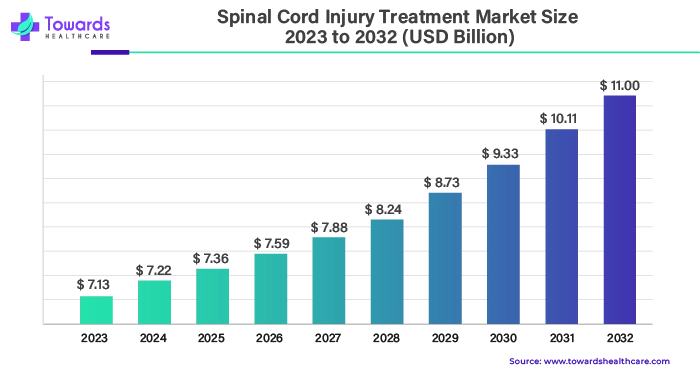
The spinal cord injury treatment market has been on a remarkable growth trajectory, a trend that is expected to continue over the coming decade. In 2023, the market reached a value of USD 7.13 billion, and projections indicate that by 2033, it is poised to reach USD 11.00 billion. This signifies a compound annual growth rate (CAGR) of 4.8% from 2024 to 2033. In this article, we delve into the factors driving this growth, the key players in the market, and the emerging trends that are shaping its future.
Factors Fueling Growth
The growth of the spinal cord injury treatment market can be attributed to several factors, including:
- Technological Advancements: Rapid advancements in medical technology have led to the development of innovative treatment options for spinal cord injuries. These include stem cell therapy, nerve stimulation techniques, and advanced surgical procedures.
- Increasing Incidence of Spinal Cord Injuries: With the rise in road accidents, sports injuries, and falls, there has been a surge in the number of spinal cord injury cases globally. This has created a growing demand for effective treatment solutions.
- Growing Aging Population: The aging population is more prone to spinal cord injuries due to age-related degeneration. As the elderly population grows, the demand for spinal cord injury treatment is expected to rise significantly.
Download a short version of this report @ https://www.towardshealthcare.com/personalized-scope/5143
Each year, approximately 18,000 new spinal cord injuries are reported in the United States. This figure is derived from an annual incidence rate of 54 per 1 million. The significant number of people living with spinal cord injuries underscores the ongoing need for prevention and treatment efforts.
A spinal cord injury occurs when the bundle of nerves within the spine is damaged, resulting in changes in sensation, movement, and strength below the injury site. Causes can range from direct trauma to the spinal cord itself to damage to the surrounding bones. Symptoms vary depending on the severity and location of the injury. Severe injuries higher up on the spine can lead to paralysis in the limbs and affect most of the body, while lower injuries may primarily impact the legs.
Symptoms of a spinal cord injury include numbness, tingling, or loss of sensation in the hands and feet, along with paralysis, pain, or pressure in the head, neck, or back. Weakness, difficulty walking, breathing, or changes in sexual function can also occur. The leading causes of spinal cord injuries in the US are motor vehicle accidents and serious falls, followed by violence, sports injuries, medical mishaps, and other accidents. Risk factors include age, alcohol use, certain diseases, and failure to use appropriate safety gear such as seat belts or sports equipment.
In the United States, motor vehicle accidents and serious falls are the primary causes of spinal cord injuries. Other causes include violence (such as gunshot wounds and assaults), sports injuries, industrial accidents, and diseases. Age is a significant risk factor, with individuals between 16 and 30 years old being at higher risk, as well as those over 65 for dangerous falls. Alcohol use, certain medical conditions, and failure to use appropriate safety equipment like seat belts or protective gear also increase the risk of spinal cord injuries.
Recent Advances in Medical Collaborations and FDA Designations
- In July 2023, NeuroSolv Therapeutics and AscellaHealth LLC announced a partnership to advance clinical trial development and expedite regulatory approval for Perineline.
- NeuroSolv Therapeutics, an ethical business headquartered in Belfast, Northern Ireland, and Baltimore, United States, joined forces with AscellaHealth LLC, a global healthcare and specialty pharmacy solutions company. Their collaboration aims to maximize the potential of bringing Perineline to market.
- In July 2021, the United States Food and Drug Administration (FDA) granted fast-track designation to Mitsubishi Tanabe Pharma Corporation’s MT-3921 for the treatment of spinal cord injuries.
- In September 2020, AbbVie announced that elezanumab (ABT-555), an investigational treatment for spinal cord injury, received Orphan Drug and Fast Track designations from the FDA.
Spinal Cord Injury Treatment Market Segments
By Injury Type
- Complete Spinal Cord Injuries
- Partial Spinal Cord Injuries
By Treatment
- Corticosteroid
- Surgery
- Spinal Traction
- Others
By End User
- Hospital
- Trauma Centers
- Ambulatory Surgical Centers
- Others
By Geography
- North America
- Europe
- Asia Pacific
- Middle East and Africa
- South America
To own our research study instantly, Click here @ https://www.towardshealthcare.com/price/5143
Read more about Spinal Cord Injury Treatment Market:
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
About Us
Healthcare Web Wire is a premier subsidiary of Towards Healthcare, dedicated to providing comprehensive insights and information related to the healthcare industry. With a commitment to delivering accurate and timely updates, Healthcare Web Wire serves as a vital resource for professionals, enthusiasts, and stakeholders within the healthcare sector. Our platform serves as a central hub for the latest news, trends and developments shaping the healthcare landscape. Join us on Healthcare Web Wire and become part of a vibrant community dedicated to advancing healthcare knowledge and shaping the future of healthcare worldwide.
Explore the comprehensive statistics and insights on healthcare industry data and its associated segmentation: Get a Subscription
For Latest Update Follow Us: https://www.linkedin.com/company/towards-healthcare