Pediatric Drugs Market Growth, Shares, Top Players and Latest Reports 2025- 2034

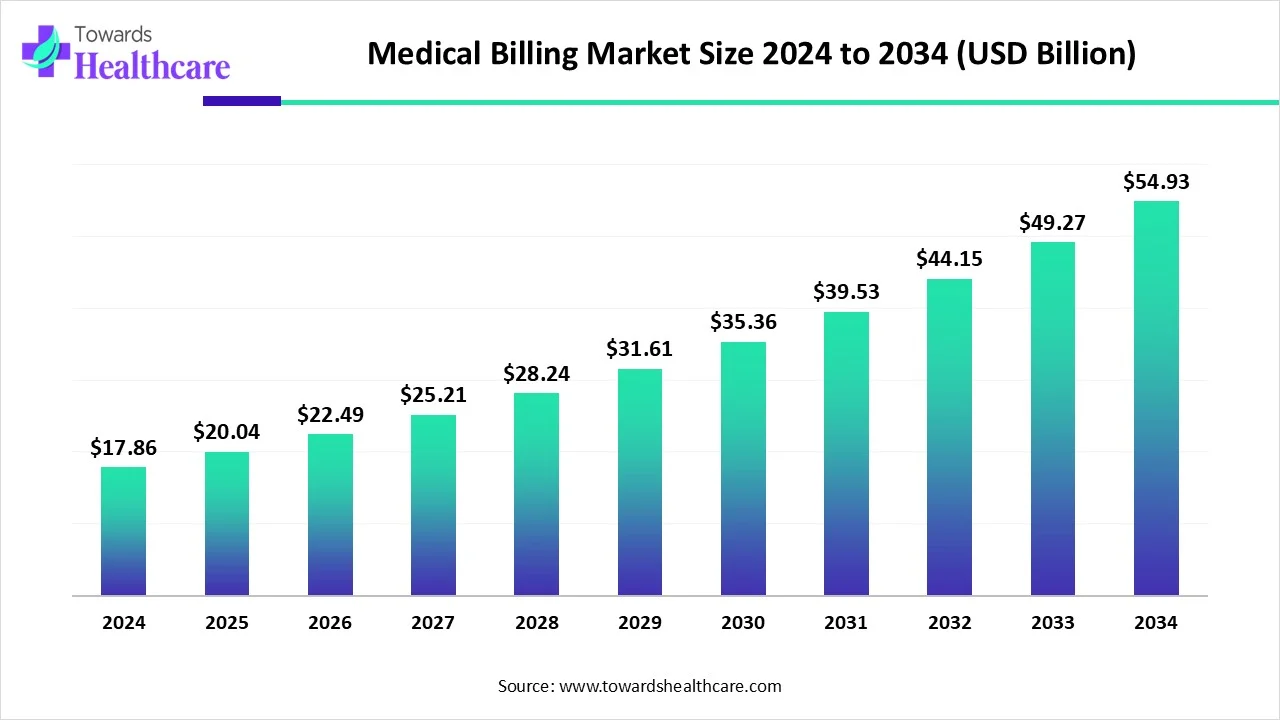
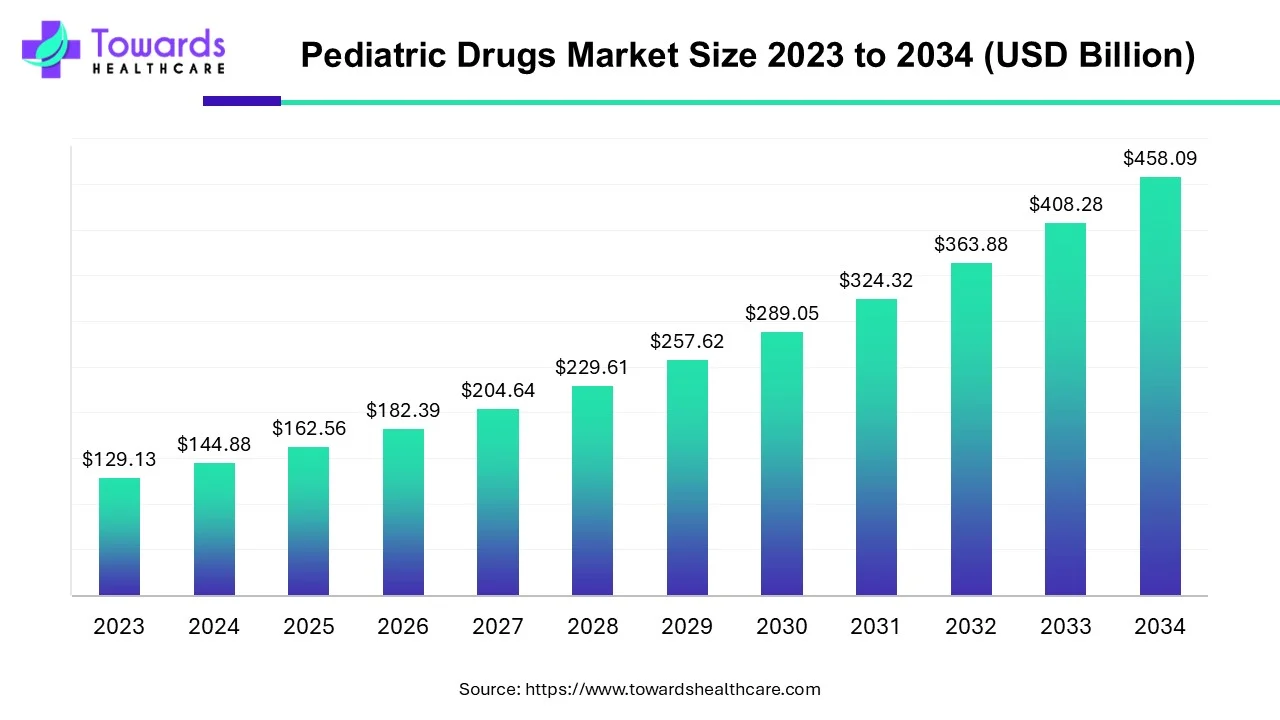
The global pediatric drugs market is projected to grow significantly from USD 162.56 billion in 2025 to USD 458.09 billion by 2034, with a CAGR of 12.2%.
Download Free Sample of Pediatric Drugs Market Now and Get the Complete Report Easily at: https://www.towardshealthcare.com/download-sample/5074
This growth is primarily driven by the increasing prevalence of pediatric disorders and the expanding pediatric population. Major market drivers include the rise in childhood diseases, government incentives for pediatric drug development, advancements in pediatric research, and the growing importance of personalized treatments tailored to children’s unique physiological needs. Additionally, WHO’s focus on developing essential antibiotics for children and the rise in clinical trials and drug approvals in pediatric oncology further contribute to the market’s expansion.
Table of Contents
ToggleMarket Size:
Overall Market Size:
2025: USD 162.56 billion
2034: USD 458.09 billion
CAGR: 12.2%
Segment-wise Market Size (2024):
Respiratory Drugs: Dominant segment due to increased prevalence of chronic respiratory conditions like asthma and COPD.
Gastrointestinal Drugs: Expected significant growth due to rising gastrointestinal disorders in children.
Oral Drugs: Largest share in route of administration.
Parenteral Drugs: Fastest-growing due to the need for injectable medications in younger children.
Hospital Pharmacies: Leading distribution channel due to infrastructure and specialized pediatric care.
Online Pharmacies: Fastest-growing distribution channel, driven by accessibility.
Pediatric Drugs Market Trends
Increasing Prevalence of Pediatric Disorders:
Respiratory and gastrointestinal disorders are on the rise, contributing to the growing demand for pediatric drugs.
Conditions such as asthma, obesity, diabetes, and neurological disorders are prevalent, driving the need for specialized medications.
Government and Regulatory Support:
WHO’s 2023 list of pediatric priority antibiotics fuels research.
FDA and EMA initiatives like orphan drug designations for pediatric conditions provide incentives for companies to focus on pediatric drug development.
Innovation in Pediatric Drug Formulations:
The development of child-friendly formulations, including flavored syrups and chewable tablets, makes drugs more palatable for children.
The use of AI and biotechnology is accelerating the development of more effective pediatric treatments.
Focus on Rare Pediatric Diseases:
Specialized drugs for diseases like Duchenne muscular dystrophy, childhood cancers, and rare genetic disorders are gaining attention.
FDA’s approval of treatments like Nivolumab for pediatric melanoma and Debrafenib for brain tumors in children showcases the expansion of the pediatric oncology segment.
Increased Investment in Pediatric Clinical Trials:
Growing number of pediatric clinical trials focused on rare diseases and genetic disorders.
Companies like Cumberland Pharmaceuticals are conducting trials to address unmet needs in rare pediatric diseases.
Role of AI in Pediatric Drugs Market
Accelerating Drug Discovery:
AI can analyze large datasets to identify potential drug candidates, speeding up the discovery of treatments for pediatric diseases.
Personalized Medicine:
AI helps tailor pediatric drugs to individual patients, considering factors such as age, weight, genetic makeup, and health conditions.
Predictive Modeling:
AI can predict how drugs will behave in children’s bodies, helping to determine the most effective dosage and treatment protocols.
Improving Drug Safety:
AI can analyze side effects data to identify patterns in pediatric populations, enhancing the safety profiles of pediatric drugs.
Optimizing Clinical Trials:
AI streamlines clinical trial processes by predicting patient responses, optimizing trial designs, and identifying suitable candidates.
Enhanced Medication Adherence:
AI-driven tools like reminder apps can help children and caregivers stay on top of medication schedules, improving adherence.
Real-Time Monitoring:
AI can facilitate real-time monitoring of pediatric patients’ conditions, adjusting treatment regimens as needed.
Supply Chain Optimization:
AI optimizes the distribution of pediatric medications, ensuring timely deliveries, especially for rare diseases or remote regions.
Data-Driven Decision Making:
AI aids healthcare providers and researchers in making informed decisions by analyzing large datasets of clinical trial outcomes.
Automating Administrative Tasks:
AI streamlines the administrative side of pediatric drug development, from regulatory compliance to drug delivery management.
Regional Insights
North America:
Dominates the global market due to robust healthcare infrastructure, high research investment, and a strong pediatric patient population.
In the U.S., initiatives like the FDA’s pediatric exclusivity and the prevalence of childhood diseases ensure continued market growth.
Asia-Pacific:
Fastest-growing region, driven by large pediatric populations in countries like India and China.
Government initiatives like the National Health Mission and private sector investments are fostering the market’s growth.
Europe:
EU regulations like the Paediatric Regulation and the Pediatric Investigation Plans (PIPs) are boosting drug development and clinical trials for pediatric use.
Countries like the UK and France show strong market growth, supported by government investments and healthcare reforms.
Latin America:
Growing pediatric drug market due to rising healthcare awareness and better access to treatments in countries like Brazil.
Middle East & Africa:
Emerging markets with increased focus on improving pediatric healthcare, though challenges remain in terms of healthcare infrastructure.
Market Dynamics of the Pediatric Drugs Market
The pediatric drugs market is shaped by various factors that drive growth, present challenges, and create opportunities. Below is a detailed breakdown of the drivers, restraints, opportunities, and challenges that influence this market.
Drivers:
Rising Pediatric Health Issues:
Prevalence of Chronic Conditions:
Asthma, obesity, diabetes, neurological disorders, and childhood cancers are among the most common health issues affecting children. These conditions require specialized treatments, thus driving the demand for pediatric drugs.
For example, the increasing rate of asthma in children, coupled with environmental factors like pollution and allergens, contributes significantly to the respiratory drugs segment.
Obesity in children, often leading to secondary diseases like type 2 diabetes and hypertension, is another major driver, necessitating the need for pediatric medications targeted at these conditions.
Government Regulations & Support:
FDA Orphan Drug Designation:
Governments are incentivizing pharmaceutical companies to focus on pediatric drug development. In the U.S., the FDA Orphan Drug Designation provides incentives like extended market exclusivity and tax credits for companies that develop drugs for rare pediatric diseases, thus encouraging the development of drugs for conditions that might otherwise be neglected due to the small patient population.
EMA Pediatric Investigation Plans (PIPs):
The European Medicines Agency (EMA) mandates PIPs for new drugs that are intended to be used in pediatric populations. These plans require companies to conduct clinical trials for pediatric use, ensuring that new treatments are safe and effective for children.
Such regulations and incentives make it financially viable for pharmaceutical companies to invest in pediatric drug development despite the smaller market size.
Technological Advances:
Artificial Intelligence (AI):
AI is playing a key role in pediatric drug development by analyzing vast datasets to discover potential drug candidates, optimize clinical trials, and identify personalized treatment options for pediatric patients. AI models can predict how children will respond to medications based on age, weight, and genetic factors, enabling more precise and safer treatments.
Biotechnology and Personalized Medicine:
Advances in biotechnology, including the development of gene therapies and monoclonal antibodies, are revolutionizing pediatric treatments, especially in rare and complex diseases like childhood cancers and genetic disorders.
Personalized medicine ensures that treatments are tailored to the individual needs of a child, considering factors like genetics and environmental influences.
Restraints:
Limited Market Exclusivity:
Shorter Patent Protection:
Pediatric drugs face a significant restraint due to the shorter patent protection periods compared to adult drugs. This limits the financial return on investment, as pharmaceutical companies cannot secure long-term exclusivity for pediatric formulations. Shorter exclusivity periods decrease the incentive to invest in pediatric-specific drugs, especially when the patient population is smaller.
As a result, the pediatric segment often lacks the same level of research and development investment seen in adult drug markets.
High Costs:
Development Costs:
The development of specialized pediatric drugs is costly due to the unique nature of clinical trials required. Clinical trials for pediatric drugs typically require smaller sample sizes, but they must still adhere to rigorous standards of safety and efficacy, especially since children have different physiological responses than adults.
Additionally, pediatric drugs often need to be formulated in specific ways (e.g., chewable tablets, syrups), which adds to the cost of production.
Smaller Clinical Trial Pools:
Pediatric trials often face logistical and ethical challenges in recruiting participants, especially for rare diseases. The smaller patient pool leads to higher trial costs and extended timelines.
Opportunities:
Emerging Markets:
Asia-Pacific and Latin America:
As the pediatric population grows in Asia-Pacific (China, India) and Latin America, these regions represent significant growth opportunities for the pediatric drugs market. The rising demand for specialized healthcare and increased access to healthcare infrastructure in these regions will further accelerate the adoption of pediatric medications.
Government Initiatives: Many countries in Asia-Pacific and Latin America are introducing healthcare reforms, improving access to medications, and encouraging pharmaceutical companies to invest in local drug manufacturing and development.
R&D Investments:
Clinical Trials and Drug Development:
Increased investments in pediatric clinical trials will fuel the growth of the pediatric drugs market. Pharmaceutical companies are allocating more resources to create drugs specifically for pediatric use, and the global increase in pediatric clinical trials will drive innovation in this market.
The increasing focus on pediatric oncology and neurological disorders is another area of investment, with an emphasis on developing drugs that cater to these complex needs.
Collaborative Research and Partnerships:
Pharmaceutical companies are forming strategic partnerships with universities, research institutes, and government organizations to advance pediatric drug research. These collaborations aim to bridge the gap in pediatric drug development and bring new treatments to market more efficiently.
Challenges:
Lack of Pediatric-Specific Formulations:
Gap in Pediatric Drug Availability:
There is a significant lack of pediatric-specific formulations, and many drugs are off-label when prescribed for children. Since children’s physiological responses differ from those of adults, adult medications may not always be safe or effective for children. The absence of age-appropriate formulations such as liquid medications, chewable tablets, or topical creams can complicate treatment.
Regulatory bodies, such as the FDA and EMA, are encouraging companies to create pediatric-friendly formulations, but the progress has been slow.
Off-Label Use:
Safety and Effectiveness Issues:
Due to the limited availability of pediatric drugs, off-label use of adult medications in children is common. However, this presents a safety risk because these medications may not have been tested for pediatric populations, leading to potential adverse effects or inadequate dosing.
The lack of age-appropriate formulations and proper dosing guidelines for children increases the likelihood of medication errors, especially in young children or those with unique medical conditions.
Top 10 Companies in Pediatric Drugs Market
Pfizer Inc.:
Overview: Leading player with a wide portfolio in pediatric vaccines and antibiotics.
Strength: Extensive research and global market reach.
Recent Developments: Developed pediatric COVID-19 vaccines.
Novartis International AG:
Overview: Focuses on pediatric oncology and gene therapies.
Strength: Strong pipeline in pediatric rare diseases.
Recent Developments: Approved treatments for pediatric cancers.
GSK plc.:
Overview: Leader in pediatric vaccines.
Strength: Strong global presence, especially in respiratory diseases.
Recent Developments: Focused on RSV vaccine for children.
Sanofi S.A.:
Overview: Specializes in pediatric allergy and respiratory drugs.
Strength: Robust portfolio in respiratory conditions.
Recent Developments: Investment in childhood diabetes treatments.
AstraZeneca plc:
Overview: Strong presence in pediatric asthma medications.
Strength: Innovation in respiratory disorders.
Recent Developments: Approved pediatric asthma treatment.
Merck & Co., Inc.:
Overview: Leading in pediatric vaccines and infectious diseases.
Strength: Pioneering in pediatric immunizations.
Recent Developments: Pediatric HPV vaccine launch.
Johnson & Johnson:
Overview: Major player in pediatric oncology and neurology treatments.
Strength: Extensive pediatric pipeline.
Recent Developments: Pediatric ADHD treatment.
Roche Holding AG:
Overview: Focus on pediatric oncology and immunology.
Strength: Expertise in genetic disorders.
Recent Developments: Pediatric gene therapy for rare diseases.
Eli Lilly and Company:
Overview: Focus on pediatric diabetes and neurological conditions.
Strength: Innovative in pediatric hormone therapies.
Recent Developments: Pediatric insulin approval.
Abbott Laboratories:
Overview: Focus on gastrointestinal and respiratory pediatric drugs.
Strength: Broad product range in child health.
Recent Developments: New GI drug formulations for children.
Latest Announcements
Cumberland Pharmaceuticals: FDA granted Orphan Drug and Rare Pediatric Disease Designation for Ifetroban for treating cardiomyopathy in children with Duchenne muscular dystrophy.
UK Charities: $36 million investment to develop new drugs for childhood tumors, forming a global consortium.
ICH: Released ICH E11A guideline for extrapolating adult drug data to pediatrics, nearing regulatory adoption.
Harmony Biosciences: FDA approved WAKIX for excessive daytime sleepiness in pediatric narcolepsy patients.
Segments Covered in the Pediatric Drugs Market
Drug Types
Respiratory Drugs:
Leading Segment: Respiratory disorders are one of the most common conditions affecting children, making respiratory drugs a dominant category in the pediatric drugs market.
Market Drivers: The rise in conditions like asthma, chronic obstructive pulmonary disease (COPD), and pneumonia is contributing to the high demand for respiratory treatments in children.
Key Products:
Montelukast for asthma and exercise-induced bronchoconstriction.
Bronchodilators and corticosteroids for conditions like asthma and chronic respiratory infections.
Market Trends: There is an increasing focus on developing long-acting, child-friendly formulations, such as inhalers and nebulizers, that are easy to use for children.
Gastrointestinal Drugs:
Expanding Segment: Gastrointestinal (GI) disorders are prevalent in pediatric populations due to issues like inflammatory bowel disease (IBD), constipation, gastroesophageal reflux disease (GERD), and diarrhea.
Market Drivers: The increasing incidence of GI diseases, especially in children with food allergies or those affected by genetic conditions, is pushing the demand for pediatric GI drugs.
Key Products:
Ursodiol for biliary atresia.
Clarithromycin and mesalamine for conditions like IBD and peptic ulcers.
Market Trends: The market is moving toward more child-friendly formulations, such as chewable tablets, liquids, and suspensions, that are easier for young children to ingest.
Oncology Drugs:
Growing Segment: Pediatric cancers, while less common than adult cancers, are seeing increasing rates, making the development of pediatric oncology drugs essential.
Market Drivers: With rising incidences of cancers like childhood leukemia, brain tumors, and lymphoma, there is a growing need for specialized drugs in this category.
Key Products:
Debrafenib and Trametinib for treating pediatric low-grade gliomas.
Nivolumab for treating pediatric melanoma.
Market Trends: The growing emphasis on personalized treatments and immunotherapies for pediatric cancers is driving the development of innovative therapies. There is also a significant focus on developing biologic therapies and gene therapies to target childhood cancers more effectively.
Routes of Administration
Oral Administration:
Dominant Route: Oral administration remains the most common route for pediatric drugs due to its non-invasive nature and ease of use for both patients and caregivers.
Market Drivers: The preference for oral medications is driven by the simplicity of administration, cost-effectiveness, and parental comfort. Moreover, children are generally more accepting of oral medications than injections.
Key Products:
Antibiotics, analgesics, and antipyretics are often available in liquid or chewable tablet formulations to suit younger children.
Market Trends: Flavored syrups, chewable tablets, and effervescent formulations are gaining traction as they help improve adherence to medication in children.
Parenteral Administration:
Fast-Growing Segment: The parenteral route, including intravenous (IV), intramuscular (IM), and subcutaneous (SC) injections, is becoming increasingly important, particularly in situations where children cannot take medications orally (e.g., infants or those with severe nausea).
Market Drivers: The increasing demand for vaccinations and the need for rapid drug delivery in critical care scenarios (such as pediatric oncology or sepsis) are contributing to this segment’s growth.
Key Products:
Vaccines for diseases such as measles, polio, and meningitis.
Immunotherapy drugs and biologic agents for diseases like cancer or autoimmune disorders.
Market Trends: There is a significant push to develop pediatric-friendly injectables, such as autoinjectors for at-home use, which increase comfort and convenience for both patients and caregivers.
Distribution Channels
Hospital Pharmacies:
Leading Channel: Hospital pharmacies are the dominant distribution channel in the pediatric drugs market due to the specialized nature of pediatric care and the need for expert consultation.
Market Drivers: Hospital pharmacies provide a centralized source for specialized pediatric formulations and can offer a higher level of support for complex therapies, including those for chronic diseases, cancer, and genetic disorders.
Key Products: Specialized formulations, such as pediatric injectable drugs, IV antibiotics, and chemotherapy drugs, are predominantly dispensed through hospital pharmacies.
Market Trends: Hospital pharmacies are expanding their roles to include pharmacogenomic counseling, which helps tailor treatments to individual pediatric patients, further enhancing their role in pediatric healthcare.
Online Pharmacies:
Fastest-Growing Channel: Online pharmacies have emerged as the fastest-growing distribution channel, particularly for non-urgent, prescription-based medications.
Market Drivers: The increasing use of the internet for healthcare needs, along with the convenience of home delivery, has made online pharmacies especially popular among caregivers seeking easy access to pediatric drugs.
Key Products: Common pediatric drugs like antibiotics, antipyretics, and over-the-counter (OTC) remedies are widely available through online platforms.
Market Trends: The growth of telemedicine and online doctor consultations is driving demand for online pharmacies. In addition, online pharmacies are becoming more integrated with e-health platforms to offer a seamless experience, from prescription to medication delivery.
Top 5 FAQs
What is the expected growth rate of the pediatric drugs market?
The market is projected to grow at a CAGR of 12.2% from USD 162.56 billion in 2025 to USD 458.09 billion by 2034.
What are the key segments in the pediatric drugs market?
Key segments include respiratory drugs, gastrointestinal drugs, oral drugs, and parenteral drugs.
Which regions dominate the pediatric drugs market?
North America leads, with Asia-Pacific expected to grow at the fastest rate.
What role does AI play in pediatric drug development?
AI accelerates drug discovery, personalizes treatments, optimizes clinical trials, and enhances medication adherence.
What are the challenges in pediatric drug development?
Challenges include limited market exclusivity, small patient populations, and the need for off-label drug use.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout Pediatric Drugs Market Report Now at: https://www.towardshealthcare.com/checkout/5074
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest