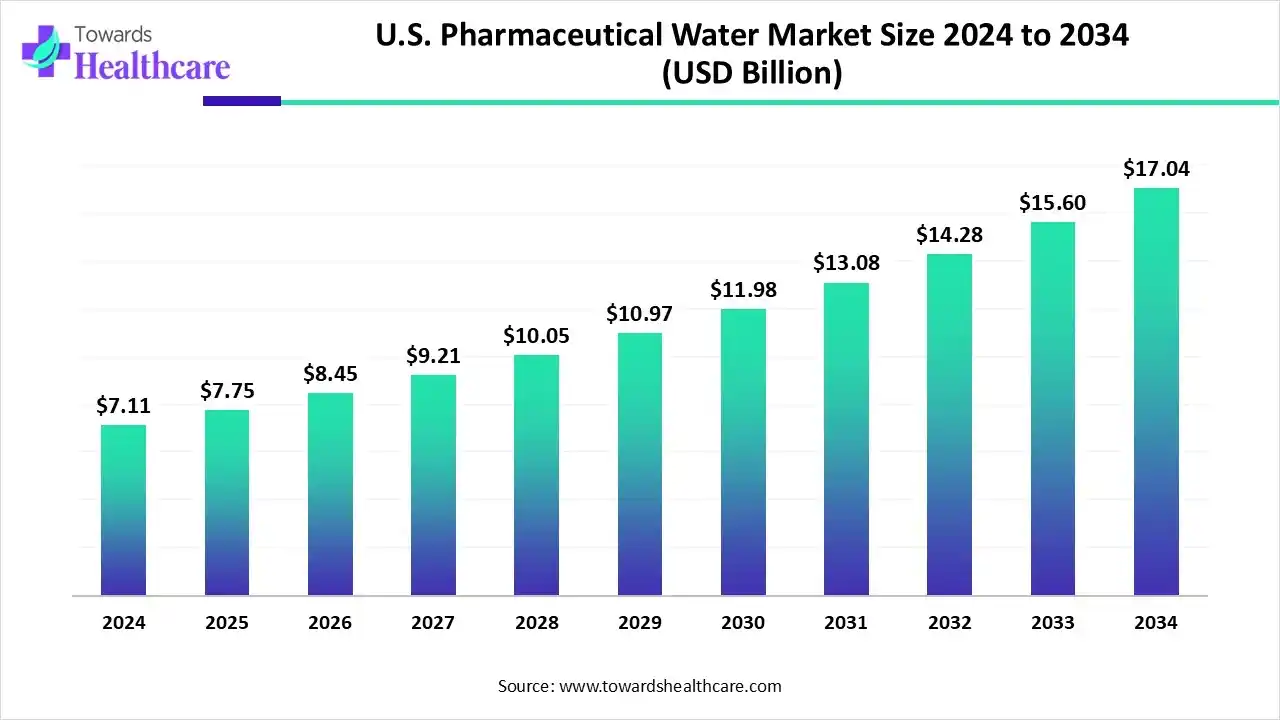
The U.S. pharmaceutical water market, valued at US$ 7.11 billion in 2024, is projected to reach US$ 17.04 billion by 2034 at a CAGR of 8.94%, driven by rising demand for high-purity water in biologics, injectables, and stringent regulatory compliance.
Download this Free Sample Now and Get the Complete Report and Insights of this Market Easily @ https://www.towardshealthcare.com/download-sample/6238
Market Size
Market Value & Growth
●2024: US$ 7.11 billion
●2025: US$ 7.75 billion
●2034 (Projected): US$ 17.04 billion
●CAGR (2025–2034): 8.94%
Regional Revenue Share (U.S.)
●Northeast/Mid-Atlantic: 55–60% revenue share in 2024.
●Southeast & Midwest: Fastest-growing regions due to facility expansion, regulatory support, and biotech investments.
Global Context
●Global pharmaceutical water market (2023): US$ 38.7 billion
●Expected global value by 2034: US$ 107.15 billion, CAGR: 9.25%
Segment-wise Market Size (Water Type / Technology / End Users / Solutions)
●Water Type/Product Type: WFI holds 79% revenue share, HPW/HPLC-grade water fastest-growing.
●Technology: RO + EDI dominates, Continuous monitoring & automation/PAT sensors fastest-growing.
●End User: Pharmaceutical & biotech companies lead with 58% revenue, CDMOs fastest-growing.
●Business Model: On-site water generation leads with 34.6% share, Water-as-a-Service growing rapidly.
Market Trends
Inorganic Growth Strategies
●Companies adopt acquisitions, strategic partnerships to expand product portfolios.
●Example: Cipla’s acquisition of InvaGen Pharmaceuticals (2025) increased demand for WFI systems.
Investment & Funding
●Focus on advanced purification, R&D, and facility expansion.
●Example: Eli Lilly invested US$ 6.5 billion in a biomanufacturing facility in Houston for high-purity water-dependent biologics production.
Adoption of Advanced Purification Technologies
●Reverse Osmosis (RO), Electrodeionization (EDI), Ultrafiltration, UV sterilization.
●Improves reliability, water quality, and regulatory compliance.
Sustainability Initiatives
●Efficient water use, recycling, and environmental compliance.
●Example: Sanofi’s wastewater treatment and reuse project won ISPE award in 2025.
Expansion of Biologics & Injectables Production
●Rising demand for sterile water in vaccines and biologics fuels growth.
Regional Facility Growth
●Southeast & Midwest regions expanding due to favorable business and regulatory environment.
Automation & Digitalization
●Adoption of continuous monitoring and PAT sensors for water systems.
Rising Analytical & QC Needs
●HPW/HPLC water demand driven by lab and analytical applications.
Outsourcing to CDMOs/CMOs
●Increasing need for high-purity water in outsourced drug manufacturing.
System Integration and Flexibility
●On-site and packaged water solutions allow scalable operations across pharma and biotech.
AI impacts/roles
Real-Time Monitoring
Function: AI continuously analyzes data from sensors monitoring conductivity, Total Organic Carbon (TOC), microbial load, and endotoxin levels.
Impact:
●Immediate detection of deviations in water purity prevents production of non-compliant batches.
●Ensures continuous compliance with FDA and USP standards for WFI and HPW.
Market Relevance: Supports fast-growing aseptic fill and biologics production, where real-time purity is critical.
Predictive Maintenance
Function: AI forecasts failures or wear in RO + EDI units, distillation systems, and filtration assemblies based on historical performance and operational parameters.
Impact:
●Reduces unplanned downtime, minimizing disruption in water supply for drug manufacturing.
●Extends equipment lifespan, optimizing CAPEX and OPEX for pharma and biotech facilities.
Example: Facilities in Northeast/Mid-Atlantic with high-volume WFI demand benefit from predictive scheduling of maintenance.
Process Optimization
Function: Machine learning algorithms dynamically adjust purification process parameters to maximize water yield and quality while minimizing energy and chemical usage.
Impact:
●Improves efficiency of energy-intensive RO + EDI and distillation operations.
●Reduces operational costs and environmental footprint, aligning with sustainability initiatives.
Market Relevance: Essential for large-scale biologics and injectable production facilities in both Northeast and emerging Southeast/Midwest hubs.
Contamination Prevention
Function: AI detects early signs of microbial growth, chemical contamination, or system deviations before water quality drops below standards.
Impact:
●Maintains sterility for WFI and HPW used in injectable drugs, vaccines, and biologics.
●Reduces batch rejection and ensures patient safety.
Example: Continuous PAT sensor data processed by AI can preemptively adjust UV sterilization or filtration cycles.
Energy Efficiency
Function: Algorithms optimize energy-intensive water purification steps, such as heating in distillation or pumps in RO + EDI systems.
Impact:
●Reduces electricity and operational costs significantly.
●Aligns with regulatory and corporate sustainability goals.
Market Relevance: Supports adoption of sustainable water systems in companies like Sanofi, Eli Lilly, and emerging CDMOs.
Automated Validation & Compliance
Function: AI accelerates documentation and reporting for IQ (Installation Qualification), OQ (Operational Qualification), PQ (Performance Qualification) and regulatory audits.
Impact:
●Reduces human error in compliance reporting.
●Shortens time-to-market for biologics and sterile injectables by ensuring regulatory approval efficiency.
Market Relevance: Critical for CDMOs/CMOs managing multiple clients and batch productions.
Quality Analytics
Function: Intelligent data analysis identifies trends, correlations, and anomalies in water quality over time.
Impact:
●Ensures consistency across batches in drug substance and drug product manufacturing.
●Supports predictive quality control for aseptic filling, vaccines, and biologics.
Example: Continuous data aggregation from multiple facilities can optimize HPW and WFI consistency across sites.
Regional Insights
Northeast / Mid-Atlantic Region
Dense Pharma & Biotech Presence
●The region hosts a high concentration of pharmaceutical and biotechnology companies, including large multinational corporations and established CDMOs.
Impact:
●High demand for ultra-pure Water for Injection (WFI) and Highly Purified Water (HPW) due to extensive biologics, sterile injectable, and vaccine production.
●Encourages investment in on-site water generation systems and advanced purification technologies.
Strong R&D Ecosystem
●Presence of research institutions, academic labs, and biotech startups fosters innovation.
Impact:
●Drives the need for laboratory-grade water (HPW/HPLC) for analytical testing, drug development, and clinical research.
●Promotes adoption of continuous monitoring systems and automation/PAT sensors to ensure precision in R&D water usage.
Advanced Manufacturing Infrastructure
●Facilities are equipped with modern water treatment systems, RO + EDI, distillation, and filtration setups.
Impact:
●Ensures reliable, scalable, and compliant water supply for large-scale production.
●Supports high-volume biologics, sterile injectables, and aseptic fill operations.
Regulatory Compliance Drives Demand
●Strong FDA oversight and strict adherence to USP water standards push continuous upgrades of water systems.
Impact:
●Manufacturers invest in monitoring, validation, and system automation to avoid regulatory risks.
●Sustains consistent market leadership for suppliers of advanced water purification and monitoring technologies.
Southeast & Midwest Regions
Fastest-Growing Regions
Emerging hubs for pharmaceutical manufacturing, with significant investments in new facilities and expansions.
Impact:
●Rapid adoption of WFI, HPW, and on-site water systems to meet production demands.
●Opportunity for suppliers to deploy turnkey water systems and Water-as-a-Service models for new entrants.
Favorable Regulatory & Business Environment
●States in these regions offer tax incentives, grants, and business-friendly regulations for pharma and biotech companies.
Impact:
●Attracts CDMOs and emerging biotech firms.
●Drives adoption of advanced purification systems, including RO + EDI and automated monitoring solutions.
Focus on Biologics, Vaccines, and Injectables
●Increasing production of biologics and sterile injectables requires high-purity water at scale.
Impact:
●Accelerates installation of on-site water generation systems, WFI distillation units, and HPW pipelines.
●Encourages investment in AI-driven water quality monitoring for continuous compliance.
Significance: These regions are becoming strategic hubs for expansion of U.S. pharmaceutical water suppliers due to growth potential.
Market Dynamics
Drivers
Advanced Purification Technologies
Technologies: Reverse Osmosis (RO), Electrodeionization (EDI), Ultrafiltration, UV sterilization, Ozone treatment.
Impact:
●Ensures consistent production of ultra-pure water for WFI, HPW, and lab applications.
●Reduces microbial contamination and endotoxin presence in injectable drug production.
Benefit to Industry:
●Enhances reliability and operational efficiency.
●Supports compliance with USP and FDA standards.
●Enables scalable and flexible manufacturing across pharmaceutical and biotech companies.
Rising Biologics, Vaccines, and Sterile Injectable Production
Trend: Expansion in biologics, vaccines, and sterile injectables increases high-purity water demand.
Reason: Biologics and vaccines require Water for Injection (WFI) due to sterility and endotoxin-free requirements.
Market Effect:
●Drives investment in advanced water systems.
●Accelerates adoption of on-site water generation and monitoring solutions.
Example: Eli Lilly’s US$6.5 billion biomanufacturing facility in Houston (2025) increases WFI system demand.
Regulatory Compliance (FDA, USP Standards)
Role: Ensures all water used in drug manufacturing meets strict quality, sterility, and safety standards.
Impact:
●Compels pharmaceutical companies and CDMOs to adopt advanced purification and monitoring systems.
●Increases demand for automated validation, documentation, and continuous quality monitoring.
Example: Continuous monitoring & automation / PAT sensors segment is growing fastest due to regulatory adherence needs.
Restraints of the U.S. Pharmaceutical Water Market
Energy-Intensive Purification Processes
Issue: WFI production and purification systems consume high amounts of energy.
Impact:
●Operational costs increase significantly for large-scale pharmaceutical manufacturing.
●RO + EDI and distillation processes, though efficient, still require considerable power and maintenance.
Industry Challenge:
●Need to balance purity standards with sustainable and cost-effective operations.
●Small and emerging biotech companies may struggle to invest in high-capex water systems.
Opportunities in the U.S. Pharmaceutical Water Market
Sustainability Initiatives
Trend: Growing focus on efficient water use, recycling, and wastewater treatment.
Impact:
●Reduces water consumption and environmental footprint.
●Aligns with global sustainability standards and corporate social responsibility goals.
Example: Sanofi’s wastewater treatment and reuse project (2025) won ISPE Facility of the Year Award, demonstrating sustainable water management in pharma.
Benefit: Drives adoption of water recycling technologies, energy-efficient RO/EDI systems, and UV/ozone disinfection for reduced effluent discharge.
AI Integration and Automation
Trend: Incorporation of AI, machine learning, and automated monitoring systems.
Impact:
Enables predictive maintenance, reducing system downtime and operational inefficiencies.
Supports real-time quality assurance, ensuring water purity for biologics and injectables.
Reduces human error in regulatory compliance and documentation processes.
Examples of Benefits:
●AI-driven analytics predict contamination risks.
●Automated PAT sensors monitor conductivity, TOC, and microbial load continuously.
●Optimizes energy consumption in WFI/HPW production.
Top 10 Companies – Overview, Products, Strengths
Evoqua Water Technologies
●Products: WFI systems, purification equipment.
●Strength: High expertise in water treatment for pharma.
Pall / Danaher
●Products: Filters, WFI/distillation, sterilization systems.
●Strength: Global reach, strong R&D.
Merck Millipore (MilliporeSigma)
●Products: High-purity water technologies, WFI systems.
●Strength: Specialty in biologics and injectables water systems.
GE Water / SUEZ WTS
●Products: RO, EDI, purification systems.
●Strength: Advanced technology integration, large-scale deployment.
Veolia / Suez
●Products: On-site water generation, WFI systems.
●Strength: Sustainability focus, global operations.
Thermo Fisher Scientific
●Products: Lab & manufacturing water purification systems.
●Strength: Technical support, continuous monitoring solutions.
Parker Hannifin
●Products: Sterile water filtration and system automation.
●Strength: High-quality precision filtration solutions.
Grundfos
●Products: Pumping solutions for pharma water systems.
●Strength: Reliable distribution and energy efficiency.
SPX Flow (APV, WFI/distillation)
●Products: Distillation, WFI systems.
●Strength: Specialized in sterile and ultrapure water systems.
Dow (Membranes)
●Products: RO membranes and filtration technologies.
●Strength: Technological efficiency and scalability.
Latest Announcement
Veolia (May 2025):
Acquired remaining 30% stake in Water Technologies & Solutions from CDPQ for US$ 1.75 billion.
Expected cost synergies: €90 million (~$102.3 million) by 2027.
Secured US$ 750 million in new contracts, including $550 million with a U.S. Midwest microelectronics plant.
Significance: Expands market presence in energy and semiconductor water systems.
Recent Developments
MilliporeSigma (Sept 2025)
Climate-neutral manufacturing facility in the U.S.
Produces filtration devices for aseptic processing, tangential-flow, and virus filtration.
Aquatech (June 2025)
Acquisition of Century Water, Singapore-based ultrapure water solutions provider.
Focus: Advanced water manufacturing, pharma, semiconductor, and wastewater recycling.
Segments Covered
By Water Type / Product Type
Water for Injection (WFI)
Dominance: Accounts for approximately 79% revenue share in 2024.
Purpose: Used primarily for sterile injectable drug production, biologics, vaccines, and critical formulations.
Regulatory Compliance: Meets strict USP/FDA standards for endotoxin-free and sterile water.
Significance: Essential in maintaining drug safety, sterility, and overall quality in manufacturing.
Purified Water (PW)
Applications: Standard pharmaceutical operations such as cleaning, formulation, and general lab use.
Characteristics: Free from particulate matter, organics, and microbes but less stringent than WFI.
Role: Supports non-injectable formulations and general manufacturing processes.
Highly Purified Water (HPW / HPLC Grade Water)
Growth: Fastest-growing water segment due to laboratory and analytical demand.
Applications: HPLC (High-Performance Liquid Chromatography), reagent preparation, analytical testing.
Importance: Critical for accuracy and consistency in testing, research, and biologics production.
Trend: Growth driven by increased R&D activity in pharmaceutical labs and biotech firms.
Sterile Water / Packaged
Format: Ready-to-use, aseptic water solutions delivered in controlled packaging.
Applications: Small-scale injections, emergency kits, or situations where on-site purification is not feasible.
Benefit: Reduces infrastructure costs, ensures sterility, and provides operational flexibility.
Specialty / Reagent Waters
Use Case: Primarily in research, diagnostics, and specialized analytical workflows.
Characteristics: Ultra-pure, contaminant-free water for precise experimental or testing purposes.
Importance: Supports quality control, method validation, and sensitive assay accuracy in pharma labs.
By Technology / Generation & Treatment Platform
Reverse Osmosis (RO) + Electrodeionization (EDI)
Dominance: Primary technology for ultra-pure water generation.
Function: Removes ions, organic compounds, and particulates effectively, producing high-purity water for WFI and HPW.
Advantage: Energy-efficient relative to distillation, reliable continuous operation, scalable for large pharma plants.
Distillation
Primary Use: WFI production in sterile environments.
Process: Boils water, condenses vapor, ensures endotoxin-free, sterile water.
Significance: Preferred for injectable formulations where regulatory compliance is critical.
Ultrafiltration / Nanofiltration + UV / Ozone Disinfection
Supplementary Use: Enhances purity by removing residual microbes, organics, or pathogens.
Applications: Used in both production and analytical workflows.
Advantage: Provides additional sterilization, often integrated into hybrid systems with RO or distillation.
Continuous Monitoring & Automation / PAT Sensors
Growth: Fastest-growing technology segment in U.S. market.
Function: Real-time monitoring of water quality parameters (TOC, conductivity, microbial load).
Benefit: Ensures compliance with FDA standards, early detection of deviations, reduces human error, enhances efficiency.
Cartridge / Sterile Filters & Single-Use Assemblies
Applications: Critical for injectable drug production and aseptic filling.
Function: Final filtration before product contact, removing particulates and microbes.
Trend: Increasing demand due to single-use system adoption for flexibility and contamination control.
By End User / Buyer Type
Pharmaceutical & Biotechnology Companies
Revenue Share: Leading segment with 58% share in 2024.
Applications: Formulation, biologics, vaccines, sterile injectables.
Importance: Heavy reliance on high-purity water for regulatory compliance and quality assurance.
Contract Development & Manufacturing Organizations (CDMOs / CMOs)
Growth: Fastest-growing end-user segment.
Trend: Outsourcing of drug development and manufacturing increasing demand for WFI and HPW.
Requirement: Flexible, scalable water systems to cater to multiple clients and diverse products.
Research & Academic Labs
Usage: High-purity water for analytical, testing, and experimental purposes.
Significance: Supports R&D workflows and analytical accuracy.
CROs & Analytical Service Providers
Purpose: Provides purified water for testing, QC, and lab services outsourced by pharma companies.
Importance: Ensures compliance and reproducibility for client projects.
Diagnostics & Device Manufacturers
Requirement: High-purity water for device sterilization, diagnostic kits, and laboratory consumables.
Trend: Increasing regulatory focus on contamination-free device manufacturing.
By Solution / Business Model
On-Site Water Generation & System Sales
Dominance: Revenue share 82% in 2024.
Description: Systems installed within pharma facilities for continuous supply of purified water.
Benefits:
Reduces dependency on external suppliers.
Enhances control over water quality.
Supports regulatory compliance and large-scale production.
Water-as-a-Service / Packaged Sterile Water
Growth: Fastest-growing business model in the U.S. pharmaceutical water market.
Concept: Outsourced supply of ready-to-use high-purity water.
Advantages:
Flexible, scalable solution for small or emerging biotech companies.
Reduces capital expenditure for infrastructure.
Ensures regulatory compliance without on-site water system management.
Top 5 FAQs
1 What is pharmaceutical water?
Specially purified water meeting USP/FDA standards, essential for biologics, injectables, cleaning, and quality assurance.
2 Why is WFI dominant in the U.S. market?
Critical for sterile injectables, biologics; endotoxin-free, FDA/USP compliant; accounts for 79% revenue share in 2024.
3 Which regions dominate the U.S. pharmaceutical water market?
Northeast/Mid-Atlantic (55–60%), with Southeast & Midwest showing fastest growth due to biotech expansion.
4 How does AI impact pharmaceutical water systems?
Enables real-time monitoring, predictive maintenance, process optimization, contamination prevention, and energy efficiency.
5 Who are the top companies in the U.S. market?
Evoqua, Pall/Danaher, Merck Millipore, GE Water/Suez, Veolia, Thermo Fisher, Parker Hannifin, Grundfos, SPX Flow, Dow Membranes.
Access our exclusive, data-rich dashboard dedicated to the pharmaceuticals sector – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Immediate Delivery Available | Buy This Premium Research @
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Web: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest