastructure reduces operational costs.
Canada
Advanced Technology Integration:
●Adoption of cloud-based systems (EDC, CTMS, eTMF, LIMS) for trial management and data security.
R&D Optimization:
●BPO services enable academic institutions and biotech firms to scale research operations efficiently.
Outsourcing Non-Core Functions:
●Companies outsource regulatory filings, pharmacovigilance, medical writing, and logistics.
●Reduces administrative burden and operational risk.
Cost Management:
●Outsourcing enables smaller Canadian biotech firms to access global expertise without large capital investments.
Asia Pacific (Fastest-Growing Region)
●Asia Pacific is expected to exhibit the highest growth during 2025–2034 due to rising clinical trials, lower labor costs, and supportive government policies.
China
Rising Disease Burden:
●Increasing prevalence of chronic and lifestyle diseases drives demand for drug development and clinical trials.
Expansion of Diagnostics & Treatment:
●Life sciences companies are conducting more clinical trials to validate therapies locally.
BPO Demand Growth:
●CROs and CDMOs are heavily relied upon for clinical trial management, regulatory submissions, and manufacturing support.
Advanced Facilities:
●Development of high-quality laboratories and manufacturing units enhances adoption of outsourced services.
India
Skilled Workforce:
●India offers a large pool of trained professionals in clinical research, regulatory affairs, and pharmacovigilance.
Cost Advantage:
●Lower labor costs compared to North America and Europe encourage multinational pharma and biotech firms to outsource.
Government Policies & Investments:
●Regulatory frameworks, tax incentives, and infrastructure support facilitate clinical trial outsourcing.
Growing Clinical Trial Volume:
●Increase in global trials conducted in India strengthens the reliance on CROs and CMOs/CDMOs.
Europe
●Europe shows steady growth driven by stringent regulations, advanced research institutions, and personalized medicine initiatives.
Germany
Advanced Research Institutions:
●Universities and biotech hubs conduct high-level R&D, relying on BPOs to manage clinical trial execution and documentation.
Operational Cost Reduction:
●Outsourcing manufacturing, logistics, and trial management helps firms optimize budgets.
Regulatory Compliance:
●BPO services assist with EMA submissions, risk management, and safety monitoring for clinical trials.
United Kingdom
Personalized Medicine Initiatives:
●Growth in individualized therapies drives adoption of digital platforms and outsourcing solutions.
Digital Platform Adoption:
●Cloud-based systems and AI-driven tools streamline trial monitoring, data management, and pharmacovigilance.
BPO Collaboration:
●Pharmaceutical and biotech companies partner with CROs and CDMOs to improve time-to-market, scale operations, and manage regulatory challenges.
Key Insights Across Regions
North America: Mature market, high-tech adoption, strong regulatory oversight.
Asia Pacific: Rapid growth, cost-effective, skilled workforce, supportive policies.
Europe: Regulatory-driven growth, advanced R&D, personalized medicine adoption.
Regional Drivers of BPO Adoption:
Trial complexity, cost optimization, global clinical expansion, AI integration, regulatory compliance, and scalable manufacturing.
Market Dynamics
Drivers
Increasing Clinical Trials
Rising Complexity:
●Modern drug development involves multiple phases, multi-country trials, and complex protocols.
●Life sciences companies rely on BPOs to manage these complexities efficiently.
Operational Efficiency:
●Outsourcing clinical trials to CROs reduces delays and administrative burdens.
●Ensures better trial monitoring, patient recruitment, and data integrity.
Regulatory Compliance:
●BPOs provide specialized expertise to navigate regional and global regulatory requirements (FDA, EMA, CDSCO, etc.).
Cost Reduction:
●Outsourcing mitigates the high internal costs of conducting clinical trials.
●Enables companies to scale trials without heavy investment in infrastructure.
Rising FDA Approvals (2024)
Increased Drug Development:
●A rise in approved drugs indicates more ongoing trials, requiring BPO support.
Expanded Clinical Programs:
●Post-approval studies, Phase IV trials, and post-marketing surveillance increase BPO demand.
Enhanced BPO Adoption:
●Companies outsource regulatory filing, pharmacovigilance, and reporting tasks to streamline approvals.
Innovation-Driven Growth:
●Approval of novel therapies, including biologics and cell/gene therapies, pushes demand for specialized BPO services (CMOs/CDMOs).
Restraints
Privacy and Security Concerns
Data Sensitivity:
●Clinical trial data includes patient records, proprietary R&D information, and confidential drug formulas.
Cybersecurity Risks:
●Data breaches or hacking can compromise sensitive information.
●Could lead to regulatory penalties and reputational damage.
Regulatory Compliance Pressure:
●BPOs must adhere to HIPAA, GDPR, 21 CFR Part 11, and other regional regulations.
●Failure to comply can slow adoption or reduce trust in outsourcing.
Financial & Ethical Implications:
●Breaches may result in financial loss, litigation, and ethical scrutiny.
●Companies may hesitate to fully outsource sensitive operations.
Opportunities
AI-Enabled Medical Writing Platforms
Faster Documentation:
●Platforms like Indegene’s NEXT Medical Writing Automation accelerate the creation of regulatory-compliant documents.
Accuracy & Compliance:
●AI reduces errors and ensures alignment with global regulatory guidelines (FDA, EMA, CDSCO).
Cost-Effectiveness:
●Reduces need for in-house medical writers, lowering operational costs.
Scalability:
●Enables life sciences companies to handle multiple submissions, clinical reports, and post-marketing documentation efficiently.
CMOs/CDMOs for Advanced Therapies
Support for Next-Gen Therapies:
●Gene and cell therapy manufacturing requires specialized expertise, scalable facilities, and strict regulatory adherence.
Flexible Manufacturing:
●CMOs/CDMOs allow companies to scale production up or down based on demand without investing in large infrastructure.
Time-to-Market Advantage:
●Outsourcing manufacturing accelerates production cycles and clinical trial readiness.
Global Reach:
●International CMOs/CDMOs provide access to global supply chains, distribution, and regulatory knowledge.
Top 10 Companies
IQVIA:
Products: AI Agents for Life Sciences, Commercial Solutions
Strength: Clinical trial analytics, insights-driven workflow, global expertise
Parexel International:
Products: CRO services, regulatory consulting
Strength: Trial management, regulatory compliance
Syneos Health:
Products: Biopharmaceutical solutions, integrated commercialization
Strength: Clinical development + commercialization expertise
Labcorp Drug Development (Covance):
Products: Drug development, global trial management
Strength: Regulatory support, extensive global network
PPD (Thermo Fisher):
Products: Clinical trials, pharmacovigilance
Strength: Advanced data analytics and regulatory compliance
Charles River Laboratories:
Products: Preclinical testing, safety studies
Strength: Lab models, toxicology expertise
ICON plc:
Products: CRO services, clinical operations
Strength: Global reach, regulatory knowledge
Wuxi AppTec:
Products: Drug development services, manufacturing
Strength: End-to-end development, clinical trial support
Pharmaron:
Products: CRO/CDMO services
Strength: Integrated solutions, R&D efficiency
Medpace Holdings / Catalent / Lonza / Samsung Biologics / Boehringer Ingelheim BioXcellence
Products: Clinical, manufacturing, biologics
Strength: Advanced platforms, global production and research capabilities
Latest Announcements
IQVIA (Jan 2025): AI Agents for Life Sciences → insights, precise workflows, collaboration with NVIDIA.
Baker Tilly (Jun 2025): stewardshipNOW → reduces administrative burden, manages external funding ethically.
Hexaware & Abluva (Jul 2025): AI compliance and safety integration for life sciences.
Auriga & SFRI (Jul 2025): AI-enhanced clinical research across Africa, India, U.S.
Recent Developments
LogicFlo AI (Jun 2025): $2.7M seed funding for global medtech, pharmaceutical, biotech expansion.
Axtria InsightsMAx (May 2025): AI-first data analytics platform for enterprise-grade ROI enhancement.
Indegene NEXT Medical Writing Automation (Jul 2025): Generative AI + medical writing expertise for compliance.
Segments Covered
By Service Type
Contract Research Services (CROs)
●CROs provide outsourced research and clinical trial support for pharmaceutical, biotech, and medical device companies.
Clinical Trial Management:
●Full management of Phase I–IV trials.
●Includes planning, site selection, monitoring, and execution.
●Reduces operational complexity and ensures adherence to regulatory standards.
Data Management:
●Collection, processing, and validation of clinical trial data.
●Ensures accuracy, integrity, and compliance with regulatory frameworks.
Medical Writing:
●Preparation of study protocols, clinical study reports, regulatory documents, and manuscripts.
●AI-powered solutions (e.g., NEXT Medical Writing Automation by Indegene) accelerate content creation while maintaining regulatory compliance.
Site Monitoring:
●Regular supervision of clinical trial sites to ensure protocol adherence and patient safety.
●Reduces risks of deviations and regulatory non-compliance.
Protocol Development:
●Designing clinical trial protocols with statistical and scientific rigor.
●Helps predict trial feasibility, optimize sample size, and reduce costs.
Contract Manufacturing Services (CMOs/CDMOs)
●These services focus on the production and supply of drugs, biologics, and therapies on behalf of clients.
API Production:
●Active Pharmaceutical Ingredient manufacturing under strict GMP compliance.
●Ensures consistent quality for clinical and commercial use.
Fill-Finish Services:
●Sterile and non-sterile drug product packaging into vials, syringes, or capsules.
●Critical for ensuring safety, accuracy, and regulatory compliance.
Packaging & Labeling:
●Custom packaging solutions, labeling, and serialization to meet regulatory standards.
●Supports traceability and market distribution requirements.
Pharmacovigilance & Safety Services
●Focus on monitoring, analyzing, and reporting drug safety post-approval.
Adverse Event Reporting:
●Collection, documentation, and analysis of adverse events reported by patients or healthcare providers.
Signal Detection:
●Identifying trends or safety signals from large datasets to prevent potential risks.
Risk Management:
●Developing strategies to minimize safety risks during clinical trials or post-marketing.
Regulatory Affairs Outsourcing
●Provides specialized support for global regulatory compliance.
Submission Filing (IND, NDA, MAA):
●Preparing and filing applications for clinical trials and product approvals.
Regulatory Strategy & Intelligence:
●Guidance on regulatory pathways, risk mitigation, and country-specific compliance.
●Ensures faster approvals and reduces regulatory hurdles.
Sales, Marketing & Market Access Services
●Supports commercialization of drugs and therapies.
KOL Engagement:
●Collaboration with key opinion leaders to drive clinical insights and product adoption.
Commercial Analytics:
●Market research, forecasting, and performance tracking.
Pricing & Reimbursement Support:
●Guidance on reimbursement strategies, insurance coverage, and cost-effectiveness evaluation.
Supply Chain & Logistics
●Ensures smooth delivery of clinical materials and commercial products.
IT & Infrastructure Management:
●Maintenance of cloud platforms, EDC, CTMS, eTMF, LIMS.
Data Security:
●Compliance with 21 CFR Part 11, GDPR, HIPAA, and other regulations.
Logistics & Distribution:
●Planning, storage, cold-chain management, and timely delivery of drugs to sites or commercial markets.
By Phase
Preclinical & Clinical Phase Services
●Support early-stage drug development and clinical trials.
Preclinical Toxicology:
●Safety testing in animal models before human trials.
IND Preparation:
●Preparation of Investigational New Drug applications for regulatory submission.
Phase I–IV Trial Execution:
●Phase I: Safety and dosage.
●Phase II: Efficacy and side effects.
●Phase III: Large-scale efficacy, comparison with standard treatments.
●Phase IV: Post-marketing surveillance, long-term effects.
Commercial Phase Services
●Support product launch and post-marketing activities.
Marketing & Distribution:
●Promotion, market access, and logistics of commercial products.
Post-Marketing Surveillance:
●Ongoing monitoring of safety, effectiveness, and adverse events after product launch.
By End User
Pharmaceutical Companies:
●Largest user segment (46% in 2024).
●Reliance on BPO for clinical trials, manufacturing, regulatory compliance, and commercialization.
Biotechnology Companies:
●Fastest-growing segment.
●Limited internal capacity, growing demand for advanced therapies like gene and cell therapy.
Medical Device Manufacturers:
●Outsource clinical studies, regulatory submissions, and safety monitoring.
Academic Institutions:
●Collaboration with CROs for research projects and clinical trials.
Government & Public Health Agencies:
●Outsource data management, trial monitoring, and pharmacovigilance.
By Region
North America
Market Share: 43% in 2024.
Drivers: Mature pharma and biotech ecosystem, high adoption of AI, global CRO/CMO presence.
Subregions:
U.S.: AI-driven clinical trials, regulatory compliance, diagnostics innovations.
Canada: Advanced technologies and BPO adoption for cost efficiency.
Asia Pacific
Fastest-Growing Region due to cost advantages, skilled workforce, and growing trials.
Subregions:
China: Rising disease burden → higher clinical trial demand.
India: Low labor cost + government support → outsourcing hub.
Japan, South Korea, Thailand: Growing R&D, clinical trial adoption.
Europe
Growth Driver: Stringent regulatory compliance.
Subregions:
Germany: Advanced research → higher BPO usage for cost reduction.
UK: Personalized medicine → digital platforms and outsourcing.
France, Italy, Spain, Sweden, Denmark, Norway: Compliance-driven demand for CRO/CMO support.
Latin America
Countries: Brazil, Mexico, Argentina.
Drivers: Emerging clinical trial markets, cost-effective outsourcing.
Middle East & Africa (MEA)
Countries: UAE, Saudi Arabia, South Africa, Kuwait.
Drivers: Growing healthcare infrastructure, outsourcing for trials, regulatory expertise.
Top 5 FAQs
1 What is the Life Sciences BPO market?
Outsourced services for pharma, biotech, medical devices to improve clinical trial management, regulatory compliance, and operational efficiency.
2 What is the current size of the Life Sciences BPO market?
USD 258 billion in 2024; projected to reach USD 895.54 billion by 2034 at a CAGR of 13.64%.
3 Which region dominates the market?
North America held 43% share in 2024; Asia Pacific fastest-growing due to cost advantages and clinical trial growth.
4 Which services dominate the market?
CROs dominate (41% share), preclinical & clinical services largest (53% share), pharmaceutical companies largest end users (46% share).
5 How is AI impacting the market?
AI enhances trial optimization, medical writing, regulatory compliance, pharmacovigilance, predictive analytics, and post-marketing surveillance.
Access our exclusive, data-rich dashboard dedicated to the life science industry – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Immediate Delivery Available | Buy This Premium Research @ https://www.towardshealthcare.com/price/5869
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Web: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest