Overview of Newborn Eye Imaging Systems
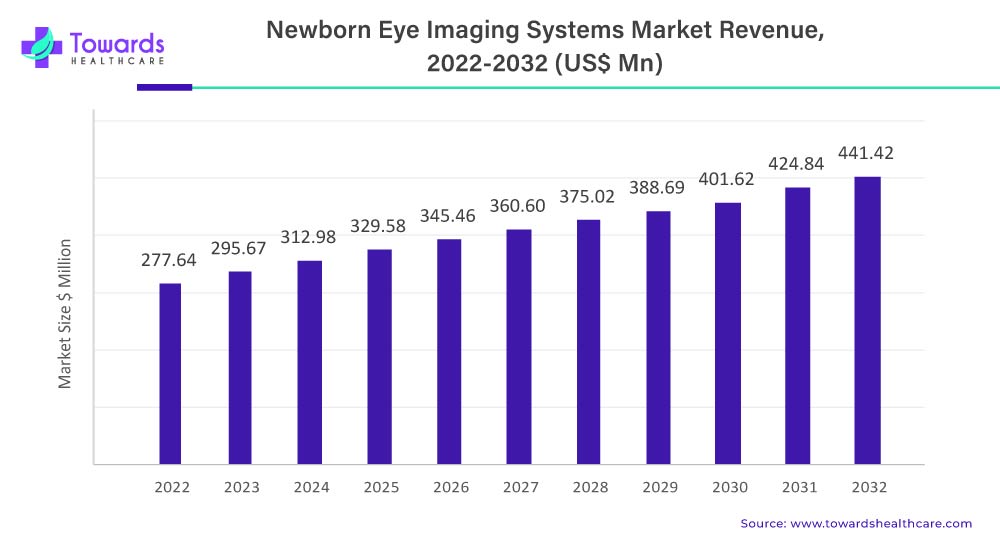
The newborn eye imaging system market is projected to grow from USD 277.64 million in 2022 to an estimated USD 441.42 million by 2032, driven by a compound annual growth rate (CAGR) of 4.6% from 2023 to 2032. This growth is primarily fueled by the rising prevalence of Retinopathy of Prematurity (ROP) and increasing healthcare spending.
Download statistics of this report @ https://www.towardshealthcare.com/download-statistics/5033
Market Drivers and Trends
Ongoing research and development in ROP diagnosis present significant growth opportunities for the market. Retinal imaging technology, which enables high-resolution, non-invasive images of the retina, plays a key role in identifying a range of eye disorders. The retina, being crucial for vision, can reveal signs of both eye and systemic diseases, making early detection vital.
Early eye exams in newborns are crucial, as untreated vision issues can lead to long-term complications, including blindness. By identifying problems early, conditions like ROP and other eye disorders—such as strabismus, refractive errors, and color vision issues—can be addressed effectively. Retinal imaging is essential in diagnosing, monitoring, and managing these conditions.
The newborn eye imaging system market specifically caters to detecting and treating eye disorders in newborns, particularly ROP, which is common in premature infants and those with low birth weight. As the incidence of premature births continues to rise globally, the demand for these specialized imaging systems is expected to grow.
Additionally, the increasing implementation of early screening programs for ROP is boosting market demand. Many countries now offer screening to detect ROP early and prevent blindness, further driving the adoption of advanced eye imaging systems. With continuous technological advancements, new imaging devices are being developed to provide clearer, more accurate images, further enhancing their use in neonatal care.
Overall, the market for newborn eye imaging systems is on a strong growth trajectory, driven by the need for early detection, the rising number of premature births, and innovations in imaging technology.
Overcoming the Challenge of High Costs
Newborn eye imaging systems are sophisticated medical devices that require advanced technology and expertise to design, manufacture, and maintain. As a result, they are often expensive, which can be a major barrier to their widespread adoption, particularly in low-resource settings. In developing countries, where healthcare budgets are limited and there are many competing priorities, the high cost of these systems can make it difficult for healthcare facilities to justify purchasing them. In some cases, the price may be too high, leading to a lack of access to the technology and potentially missing early detection of retinopathy of prematurity (ROP) in newborns.
Even when these systems are acquired, the ongoing costs of maintenance and repair can pose a significant challenge. Without trained personnel or the right equipment, healthcare facilities may face extended downtime and higher repair costs, further stretching their limited budgets.
To address these challenges, several solutions could be explored. One approach is to foster collaboration between governments, healthcare providers, and manufacturers to help reduce costs. Governments could offer subsidies or tax incentives to manufacturers, lowering the production cost, while healthcare providers could team up to buy systems in bulk, benefiting from economies of scale.
Another potential solution is to introduce flexible financing options, like leasing or pay-per-use models. This would allow healthcare facilities to pay for the systems over time, easing their budget constraints and allowing them to prioritize other healthcare needs.
Additionally, ongoing investment in research and development is essential to making these systems more affordable. This could lead to the creation of lower-cost devices that are easier to use and maintain. Incorporating artificial intelligence (AI) and other advanced technologies could also enhance the systems’ accuracy and efficiency, further reducing costs and making them more accessible to healthcare providers.
Addressing the high cost of newborn eye imaging systems is crucial to ensuring that this life-saving technology reaches more healthcare facilities, especially in regions where the burden of ROP is the highest.
Visionary Advancements: The Future of Newborn Eye Imaging Systems
The development of more affordable and efficient imaging systems could significantly increase their use in low-resource settings. As technology continues to evolve, there are opportunities to create more compact and portable devices that are cheaper to manufacture and easier to transport, making it easier for healthcare facilities in remote areas to access the technology. This would greatly enhance the diagnosis and treatment of retinopathy of prematurity and other newborn eye conditions.
Another exciting opportunity is the integration of artificial intelligence (AI) into these systems. AI-powered imaging can analyze images in real-time and provide immediate results, reducing the need for specialized personnel to interpret the images. This would make the technology more affordable and efficient, while also improving diagnostic accuracy.
Telemedicine also presents an important opportunity. By enabling remote diagnosis and treatment, telemedicine can be especially useful in low-resource settings where specialized healthcare professionals are scarce. Healthcare facilities could send eye images to experts in other locations for interpretation, eliminating the need for costly and time-consuming travel.
In conclusion, continued technological advancements hold great promise for making newborn eye imaging systems more accessible and affordable. With these innovations, it will be easier to detect and treat conditions like retinopathy of prematurity, especially in regions that need it the most.
Discover our detailed Table of Contents (TOC) for the Industry, providing a thorough examination of market segments, material, emerging technologies and key trends. Our TOC offers a structured analysis of market dynamics, emerging innovations, and regional dynamics to guide your strategic decisions in this rapidly evolving healthcare field – https://www.towardshealthcare.com/table-of-content/newborn-imaging-systems-market
To own our research study instantly, Click here @ https://www.towardshealthcare.com/price/5033
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
About Us
Towards Healthcare is a leading global provider of technological solutions, clinical research services, and advanced analytics to the healthcare sector, committed to forming creative connections that result in actionable insights and creative innovations. We are a global strategy consulting firm that assists business leaders in gaining a competitive edge and accelerating growth. We are a provider of technological solutions, clinical research services, and advanced analytics to the healthcare sector, committed to forming creative connections that result in actionable insights and creative innovations.
Visit Dental Specifics: https://www.towardsdental.com
Explore the comprehensive statistics and insights on healthcare industry data and its associated segmentation: Get a Subscription
For Latest Update Follow Us: https://www.linkedin.com/company/towards-healthcare