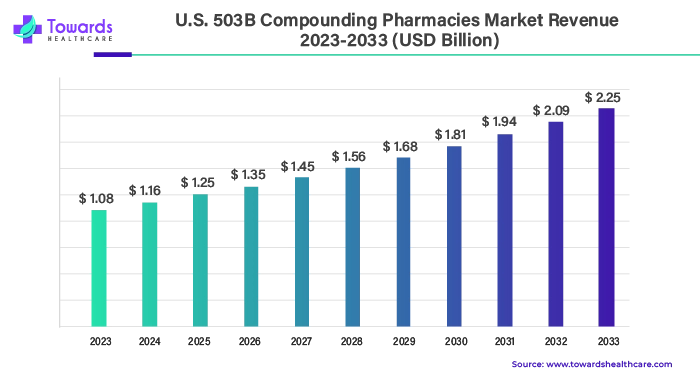
The U.S. 503B compounding pharmacies market, valued at US$ 1.25 billion in 2025, is projected to reach US$ 2.42 billion by 2034, growing at a CAGR of 7.63%, driven by rising sterile injectable demand, AI integration, and regulatory support.
Download the free sample and get the complete insights and forecasts report on this market @ https://www.towardshealthcare.com/download-sample/5180
Market Size
Market Value & Growth
➤2024: US$ 1.16 billion
➤2025: US$ 1.25 billion
➤Projected 2034: US$ 2.42 billion
➤CAGR (2025–2034): 7.63%
Drivers of Market Size Expansion
➤Surge in demand for sterile injectables in hospitals, ICUs, ERs, and surgical centers.
➤Increased adoption of cancer care, anesthesia, and high-potency drugs.
➤Growing trust in FDA-regulated 503B facilities producing bulk sterile medications.
Regulatory Influence on Market Growth
➤FDA guidance (Jan 2025) on bulk drug substance compounding under 503B enables more robust production, increasing market scalability.
➤Regular inspections and CGMP adherence increase market credibility and acceptance.
Investment & Expansion Trends
➤Acquisitions, such as Revelation Pharma acquiring AleraCare Pharmacy, expand production capacity and market share.
➤Emerging technologies and AI automation improve productivity and market efficiency.
Market Trends
Expansion of Compounding Facilities
➤Increased investment from pharmaceutical and healthcare industries.
➤FDA guidance supporting bulk drug compounding accelerates growth.
Acquisitions & Collaborations
➤Example: Revelation Pharma’s acquisition of AleraCare Pharmacy operations, rebranded as OmniScript Compounding.
Rising Sterile Injectable Usage
➤Essential for pain management, antibiotics, antivirals, sedatives, anesthesia, and electrolyte replacement therapy.
➤Expected global demand growth: over 7% annually over the next decade.
AI Integration in Compounding
➤Monitoring processes in real-time.
➤Detecting anomalies in documentation and ensuring compliance.
➤Automating batch production and minimizing contamination.
Addressing Drug Shortages
➤Over 300 medications are in short supply in the U.S. (FDA 2025).
➤503B facilities produce essential drugs at scale to meet hospital demand.
Regulatory Guidance Boosts Innovation
➤FDA updates allow for safer, legal, and faster adoption of new compounding methods.
Adoption by Hospitals and Ambulatory Surgical Centers
➤Over 60% of U.S. hospitals source sterile compounds from 503B pharmacies.
North America as a Market Leader
➤Advanced healthcare infrastructure drives demand.
➤U.S.: 80+ FDA-registered 503B outsourcing facilities with continuous growth.
Canada’s Growing Market
➤Federal/provincial funding supports upgrades in compounding practices.
➤Emphasis on cleanroom expansion, sterility assurance, and pharmacy automation.
Innovation in Operations
➤Digital Factory Platforms and data-driven manufacturing are redefining 503B production efficiency.
AI Impact on U.S. 503B Compounding Pharmacies Market
Real-Time Process Monitoring
➤AI monitors every step of sterile compounding, including mixing, filling, and packaging.
➤Sensors and AI software track temperature, humidity, and pressure in cleanrooms to ensure sterility.
➤Early detection of deviations reduces batch failures and wastage, improving overall efficiency.
Quality Control & Error Detection
➤AI algorithms compare current production batches to historical data and standard formulations.
➤Identifies errors such as incorrect concentrations, improper mixing times, or contamination risks.
➤Facilities leveraging AI report up to 40% reduction in batch errors, enhancing safety.
Regulatory Compliance Automation
➤AI automates documentation, ensuring CGMP and FDA record compliance.
➤Reduces human error in maintaining batch logs, ingredient verification, and audit trails.
➤Facilitates faster FDA inspections by generating accurate, real-time compliance reports.
Predictive Maintenance of Equipment
➤AI predicts when critical equipment (autoclaves, filling machines, isolators) may fail.
➤Prevents unplanned downtime in sterile compounding facilities.
➤Ensures continuous production for high-demand injectables in hospitals and ICUs.
Contamination Risk Minimization
➤Robotic arms and AI-assisted compounding limit human touch, reducing contamination.
➤Real-time air quality monitoring and AI-controlled environmental adjustments maintain cleanroom integrity.
Batch Optimization
➤AI analyzes past production data to determine optimal batch sizes for efficiency.
➤Balances inventory availability, demand forecasts, and cost reduction.
➤Prevents both overproduction (waste) and underproduction (drug shortages).
Inventory Forecasting
➤AI predicts hospital demand for sterile injectables based on historical consumption, seasonal trends, and emergency cases.
➤Ensures hospitals never face shortages of critical medications like antibiotics or oncology injectables.
Data-Driven Formulation Development
➤AI accelerates the development of new compounded drugs by analyzing chemical stability and sterility compatibility.
➤Supports formulation innovation for high-risk medications, such as chemotherapy and anesthetics.
Supply Chain Efficiency
➤AI optimizes distribution routes, delivery schedules, and storage conditions.
➤Reduces delays in shipping sterile injectables to hospitals, ERs, and ICUs.
➤Improves cost-efficiency and patient access to critical drugs.
Operational Cost Reduction
➤AI and automation streamline repetitive tasks like labeling, filling, and batch documentation.
➤Reduces labor dependency, lowers production costs, and speeds up turnaround times.
➤Enables facilities to scale operations without proportionally increasing workforce or errors.
Regional Insights
North America
Market Leadership
➤Advanced healthcare infrastructure and regulatory oversight make North America the leader in 503B compounding.
➤Hospitals and clinics depend on outsourcing facilities for consistent, sterile injectable supply.
Hospital Dependence
➤Over 60% of U.S. hospitals source sterile compounds from 503B pharmacies due to reliability and FDA oversight.
FDA Oversight
➤80+ FDA-registered outsourcing facilities ensure adherence to CGMP, sterility, and quality standards.
➤Regular unannounced inspections maintain high compliance and safety levels.
Drivers
➤Rising oncology treatments and anesthesia demand in surgical centers.
➤Increased use of small-dose, high-potency injectables in ICUs, ERs, and ambulatory surgical centers (ASCs).
Trends
➤Growth of ASCs is driving demand for ready-to-use sterile injectables.
➤AI and digital platforms are increasingly adopted for efficiency, error reduction, and regulatory compliance.
Canada
Government Support
➤Federal and provincial funding accelerates sterile compounding adoption in hospitals.
Regulatory Encouragement
➤Cleanroom expansions, sterility assurance, and alignment with U.S. FDA standards.
Technological Investment
➤Hospitals adopt AI-assisted automation for safer and consistent sterile drug production.
➤Investments enhance patient care quality, reduce shortages, and standardize compounding procedures.
Market Dynamics
Drivers
➤Rising infectious diseases, chronic illnesses, and emergency care needs.
➤Increased demand for sterile injectables in ICUs, ERs, and surgical centers.
➤AI-driven accuracy and automation reduce production errors, improve efficiency, and enhance sterility.
➤FDA guidance on bulk drug compounding supports innovation and market growth.
➤Strategic acquisitions and partnerships expand production capacity and market reach.
Restraints
Stringent CGMP regulations require continuous compliance investment.
➤Frequent FDA inspections and audits increase operational pressure.
➤High cost of automation, AI integration, and cleanroom infrastructure.
➤Skilled workforce required for managing advanced AI systems and robotics.
Opportunities
➤Filling critical hospital drug shortages, particularly for oncology and emergency injectables.
➤Expansion into high-demand segments like oncology, anesthetics, and ICU-focused medications.
➤Integration of digital factory and AI-driven operational platforms for efficiency and compliance.
➤Cross-border expansion, particularly into Canadian hospitals, leveraging funding and regulatory alignment.
➤Research-driven development of new sterile injectable formulations for unmet medical needs.
Challenges
➤Rapidly evolving FDA regulations require constant adaptation and updates.
➤Workforce training and retention for AI and automated systems in sterile compounding.
➤Ensuring sterility and compliance while scaling up bulk production.
➤Managing high capital investments in AI, robotics, and cleanroom infrastructure.
Top 10 Companies in the 503B Compounding Pharmacies Market
1. SCA Pharma
Overview: SCA Pharma is a leading provider of customized sterile injectable solutions, serving hospitals, surgery centers, and healthcare systems across the U.S.
Products: Offers a wide range of hospital-compounded sterile injectables, including anesthetics, analgesics, antibiotics, and oncology supportive care drugs.
Strengths:
➤Nationwide distribution network ensures timely delivery to high-demand facilities.
➤Strong regulatory compliance expertise, adhering to CGMP and FDA guidelines.
➤Focused on patient-specific compounding, enhancing clinical outcomes.
Strategic Advantage: Integrates advanced quality management systems and process validation, minimizing batch errors.
2. Athenex, Inc.
Overview: Specializes in oncology and critical care sterile injectables, leveraging advanced compounding technology.
Products: Oncology injectables, supportive care medications, and critical care drugs for hospital use.
Strengths:
➤High-quality production with stringent sterility and potency control.
➤Emphasis on patient safety, particularly in chemotherapy and ICU care.
➤Operational scalability to meet increased hospital demand during emergency situations.
Strategic Advantage: Invests in AI monitoring and automated compounding systems for precision and efficiency.
3. Nephron Pharmaceuticals Corporation
Overview: Large-scale production of sterile injectables and respiratory medications.
Products: Inhalation solutions, IV antibiotics, anesthesia drugs, and other high-volume sterile injectables.
Strengths:
➤Nationwide hospital supply chain ensures broad accessibility.
➤High-volume capacity allows for rapid response to demand surges.
➤Focus on regulatory compliance and quality control, reducing risk of recalls.
Strategic Advantage: Integrates process automation and digital quality checks to optimize production efficiency.
4. Wells Pharma of Houston LLC
Overview: Focuses on providing 503B sterile compounded products to hospitals and clinics.
Products: Pain management injectables, anesthetics, antibiotics, sedatives, and electrolyte replacement therapies.
Strengths:
➤Fast turnaround for emergency and surgical care medications.
➤Compliance-focused approach ensures FDA and CGMP adherence.
➤Flexible production capacity for small-batch and high-potency compounds.
Strategic Advantage: Strong reputation in critical care pharmacy support, making it a preferred vendor for ER and ICU medications.
5. Fagron Compounding Pharmacies
Overview: Global leader in pharmaceutical compounding, delivering sterile and non-sterile solutions.
Products: Oncology injectables, hormone therapies, personalized sterile compounds, and wellness medications.
Strengths:
➤Highly customizable solutions for individual patient needs.
➤Innovation-focused, leveraging new compounding technologies and formulations.
➤Global operational experience ensures best practices and quality consistency.
Strategic Advantage: Strong R&D pipeline for new sterile formulations, supporting both hospital and outpatient care.
6. Olympia Pharmacy
Overview: Offers a broad portfolio of 503B compounded injectables, including hormone therapy and wellness products.
Products: Hormone replacement therapies, aesthetic injectables, and sterile medications for hospital use.
Strengths:
➤Specialized solutions for niche medical areas like hormone therapy and wellness care.
➤Patient-centered services, ensuring customization and safety.
➤Rapid adaptation to new FDA guidelines and bulk drug substance approvals.
Strategic Advantage: Combines clinical expertise with personalized compounding, targeting both hospital and outpatient markets.
7. Revelation Pharma
Overview: Nationwide network of 503A and 503B compounding pharmacies.
Products: Sterile injectable medications for oncology, anesthesia, and ICU needs.
Strengths:
➤Strong regulatory compliance and quality assurance programs.
➤Expansion via acquisitions, e.g., AleraCare Pharmacy operations rebranded as OmniScript Compounding.
➤Centralized operations for uniform standards across multiple facilities.
Strategic Advantage: Robust regulatory and operational expertise, allowing rapid scaling in response to drug shortages.
8. Asteria Health
Overview: 503B outsourcing facility leveraging data-driven, AI-enhanced operations.
Products: Sterile injectables for oncology, anesthesia, and critical care.
Strengths:
➤Integration of Digital Factory Platform for optimized production and error reduction.
➤Focused on operational excellence and high-quality batch consistency.
➤Scalable solutions to meet hospital and ICU demand efficiently.
Strategic Advantage: Combines technology-driven manufacturing with regulatory compliance, enabling innovative compounding.
9. BayCare Health System 503B Facility
Overview: FDA-approved hospital compounding center ensuring local drug availability.
Products: Sterile injectables for surgical centers, oncology units, and emergency care.
Strengths:
➤Focused on eliminating local drug shortages, improving patient care.
➤High standards of CGMP compliance and sterility assurance.
➤Recognition via awards, e.g., Pharmacy Compounding Excellence Award 2025.
Strategic Advantage: Hospital-centered approach enables direct alignment with clinical needs and faster delivery.
10. SCW Pharma
Overview: Next-generation pharmaceutical manufacturing company.
Products: Sterile injectable drugs, oncology medications, and critical care compounds.
Strengths:
➤Integration of advanced digital platforms for production optimization.
➤Operational excellence and quality assurance embedded in all processes.
➤Collaborative projects, e.g., with Asteria Health, to enhance 503B compounding efficiency.
Strategic Advantage: Combines innovation, automation, and strategic partnerships to drive growth and market leadership.
Latest Announcements
July 2025: BayCare Health System received the Pharmacy Compounding Excellence Award for building one of four FDA-approved 503B facilities in the U.S., ensuring drug supply for the Tampa Bay community.
January 2025: FDA released guidance on bulk drug substances for 503A and 503B pharmacies, supporting safe and scalable compounding.
January 2025: Revelation Pharma acquired AleraCare Pharmacy operations; rebranded as OmniScript Compounding.
Recent Developments
August 2025: SCW and Asteria Health launched a next-gen data-driven manufacturing site in Birmingham, Alabama.
Impact: Combines AI, Digital Factory Platform, and operational excellence for 503B compounding.
February 2025: Nancy Fingerhut promoted as VP of Regulatory Affairs & Quality Assurance at Revelation Pharma.
Impact: Enhances compliance, patient safety, and adherence to industry standards.
Segments Covered
1. Facility Type
503B Outsourcing Pharmacies: These are FDA-registered facilities authorized to compound sterile medications in bulk without individual patient prescriptions. They adhere to Current Good Manufacturing Practices (CGMP) and undergo regular FDA inspections. U.S. Food and Drug Administration
503A Pharmacies: Traditional compounding pharmacies that prepare medications based on individual patient prescriptions. They are regulated by state boards of pharmacy and are not permitted to compound drugs in bulk.
2. Drug Type
Sterile Injectables: These are medications administered through injection or infusion, requiring strict sterility standards. They are critical in various medical settings, including:
Oncology: Chemotherapy agents and supportive care medications.
Anesthesia: Induction and maintenance agents for surgical procedures.
Antibiotics and Antivirals: For treating infections in hospitalized patients.
Sedatives: Used in intensive care units and emergency departments.
Electrolyte Replacement Therapies: To correct imbalances in critically ill patients.
3. Technology Integration
Artificial Intelligence (AI): AI is increasingly utilized in 503B pharmacies to enhance various aspects of compounding:
Real-Time Monitoring: AI systems monitor compounding processes to ensure adherence to protocols and detect anomalies.
Documentation and Compliance: AI assists in verifying batch records and ensuring compliance with regulatory standards.
Automation of Batch Production: AI-driven automation improves efficiency and consistency in producing sterile injectables.
Contamination Risk Reduction: By minimizing manual handling, AI reduces the risk of contamination during compounding.
Error Reduction: Facilities leveraging AI have reported up to a 40% reduction in errors and significantly faster turnaround times.
Automation: Robotic systems and automated filling equipment streamline production processes, enhancing throughput and consistency.
4. Geography
North America: The U.S. and Canada are at the forefront of the 503B compounding market due to:
Infrastructure: Advanced healthcare facilities and regulatory frameworks support the growth of 503B pharmacies.
Regulation: The FDA’s oversight ensures adherence to stringent manufacturing standards.
Hospital Adoption: Over 60% of U.S. hospitals source sterile compounds from 503B pharmacies.
Canada: While smaller in scale, Canada’s focus on improving patient care through cleanroom expansion and sterility assurance is driving growth in sterile compounding.
5. Investment & Expansion
Acquisitions and Partnerships: Companies are expanding their reach and capabilities through strategic acquisitions and partnerships.
Example: In January 2025, Revelation Pharma acquired AleraCare Pharmacy’s compounding operations, rebranding them as OmniScript Compounding.
Digital Innovation: Investments in digital platforms and AI technologies are enhancing operational efficiency and compliance.
Example: In August 2025, SCW and Asteria Health announced a collaboration to launch a next-generation, data-driven pharmaceutical manufacturing site in Birmingham, Alabama.
Top 5 FAQs
1 What are 503B compounding pharmacies?
FDA-regulated facilities producing sterile medications in bulk for hospitals, ICUs, and ERs, adhering to CGMP standards.
2 Why is the U.S. 503B market growing?
Driven by increased sterile injectable usage, AI adoption, and regulatory guidance supporting bulk compounding.
3 What role does AI play in 503B compounding?
Ensures accuracy, reduces errors, optimizes production, and maintains regulatory compliance.
4 Which regions lead the 503B compounding market?
North America dominates due to advanced infrastructure, hospital dependence, and FDA oversight; Canada is expanding via government funding.
5 Who are the top companies in this market?
SCA Pharma, Athenex, Nephron Pharmaceuticals, Wells Pharma, Fagron, Olympia Pharmacy, Revelation Pharma, Asteria Health, BayCare Health System, SCW Pharma.
Access our exclusive, data-rich dashboard dedicated to the therapeutic area sector – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Immediate Delivery Available | Buy This Premium Research @ https://www.towardshealthcare.com/price/5180
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Web: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest