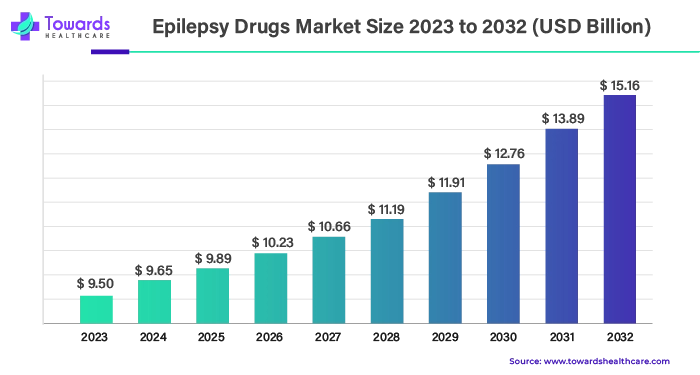
The epilepsy drugs market size attained a commendable value of USD 9.50 billion in the year 2023, reflecting the escalating demand and investments in treatment solutions. As we stride into the future, projections unveil an even more promising outlook, with an anticipated surge to USD 15.16 billion by the year 2032. This trajectory is underscored by a projected compound annual growth rate (CAGR) of 5.1% during the period spanning 2024 to 2032.
Factors Driving Epilepsy Drugs Market Size Growth
The exponential growth of the epilepsy drugs market can be attributed to multifaceted factors, including:
- Technological Advancements: Breakthroughs in drug delivery systems and formulation techniques have bolstered the efficacy and safety profiles of epilepsy medications, fostering patient compliance and treatment outcomes.
- Rising Disease Burden: The escalating prevalence of epilepsy, coupled with a growing awareness regarding the importance of early diagnosis and intervention, has fueled the demand for innovative therapeutic solutions.
- Strategic Collaborations and Partnerships: Collaborative initiatives between pharmaceutical companies, research institutions, and healthcare organizations have catalyzed research endeavors, leading to the development of novel drug candidates and treatment modalities.
- Regulatory Support and Market Access: Favorable regulatory frameworks and expedited approval processes have facilitated market entry for new drugs, thereby augmenting market competitiveness and accessibility.
Download a short version of this report @ https://www.towardshealthcare.com/personalized-scope/5144
Exploring Therapeutic Landscape
Classification of Epilepsy Drugs
Epilepsy drugs encompass a diverse array of pharmacological agents classified based on their mechanism of action and therapeutic efficacy. These include:
- First-generation Antiepileptic Drugs (AEDs): Traditional AEDs such as phenytoin, carbamazepine, and valproate constitute the cornerstone of epilepsy treatment, exerting their effects through modulation of neuronal excitability and neurotransmitter balance.
- Second-generation AEDs: With the advent of second-generation AEDs including levetiracetam, lamotrigine, and oxcarbazepine, clinicians have gained access to a broader armamentarium of treatment options characterized by improved tolerability and reduced side effects.
Emerging Trends and Innovations
The evolving landscape of epilepsy therapeutics is marked by several notable trends and innovations, including:
- Precision Medicine Approaches: Advances in pharmacogenomics and biomarker discovery are paving the way for personalized treatment regimens tailored to individual patient profiles, optimizing therapeutic outcomes and minimizing adverse effects.
- Gene Therapy and Novel Drug Targets: Exploration of novel drug targets and gene therapy strategies holds immense promise for refractory epilepsy cases, offering potential avenues for disease modification and seizure control.
Navigating Market Challenges
Despite the remarkable growth prospects, the epilepsy drugs market is not devoid of challenges, including:
- Generic Erosion and Pricing Pressures: The impending patent expirations of key epilepsy drugs pose significant challenges to market incumbents, necessitating strategic maneuvers to mitigate the impact of generic erosion and pricing pressures.
- Treatment Gaps and Access Disparities: Disparities in access to epilepsy care, particularly in low- and middle-income countries, underscore the need for concerted efforts to address treatment gaps and enhance healthcare infrastructure.
Future Outlook and Opportunities
Looking ahead, the epilepsy drugs market is poised for sustained growth and innovation, driven by:
- Advancements in Drug Delivery Technologies: Innovations in drug delivery systems, including extended-release formulations and implantable devices, hold promise for optimizing treatment adherence and therapeutic outcomes.
- Integration of Digital Health Solutions: The integration of digital health technologies, such as telemedicine platforms and wearable devices, is poised to revolutionize epilepsy management, enabling real-time monitoring and personalized interventions.
- Collaborative Research Initiatives: Continued collaboration between academia, industry, and regulatory bodies is essential for driving forward the frontiers of epilepsy research and drug development, ultimately benefiting patients worldwide.
According to the World Health Organization, around 50 million individuals in the United States are affected by epilepsy as of February 2024, and they face a heightened risk of premature death.
Epilepsy is a neurological condition characterized by recurrent seizures, which occur due to sudden bursts of electrical activity in the brain, temporarily disrupting normal function. Symptoms can vary widely, ranging from mild to severe manifestations. Seizures may present as convulsions, muscle spasms, loss of consciousness, blank staring, or unusual movements. Following a seizure, individuals may experience confusion, memory lapses, or difficulties with speech. Some seizures may involve brief disconnection from surroundings or psychic symptoms such as fear or déjà vu beforehand. Treatment of epilepsy is crucial for several reasons. It aims to decrease the frequency and severity of seizures, thereby improving the quality of life for affected individuals. Safety is a significant concern, as seizures can lead to accidents during activities like driving or swimming.
Epilepsy can be likened to an electrical storm in the brain, resulting in sudden bursts of activity leading to seizures. These seizures can manifest in various ways, from uncontrollable muscle jerking to periods of lost awareness where individuals appear temporarily disconnected.
Why is it imperative to treat epilepsy? Well, untreated seizures can profoundly disrupt a person’s life. They can occur unexpectedly, putting individuals at risk of accidents or injuries. Moreover, they can complicate everyday tasks like driving or working. However, the importance of epilepsy treatment extends beyond safety concerns. Consider living in constant apprehension of when the next seizure might occur—an emotionally and mentally draining experience. Treatment seeks to reduce the frequency and intensity of seizures, granting individuals more control and peace of mind.
Globally, approximately 10.1 million people with epilepsy could benefit from surgical treatment, with around 1.4 million new cases eligible for surgical intervention annually.
Uncontrolled seizures can detrimentally impact brain function over time, affecting memory, concentration, and mood. It’s akin to experiencing a malfunction in the brain’s system that worsens without intervention. Additionally, there are social ramifications to consider. Imagine feeling embarrassed or anxious about going out in public due to the fear of experiencing a seizure in front of others. Such concerns can hinder the formation and maintenance of relationships.
Treating epilepsy isn’t solely about stopping seizures; it’s about reinstating safety, control, and quality of life. It’s about affording individuals with epilepsy the opportunity to live their lives fully, free from the constant fear and uncertainty that seizures entail.
Overview of Epilepsy Drug Market Dynamics
Epilepsy, a neurological disorder characterized by recurrent seizures, affects millions of individuals worldwide. The demand for epilepsy drugs is substantial, particularly in regions like North America and Asia, where healthcare infrastructure, prevalence rates, and regulatory frameworks significantly impact market dynamics.
North America: A Key Market for Epilepsy Drugs
Healthcare Spending and Infrastructure
North America stands out as a significant market for epilepsy drugs due to its robust healthcare spending and well-established infrastructure. The region comprises the United States and Canada, both of which prioritize healthcare expenditure to ensure quality medical services for their citizens.
Stringent Regulatory Standards
The approval and sale of epilepsy drugs in North America are subject to rigorous scrutiny by regulatory bodies such as the U.S. Food and Drug Administration (FDA) and Health Canada. These agencies evaluate the safety, efficacy, and quality of drugs before granting approval, instilling confidence in patients and healthcare providers.
Insurance Coverage Influence
Insurance coverage plays a pivotal role in facilitating access to epilepsy drugs in North America. Whether through private insurance plans or government-funded schemes, comprehensive coverage enables patients to afford necessary medications. The extent of coverage often influences prescribing patterns and market dynamics, shaping the landscape for epilepsy drug manufacturers.
Factors Driving Demand in Asia
Rapid Economic Growth
Many countries in Asia have experienced rapid economic growth, leading to increased spending on healthcare infrastructure and services. As a result, more individuals have access to medical assistance, including treatment for neurological disorders like epilepsy.
Diverse Regulatory Landscape
The regulatory landscape for epilepsy drugs varies across different Asian countries, posing challenges for manufacturers seeking approval and market entry. Varied regulations pertaining to drug approval, pricing mechanisms, and intellectual property rights necessitate careful navigation and adaptation strategies.
Cultural Perceptions and Awareness
Cultural attitudes and perceptions towards epilepsy vary widely across Asia, influencing individuals’ willingness to seek treatment and adhere to medication regimens. Addressing cultural stigmas and raising awareness about epilepsy are crucial steps in ensuring equitable access to treatment across diverse populations.
Strategies for Market Penetration and Education
Tailored Marketing and Distribution
Effective market penetration in North America and Asia requires tailored marketing strategies that account for regional differences in healthcare systems, regulatory frameworks, and patient preferences. Collaborating with local stakeholders and leveraging digital platforms can enhance visibility and accessibility of epilepsy drugs.
Educational Initiatives
Education plays a pivotal role in destigmatizing epilepsy and promoting early intervention and treatment-seeking behaviors. Comprehensive educational initiatives targeting healthcare professionals, patients, and communities can improve awareness, diagnosis rates, and treatment outcomes.
Recent Developments
- In January 2023, Zydus Lifesciences introduced Topiramate extended-release capsules, a generic epilepsy treatment, in the US market.
- In March 2022, the FDA approved Ztamy (ganaxolone) to treat seizures in patients aged 2 and up with CDKL5 deficiency disorder (CDD). CDD is an exceptional developmental epileptic caused encephalopathy by CDKL5 gene mutations.
Epilepsy Drugs Market Players
- Sanofi
- Pfizer, Inc.
- UCB S.A
- Belgium
- Otsuka America Pharmaceutical, Inc.
- Novartis AG
- GlaxoSmithKline plc.
- Abbott Laboratories, Inc.
- Neurelis Inc.
- Jazz Pharmaceuticals plc.
Epilepsy Drugs Market Segments
By Product Type
- First Generation Anti-epileptics
- Second Generation Anti-epileptics
- Third Generation Anti-epileptics
By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
By Geography
- North America
- Europe
- Asia Pacific
- Middle East and Africa
- South America
To own our research study instantly, Click here @ https://www.towardshealthcare.com/price/5144
Read More Epilepsy Drugs Market:
Unlock Infinite Advantages: Subscribe to Annual Membership
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
About Us
Healthcare Web Wire is a premier subsidiary of Towards Healthcare, dedicated to providing comprehensive insights and information related to the healthcare industry. With a commitment to delivering accurate and timely updates, Healthcare Web Wire serves as a vital resource for professionals, enthusiasts, and stakeholders within the healthcare sector. Our platform serves as a central hub for the latest news, trends and developments shaping the healthcare landscape. Join us on Healthcare Web Wire and become part of a vibrant community dedicated to advancing healthcare knowledge and shaping the future of healthcare worldwide.
Explore the comprehensive statistics and insights on healthcare industry data and its associated segmentation: Get a Subscription
For Latest Update Follow Us: https://www.linkedin.com/company/towards-healthcare