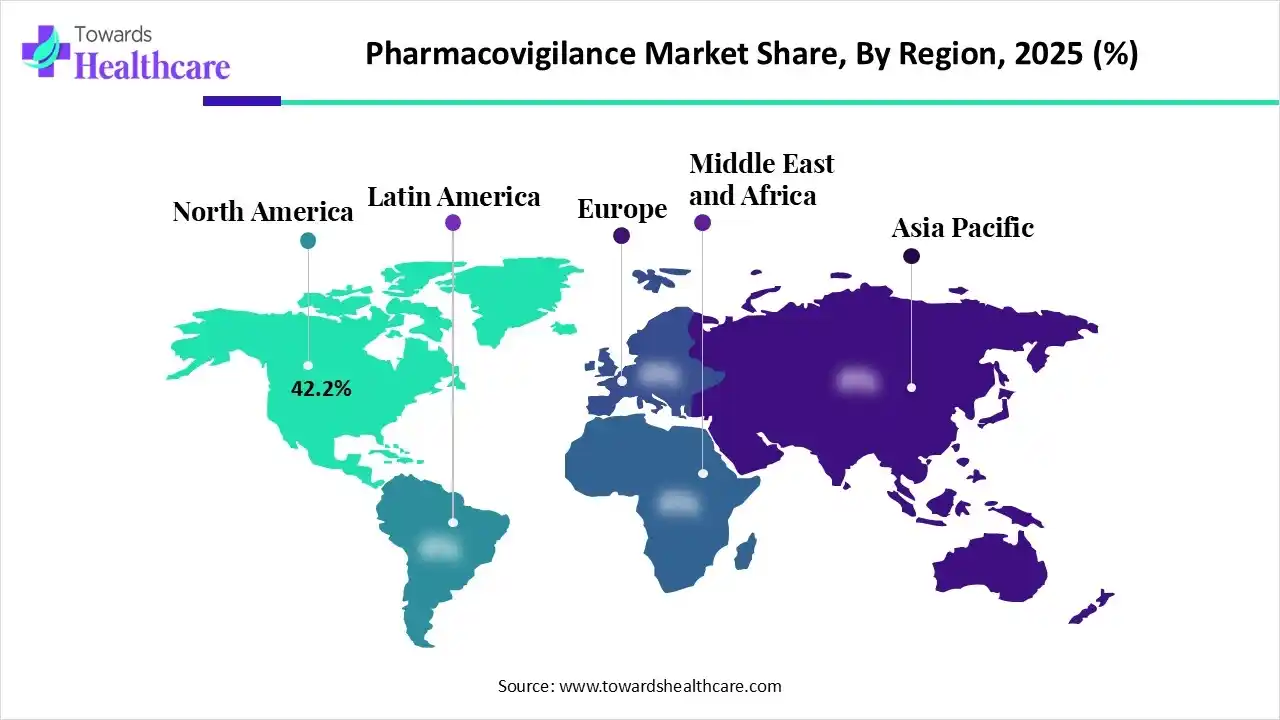
Pharmacovigilance focuses on identifying, monitoring, and preventing adverse drug reactions (ADRs). It ensures that medicines remain safe even after large populations begin using them in real-world conditions.
As healthcare systems become more complex and drug consumption rises globally, pharmacovigilance is transforming into a data-driven, technology-enabled discipline.
The global pharmacovigilance ecosystem is expanding rapidly. Regions around the world are strengthening regulatory frameworks, investing in AI-powered safety systems, and building stronger reporting networks to track drug safety.
In 2025, regional dynamics clearly show how different healthcare systems contribute to the growth and evolution of this crucial sector.
North America Sets the Global Benchmark in Drug Safety
North America continues to dominate the pharmacovigilance landscape.
In 2025, the region captured 42.5% of the global market share, reflecting its well-established regulatory ecosystem and advanced healthcare infrastructure.
The region benefits from strict safety protocols and a culture that prioritizes patient protection.
Regulatory bodies enforce detailed monitoring requirements that compel pharmaceutical companies to continuously track and report drug safety data.
Several factors strengthen North America’s leadership in pharmacovigilance:
-
A strong regulatory framework enforcing strict safety monitoring
-
High adoption of artificial intelligence in safety signal detection
-
Extensive clinical research activities generating large safety datasets
-
Robust adverse drug reaction reporting systems across healthcare networks
Together, these elements create a sophisticated safety environment where drug monitoring continues throughout a medicine’s lifecycle.
Pharmaceutical companies, regulators, and healthcare providers collaborate closely to ensure that safety signals are identified early and addressed quickly.
The United States Drives the Region’s Pharmacovigilance Power
Within North America, the United States leads the pharmacovigilance market with the largest revenue share.
The country’s pharmaceutical industry produces enormous volumes of clinical data, which strengthens drug monitoring capabilities.
Advanced healthcare infrastructure and a strong culture of regulatory compliance help the country maintain leadership in drug safety surveillance.
Key drivers behind the U.S. dominance include:
-
Large-scale clinical trial activity generating continuous safety insights
-
Strong investments in AI-driven pharmacovigilance platforms
-
Highly structured drug safety reporting frameworks
-
Active participation from global pharmaceutical companies
These elements create an environment where safety monitoring becomes proactive rather than reactive.
Digital tools, automation, and advanced analytics allow researchers to detect safety signals earlier than ever before.
Asia-Pacific Emerges as the Fastest-Growing Pharmacovigilance Hub
While North America leads the market today, the Asia-Pacific region is rapidly reshaping the future of pharmacovigilance.
The region is projected to grow at the fastest pace, with a CAGR of 7.8% during the forecast period.
Several structural shifts in the healthcare and pharmaceutical industries are fueling this rapid expansion.
Governments across Asia-Pacific are strengthening drug safety regulations while pharmaceutical production continues to rise across the region.
This transformation is turning Asia-Pacific into a global hub for pharmacovigilance services.
Major growth drivers include:
-
Expanding pharmaceutical manufacturing capacity
-
Rapid growth in clinical trial activities
-
Increasing digitalization of healthcare systems
-
Rising awareness of adverse drug reactions among healthcare professionals
In addition, many global pharmaceutical companies are outsourcing pharmacovigilance operations to Asia-Pacific due to its highly skilled workforce and cost advantages.
Countries such as India and China are becoming key service centers for global drug safety monitoring.
China Accelerates Drug Safety Innovation
China stands out as a major force behind Asia-Pacific’s pharmacovigilance expansion.
The country’s rapidly growing pharmaceutical sector continues to generate large volumes of clinical and post-marketing safety data.
China has also strengthened its regulatory environment in recent years, creating stricter monitoring requirements for drug safety reporting.
The national healthcare system is investing heavily in advanced digital pharmacovigilance infrastructure.
Several developments reinforce China’s leadership in the region:
-
Expansion of large-scale clinical trials across multiple therapeutic areas
-
Strong government regulation focused on patient safety
-
Rapid adoption of artificial intelligence for adverse event detection
-
Growth of real-world data platforms for drug monitoring
China’s massive population also contributes to the availability of extensive drug safety data, which strengthens pharmacovigilance research and surveillance.
As the country continues to modernize its healthcare ecosystem, its role in global drug safety monitoring is expected to expand significantly.
Europe Builds a Collaborative Safety Network
Europe represents another important pillar in the global pharmacovigilance landscape.
The region’s approach emphasizes collaboration between regulatory agencies, healthcare providers, and pharmaceutical companies.
Strong regulatory oversight combined with cross-border cooperation enables Europe to maintain a robust drug safety monitoring environment.
European healthcare systems increasingly rely on digital reporting platforms and real-world evidence programs to track medicine performance.
The region’s pharmacovigilance growth is supported by several structural strengths:
-
Well-established drug safety networks across multiple countries
-
Expanding use of digital adverse event reporting systems
-
Increasing use of real-world data for monitoring medicine outcomes
-
Growing investment in AI-based safety monitoring technologies
European authorities also place strong emphasis on patient safety and transparency, ensuring that safety information flows quickly across borders.
The United Kingdom Strengthens Digital Drug Safety Systems
Within Europe, the United Kingdom has emerged as a leading contributor to pharmacovigilance innovation.
The country’s drug safety monitoring framework operates with a highly structured regulatory system and strong collaboration between healthcare providers and regulatory authorities.
One of the most recognized pharmacovigilance initiatives in the country is its national adverse drug reaction reporting platform.
This system encourages healthcare professionals and patients to report drug-related issues, creating a rich safety database for analysis.
The United Kingdom continues to enhance its leadership through:
-
Advanced digital reporting platforms for adverse drug reactions
-
Strong integration of AI technologies into drug safety monitoring
-
Active clinical research programs generating valuable safety insights
-
Close cooperation between regulators, hospitals, and pharmaceutical companies
These initiatives enable faster detection of safety signals and strengthen the country’s proactive approach to medicine monitoring.
Access our exclusive, data-rich dashboard dedicated to the pharmaceutical market – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout the Market Report Now at: https://www.towardshealthcare.com/checkout/6504
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium