
The U.S. cell therapy landscape is standing at an important inflection point. For nearly three decades, the scientific community has envisioned a future where living cells could be engineered, expanded, and re-infused to repair organs, reverse degenerative processes, and even eradicate aggressive cancers that resist conventional therapies. That future is no longer theoretical.
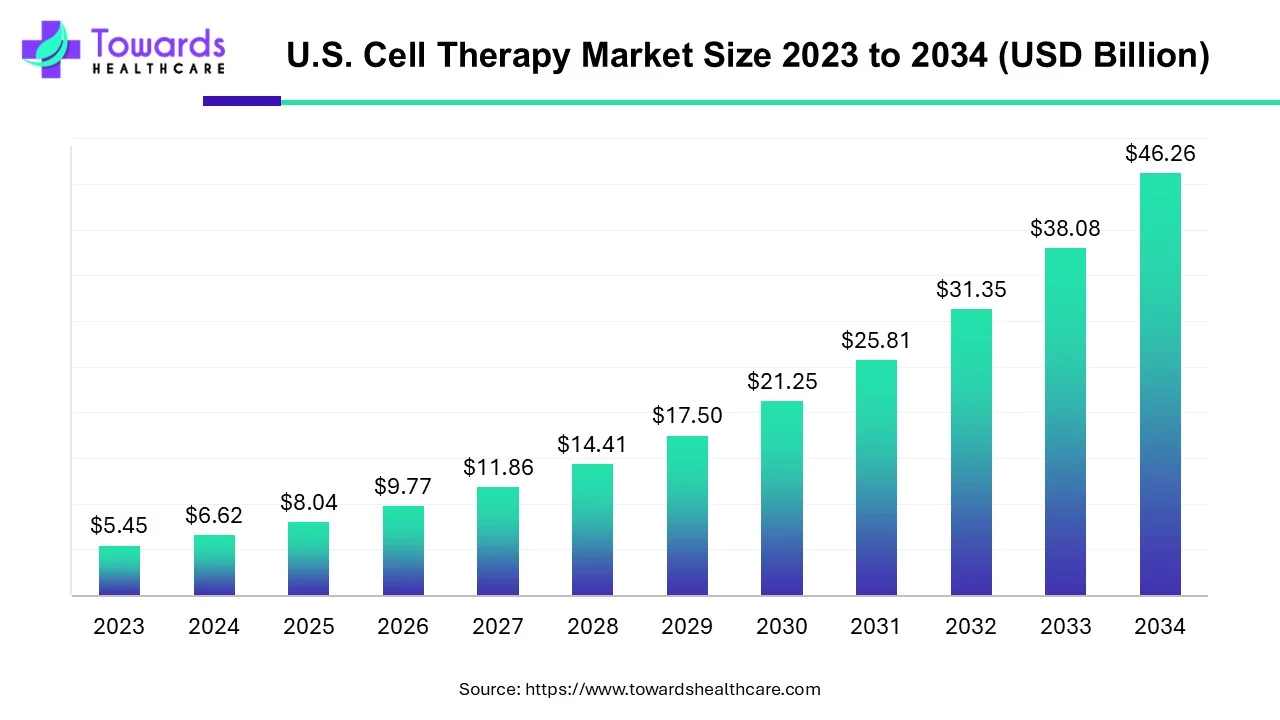
As the U.S. cell therapy market accelerates from USD 8.04 billion in 2025 to an expected USD 46.26 billion by 2034, growing at a striking CAGR of 21.46%, the question is no longer whether cell therapy will reshape modern medicine, but how quickly and at what scale it will transform clinical care.
Download Free Sample of this Insightful Report: https://www.towardshealthcare.com/download-sample/5159
The momentum is unmistakable. From oncology and neurology to autoimmune disorders and tissue repair, cell therapy is emerging as one of the most consequential innovations in today’s healthcare ecosystem. What was once a specialized niche dominated by stem-cell transplants and academic experiments has evolved into a sophisticated, regulated, and commercially relevant industry supported by advancements in biomanufacturing, genetic engineering, automation, and translational research. The U.S., with its world-leading research institutions, biopharma giants, and rapidly growing patient demand, continues to serve as the epicenter of this new era.
Yet, as promising as this revolution appears, it comes with scientific, economic, and infrastructural challenges that require careful reflection. Market expansion is impressive, but sustainable clinical integration depends on affordability, regulatory adaptability, safety, and continued investment in manufacturing innovations.
This article explores the evolving opportunities and complexities of the U.S. cell therapy market, combining market insights, recent breakthroughs, scientific advancements, and industry dynamics to present a full picture of where the field is heading.
The Rapid Expansion of Cell Therapy: A Market Ready for Scale
Cell therapy is no longer confined to bone marrow transplantation. Today, it encompasses a wide spectrum of autologous and allogeneic platforms, including CAR-T cells, MSC-based therapies, hematopoietic stem cell treatments, immune-cell engineering, and regenerative technologies. The U.S. market is benefiting from both increasing clinical validation and expanding patient demand.
The oncology segment remains the dominant therapeutic area in the U.S. This isn’t surprising, blood cancers such as multiple myeloma, lymphoma, and leukemia have shown some of the most compelling responses to CAR-T therapies in history. Autologous therapy led the market in 2023 due to personalized and lower-rejection profiles, while allogeneic treatments, often described as the “holy grail” of cell therapy, are progressing rapidly and expected to grow at the fastest rate over the coming decade.
This dual-path evolution, personalized autologous interventions alongside off-the-shelf allogeneic products, signals a shift toward treatment models that balance personalization with scalability.
Scientific Developments Pushing the Boundaries of What’s Possible
Over the past two years, scientific advancements have accelerated at a pace rarely seen in the field of cellular medicine.
In April 2025 alone, multiple high-impact breakthroughs were reported:
• Stem cell therapies for Parkinson’s disease demonstrated safety and therapeutic promise in two clinical trials using cells derived from human embryonic and induced pluripotent stem cells.
• Researchers at the University of California, Irvine developed lab-grown immune cells capable of clearing toxic brain buildup and restoring memory in mice, a breakthrough with significant implications for Alzheimer’s and other neurodegenerative disorders.
• Investigators at MD Anderson Cancer Center showcased encouraging safety profiles for AIC100, a next-generation CAR-T therapy targeting advanced thyroid cancer.
• A collaboration between the Sant Pau Research Institute, Sant Pau Hospital, and the Josep Carreras Leukaemia Research Institute reported strong early-stage efficacy for a CD30-targeting CAR-T therapy, published in Blood.
• USC researchers announced the creation of EchoBack CAR-T cells, designed to activate only around tumor cells using focused ultrasound, minimizing collateral damage to healthy tissue.
These developments reveal two important trends shaping the future of cell therapy:
-
The technology is expanding beyond oncology into neurology, autoimmune conditions, metabolic disorders, and regenerative applications.
-
Engineering sophistication is increasing, moving from simple antigen targeting to tissue-specific activation, memory phenotype enhancement, next-gen co-stimulatory domains, and improved safety control.
The field is no longer about simply engineering cells, it’s about building intelligent therapeutic systems inside the human body.
A Market Driven by the Burden of Complex Diseases
The rise in chronic and life-threatening diseases is one of the most significant demand drivers behind the U.S. cell therapy market.
Cardiovascular disease continues to be the leading cause of death in the country, claiming one life every 34 seconds. Neurodegenerative diseases such as Alzheimer’s and Parkinson’s affect millions of Americans. Autoimmune disorders impact almost 50 million individuals, 80% of whom are women. These diseases require treatment options that go beyond symptom management to target underlying cellular and molecular dysfunctions.
Cell therapies offer the ability to regenerate tissue, modulate immune responses, target malignant cells, and potentially reverse conditions previously considered irreversible. This unmatched therapeutic potential is driving both investment and patient interest.
The High Cost Barrier: A Persistent Reality
Despite extraordinary breakthroughs, cost remains one of the biggest constraints in the U.S. cell therapy market. Many current therapies exceed USD 400,000 per patient, while some cross the million-dollar mark. These price points challenge the sustainability of widespread adoption.
The core reasons are well-known to industry insiders:
• labor-intensive cell collection
• complex genetic engineering processes
• reliance on viral vectors
• stringent regulatory manufacturing conditions
• limited automation
• individualized manufacturing for autologous therapies
If the U.S. wants cell therapy to move from specialized cancer centers to mainstream clinical practice, manufacturing scalability and cost reduction must advance in parallel with scientific innovation.
Fortunately, this is beginning to happen.
With the launch of next-generation platforms such as Cytiva’s Sefia manufacturing suite, and increasing collaboration between therapy developers and manufacturing technology companies, the industry is clearly shifting toward streamlined, modular, and more standardized production models.
Autologous vs. Allogeneic: A Dual-Track Growth Path
Autologous therapies continue to lead in clinical adoption due to their lower risk of immune rejection. Cancer centers across the U.S. are expanding autologous CAR-T and stem-cell capabilities, with recent milestones such as City of Hope Atlanta introducing its autologous transplant program.
But the industry is also preparing for a transition. Allogeneic therapies, off-the-shelf solutions manufactured from donor cells, offer scalability, lower cost of goods, and reduced treatment preparation timelines. These approaches could eventually democratize access and relieve capacity bottlenecks.
Examples of progress include:
• VetStem introducing the first FDA-reviewed off-the-shelf allogeneic PRP product for veterinary use, signaling steps toward broader translational applications.
• Companies such as Atara Biotherapeutics advancing an allogeneic EBV T-cell platform, with multiple programs including ATA3219, ATA3431, and ATA188.
• Gilead/Kite’s extensive allogeneic CAR-T pipeline in early- and late-stage trials.
Both therapy types will coexist for the foreseeable future, each addressing different clinical and economic needs.
Oncology: The Flagship Segment of U.S. Cell Therapy
Cancer remains the backbone of the cell therapy market. The remarkable success of CAR-T therapies, starting with the approvals of Abecma, Yescarta, Tecartus, and Carvykti, has emboldened clinicians and researchers to push boundaries.
Cell therapies are being developed not only to treat blood cancers but also to support post-treatment recovery, enhance immune function, and deliver targeted payloads directly to tumors. HDR-based approaches, MSC carriers, and immune-effector engineering are rapidly expanding the oncology pipeline.
The FDA has also played a role by approving increasingly innovative therapeutic designs, such as J&J’s Carvykti for multiple myeloma, strengthening confidence in next-generation oncology programs.
As cancer prevalence continues to rise across the U.S., demand for advanced cell-based interventions will only accelerate.
Innovation Across the Ecosystem: Academia, Pharma, and Startups Moving in Sync
One of the most defining elements of the U.S. cell therapy landscape is collaboration. Academic centers, biotech startups, and large pharmaceutical companies are working closer than ever to translate discoveries into real-world treatments.
Recent examples include:
• Seattle Children’s receiving FDA approval to begin the first CAR-T trial for pediatric lupus.
• Walgreens expanding its specialty pharmacy model to include services for gene and cell therapy.
• The UC San Diego Alpha Stem Cell Clinic receiving a major state-funded investment to advance stem-cell-based treatments.
Industry leaders such as Atara, Gilead, J&J, Nkarta, Vertex, and others continue to push the pipeline forward with platforms spanning EBV-targeted therapies, NK-cell engineering, and multi-antigen CAR designs.
This interconnected ecosystem, research, clinical capacity, manufacturing innovation, and commercial experience, is one of the U.S.’s greatest competitive advantages.
Where the Market Is Heading: The Next Decade of U.S. Cell Therapy
The next decade will likely be defined by five major shifts:
1. Allogeneic therapies becoming mainstream
Large-scale, off-the-shelf treatments will drastically reduce turnaround times and costs.
2. Automation and AI-driven manufacturing
From closed-loop bioreactors to digital twins, automated cell expansion will become the industry standard.
3. Expansion into non-oncology indications
Neurodegeneration, autoimmune disorders, cardiovascular repair, metabolic diseases, and tissue regeneration will unlock large patient populations.
4. Combination therapies with gene editing, biologics, and small molecules
Synergy-based treatment models will emerge, especially in rare diseases and complex cancers.
5. Greater emphasis on long-term safety controls
Suicide switches, controllable activation systems, and ultrasound-guided CAR-Ts will shape next-generation design.
The U.S. is poised to remain the global leader in cell therapy innovation, but this leadership will depend on addressing cost, capacity, and equitable access.
Access our exclusive, data-rich dashboard dedicated to the therapeutic area sector – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout U.S. Cell Therapy Market Report Now at: https://www.towardshealthcare.com/checkout/5159
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest




