The future of medicine is not arriving suddenly—it is unfolding inside clinical trial labs across the United States.
Cell and gene therapy (CGT) clinical trials are no longer experimental ideas. They are becoming structured, large-scale efforts aimed at curing diseases rather than just managing them.
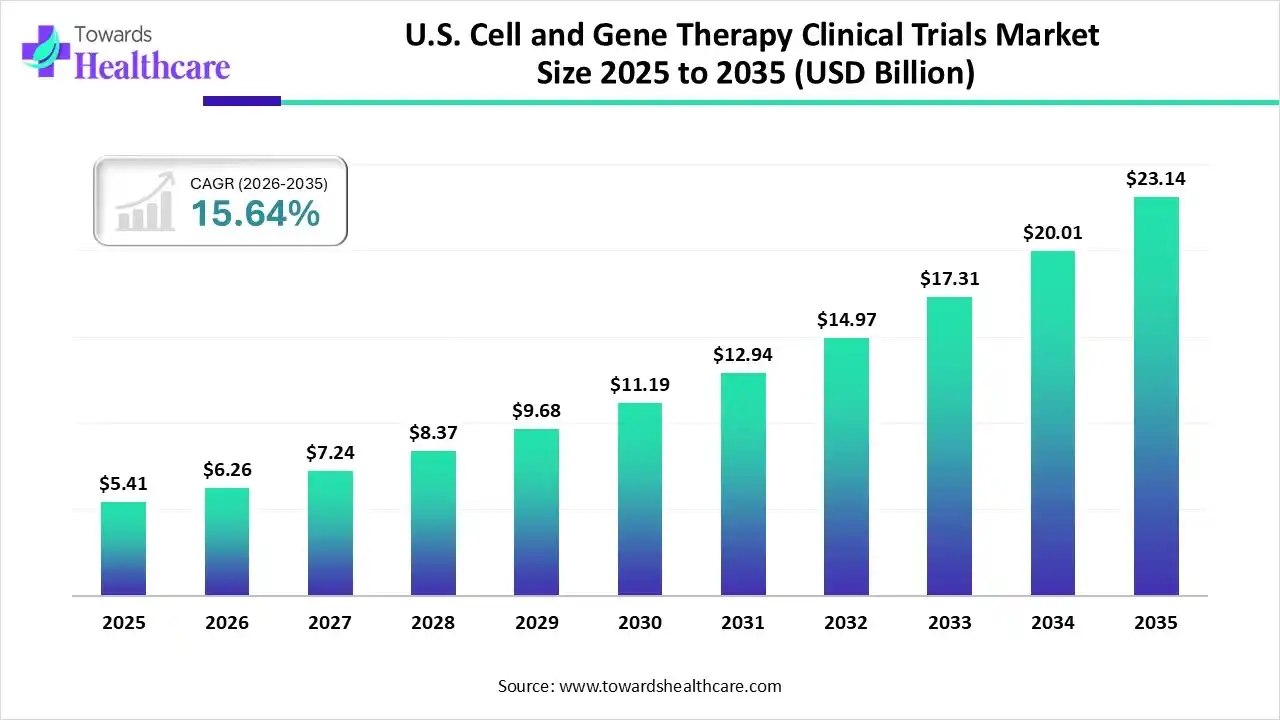
With the U.S. Cell and Gene Therapy Clinical Trials Market projected to reach $23.14 billion by 2035, the pace of transformation is impossible to ignore. This is not just growth; it is a shift in how healthcare thinks, tests, and treats.
Download Free Sample: https://www.towardshealthcare.com/download-sample/6607
Why This Market Is Growing So Fast
Several powerful forces are driving this expansion. The most important one is simple: unmet medical needs.
Diseases like cancer, rare genetic disorders, and inherited conditions still lack effective long-term treatments. CGT offers something different—potential one-time therapies with lasting impact.
At the same time, the U.S. ecosystem supports innovation like no other. Strong biotech startups, academic research, and funding pipelines are working together to accelerate progress.
Key growth drivers include:
- Rising investment in advanced biologics and gene-editing technologies
- Increasing number of patients with chronic and genetic diseases
- Strong regulatory support through faster approval pathways
- Continuous breakthroughs in viral vectors and cell engineering
This combination creates a high-momentum environment where ideas quickly turn into clinical trials.
Clinical Trials: Where Science Meets Reality
Clinical trials are the backbone of this market. They test whether new therapies actually work—and whether they are safe.
In the CGT space, trials are more complex than traditional drug testing. They involve living cells, genetic material, and long-term monitoring.
Among all phases, Phase III trials dominate the market. These trials require large patient groups and longer timelines, making them expensive but essential.
They act as the final checkpoint before therapies move toward regulatory approval and commercialization.
Early-Stage Trials Are Gaining Speed
While Phase III holds the largest share, Phase I trials are growing the fastest.
This signals a healthy pipeline. More early-stage therapies are entering the system, especially from biotech startups and research labs.
These early trials focus on safety and dosage. But behind them lies something bigger—a wave of innovation that will define the next decade of medicine.
New ideas are not just coming in—they are coming in faster than ever.
Oncology Leads, But Not Alone
Cancer remains the biggest focus area in CGT clinical trials.
The reason is clear. Cancer cases continue to rise, and traditional treatments often fall short. Therapies like CAR-T cells and gene editing have shown promising results, pushing oncology to the front of research efforts.
However, another area is quietly gaining attention—cardiology.
Heart diseases are increasing globally, and current treatments do not always offer long-term solutions. Researchers are now exploring gene therapies for inherited cardiac conditions and regenerative cell therapies for heart failure.
This shift indicates that CGT is moving beyond oncology into broader therapeutic areas.
AI Is Changing How Trials Work
Artificial Intelligence is becoming a critical tool in this market.
It is not just speeding things up—it is changing how trials are designed and executed.
AI helps researchers identify the right patients, predict treatment outcomes, and monitor safety in real time. It reduces guesswork and improves precision.
Here’s how AI is making an impact:
- Faster patient recruitment through data-driven matching
- Better trial design with predictive analytics
- Real-time monitoring of patient responses and safety
- Improved success rates through biomarker identification
This integration of AI is turning clinical trials into smarter, more efficient systems.
Investment Is Fueling the Momentum
Money is flowing into this space—and it is not slowing down.
Large pharmaceutical companies are actively acquiring smaller biotech firms to strengthen their pipelines. At the same time, startups are raising significant funding to push therapies into mid and late-stage trials.
These investments are not random. They reflect confidence in the long-term potential of CGT.
When capital, innovation, and demand align, markets tend to grow rapidly—and that is exactly what is happening here.
The Role of Regulation and Collaboration
Regulatory frameworks in the U.S. play a crucial role in this market’s growth.
Agencies are offering faster pathways for therapies that address serious or life-threatening conditions. This reduces the time it takes for new treatments to reach patients.
Collaboration is equally important. Biotech firms, academic institutions, and pharmaceutical companies are working together more than ever before.
This shared approach helps reduce risk, improve research quality, and accelerate development timelines.
Challenges Still Exist
Despite strong growth, the market faces several hurdles.
Clinical trials in CGT are expensive and time-consuming. Manufacturing processes are complex, and supply chains require precision.
Patient recruitment can also be difficult, especially for rare diseases where eligible participants are limited.
There are also safety concerns. Because these therapies involve genetic modification, even small risks can have significant consequences.
Balancing innovation with safety remains a key challenge for the industry.
A Patient-Centric Shift in Trials
One noticeable change in this market is the focus on patient experience.
CGT trials often require long-term follow-up and complex procedures. To address this, companies are offering better support systems.
Patients now receive help with travel, scheduling, and understanding treatment protocols.
This shift reflects a broader trend—clinical trials are no longer just about data. They are about people.
The future of the U.S. cell and gene therapy clinical trials market looks promising, but also demanding.
As more therapies enter the pipeline, competition will increase. Only the most effective and safe treatments will succeed.
At the same time, advancements in technology, AI, and genetic research will continue to push boundaries.
The next decade could bring breakthroughs that were once considered impossible—curing genetic disorders, reversing chronic diseases, and redefining treatment standards.
Access our exclusive, data-rich dashboard dedicated to the healthcare market – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout the Market Report Now at: https://www.towardshealthcare.com/checkout/6607
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium