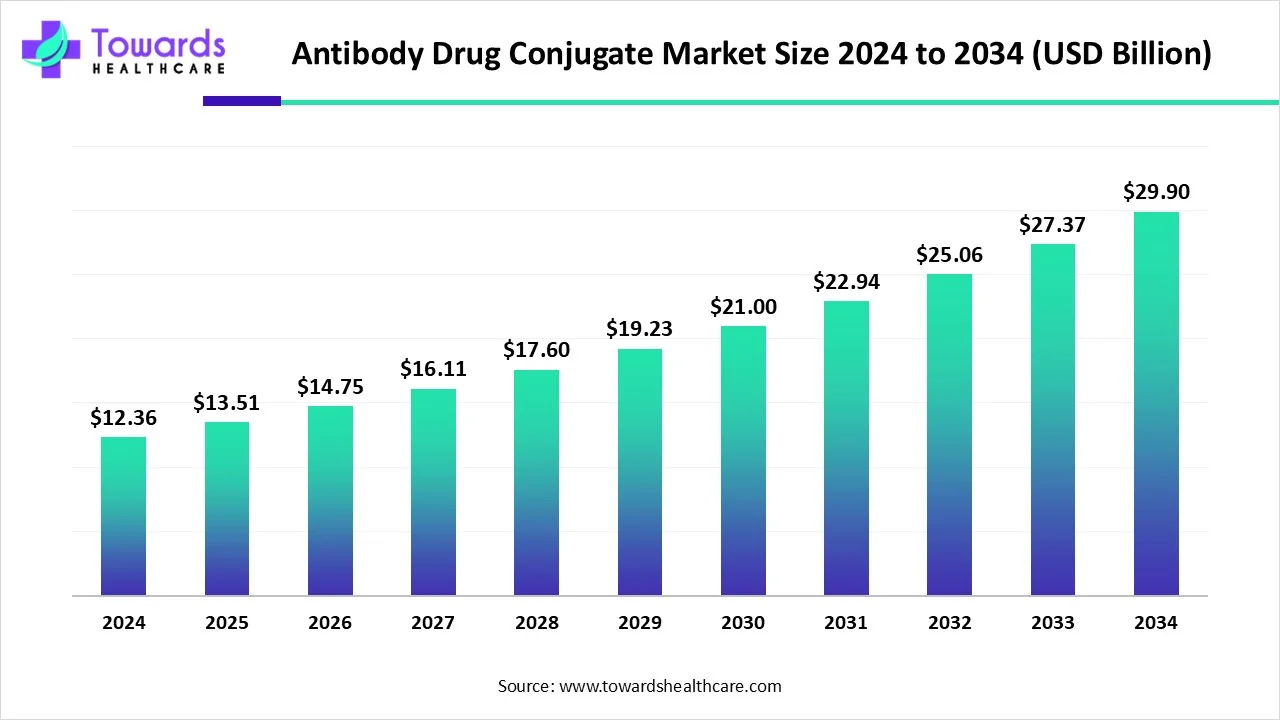
The global Antibody Drug Conjugate (ADC) market is rapidly expanding, projected to grow from USD 13.51 billion in 2025 to USD 29.9 billion by 2034, at a CAGR of 9.23%, driven by advancements in targeted cancer therapies, AI integration, and innovative ADC research.
Download this Free Sample Now and Get the Complete Report and Insights of this Market Easily @ https://www.towardshealthcare.com/download-sample/5178
Market Size
Global Market Growth:
●2024: USD 12.36 Billion
●2025: USD 13.51 Billion
●2034 Projection: USD 29.9 Billion
●CAGR (2025-2034): 9.23%
Leading Region:
●North America dominated the market in 2023 with 53% revenue share, driven by FDA approvals, well-funded biotech startups, and high cancer incidence.
Key Market Metrics:
●Segmentations: Product, Target, Technology, Region
●Top Products: Kadcyla, Enhertu, Adcetris, Padcev, Trodelvy, Polivy
●Top Targets: HER2, CD22, CD30
●Technologies: Cleavable linkers dominate with 73% revenue share in 2023.
Market Trends in Antibody Drug Conjugate (ADC) Market
Targeted Cancer Therapy Growth:
●ADCs combine monoclonal antibodies with cytotoxic drugs, allowing highly precise tumor targeting with minimal healthy tissue damage.
R&D Expansion:
●Continuous advancements in antibody design, linker chemistry, and cytotoxic payloads are accelerating ADC innovation.
Breast Cancer Dominance:
●Breast cancer ADCs (like Kadcyla) generated the largest revenue share (49%) in 2023 due to high prevalence (2.3 million women diagnosed globally in 2022).
Emerging Blood Cancer Segment:
●Blood cancer ADCs expected to grow fastest due to easier access to target cells and higher success rates in hematological malignancies.
Product Innovation:
●Enhertu expected to grow fastest (2024–2033) targeting HER2-positive breast and non-small cell lung cancer.
Technological Adoption:
●Cleavable linkers dominate; over 80% of approved ADCs use linkers that release cytotoxins selectively in tumors.
Regulatory Support:
●FDA approvals and accelerated review pathways in North America are driving faster adoption of new ADCs.
Personalized Medicine Opportunity:
●ADCs tailored to individual tumor antigens can replace chemotherapy, enabling “smart chemotherapy” and highly personalized treatment plans.
AI-Driven Discovery:
●Integration of AI into research and development accelerates drug discovery, patient stratification, and manufacturing efficiency.
Global Expansion:
●Emerging regions like Asia Pacific, India, and China show growing adoption due to rising disease incidence, government support, and healthcare investment.
AI Impact on ADC Market
Accelerated Drug Discovery:
●AI analyzes large datasets to identify promising antibody-drug combinations, reducing trial-and-error time.
Precision ADC Design:
●Predicts molecular interactions between antibodies, linkers, and cytotoxins to enhance efficacy and minimize side effects.
Patient Stratification:
●AI predicts patient response to specific ADCs, enabling personalized treatment plans.
Clinical Trial Optimization:
●AI-driven analytics help identify suitable patient cohorts and anticipate adverse reactions.
Manufacturing Efficiency:
●Real-time AI monitoring ensures consistent ADC quality and identifies production issues early.
Literature & Data Mining:
●AI processes biomedical literature to uncover novel targets, linkers, and payload insights.
Toxicity Prediction:
●Predicts potential side effects before clinical trials, lowering the risk of therapy discontinuation.
Cost Reduction:
●Optimizes R&D, clinical trials, and manufacturing, reducing time-to-market and overall costs.
Regulatory Compliance:
●AI models anticipate regulatory requirements and streamline approval submissions.
Future Research Guidance:
●AI identifies emerging ADC trends and innovative therapeutic combinations for continuous market growth.
Regional Insights
North America – Market Leader
●Market Share: Dominated the ADC market with 53% share in 2023.
Key Drivers:
High Cancer Incidence:
●Estimated 2,001,140 new cancer cases in the U.S. in 2024 and 611,720 deaths.
●Increasing prevalence of breast, blood, and lung cancers drives demand for targeted therapies like ADCs.
Regulatory Support (FDA):
●Rapid approvals of ADCs such as Kadcyla and Trodelvy encourage adoption.
●FDA’s review process and clinical trial support reduce time-to-market for novel ADC therapies.
Strong Biotech Ecosystem:
●Presence of numerous startups and established firms specializing in ADCs, like Seagen, Pfizer, and AbbVie.
●Investment in R&D for innovative linkers, payloads, and antibodies.
Healthcare Expenditure:
●High healthcare spend facilitates access to cutting-edge therapies. U.S. national cancer care spend in 2020 was $208.9 billion; expected to increase with rising cases.
Trend Insights:
●Focus on Personalized Medicine: Increasing use of HER2-targeting ADCs (Kadcyla, Enhertu) for tailored treatments.
●Clinical Trials Hub: North America hosts numerous Phase 1–3 trials for novel ADCs, particularly in hematologic malignancies and solid tumors.
Asia Pacific – Fastest Growing Market
Growth Dynamics:
●Rapid adoption of ADC therapies due to rising disease prevalence, healthcare infrastructure development, and government initiatives.
●Growth accelerated by technological integration, including advanced linker-payload combinations and molecular biology techniques.
Key Countries Driving Growth:
China:
●Large population → higher disease burden → greater demand for ADC therapies.
●Strong government support for oncology innovation, clinical trials, and domestic ADC manufacturing.
●Increasing industry-academic collaborations to develop next-gen ADCs targeting HER2, Nectin-4, and TROP2.
India:
●Rising cancer incidence and growing healthcare industry increase demand for effective treatments.
●Adoption of new ADC technologies for faster, higher-quality production.
●Government-led affordability programs improving patient access to ADCs.
Japan:
●Advanced biotech and pharmaceutical research capabilities.
●High adoption rate of novel ADCs for breast and blood cancers.
Singapore:
●AstraZeneca’s $1.5B ADC plant highlights the country as a production and innovation hub in Asia.
Market Drivers:
●Growing middle-class population with access to advanced healthcare.
●Rising prevalence of HER2-positive breast cancer and hematologic malignancies.
●Increased clinical trials and ADC pipeline expansion by global companies in the region.
Trend Insights:
●Government initiatives supporting research and development for ADCs.
●Regional collaborations between pharma companies and academic institutes accelerate innovation.
●Emerging personalized therapy adoption, particularly for breast cancer and blood cancers.
China – Regional Spotlight
●Population Factor: Largest population in APAC → high incidence of cancer cases.
Industry Growth:
●Collaborations between pharma companies and research institutes enhance ADC development.
●Investment in advanced technologies, e.g., cleavable linkers and novel payloads.
●Government Support: Policies to promote innovative cancer treatments and facilitate regulatory approvals.
India – Regional Spotlight
●Healthcare Development: Expansion of hospitals, oncology centers, and cancer research facilities.
●Technological Adoption: Companies adopting faster, quality-focused ADC production technologies.
●Patient Accessibility: Government schemes and subsidy programs make ADC therapies more affordable, increasing uptake.
Market Dynamics
1. Market Drivers – Factors Fueling Growth
Rising Cancer Incidence
●Globally increasing rates of breast, blood, lung, and pancreatic cancers drive the demand for targeted therapies.
Example: North America recorded 2,001,140 new cancer cases in 2024, emphasizing the urgent need for effective ADC therapies.
Impact: Higher patient populations and unmet medical needs accelerate ADC adoption.
Advanced ADC Research & Development
●Innovations in antibody engineering, linker chemistry, and cytotoxic payloads improve therapeutic efficacy and reduce off-target effects.
Example: Site-specific antibody conjugation and genetically engineered antibodies enhance safety and performance.
Impact: Expands pipeline of novel ADCs for both solid tumors and hematologic malignancies.
AI Integration
●Artificial Intelligence accelerates discovery by predicting optimal antibody-drug combinations and simulating molecular interactions.
●Clinical Advantage: AI-driven analytics forecast patient responses, enabling personalized treatment plans.
●Manufacturing Advantage: Real-time monitoring and predictive analytics ensure consistent product quality, reducing errors and wastage.
Regulatory Support
●FDA approvals and expedited review processes in regions like North America streamline ADC market entry.
●Impact: Encourages investment from pharmaceutical companies and shortens time-to-market for innovative ADCs.
2. Market Restraints – Factors Limiting Growth
Severe Toxicity and Adverse Events
●Many ADCs exhibit high systemic toxicity, leading to dose reductions, treatment delays, or therapy withdrawal.
●Example: Even clinically approved ADCs may cause unacceptable side effects in a significant portion of patients.
●Impact: Limits patient adoption and can slow regulatory approvals.
High Clinical Development Failure Rate
●Approximately 12 ADC compounds are in clinical use, but many fail during development due to negative risk-benefit profiles.
●Impact: Increases investment risk for pharmaceutical companies and slows pipeline expansion.
3. Market Opportunities – Areas for Growth
Personalized ADC Therapies
●Future ADCs may be tailored based on a patient’s tumor antigens and molecular profile.
●Impact: “Smart chemotherapy” could replace traditional chemotherapy, providing highly targeted treatment with fewer side effects.
Expansion in Emerging Markets
●Rapid growth expected in Asia Pacific, including China, India, and Singapore, due to rising disease prevalence and government healthcare initiatives.
●Impact: Increased patient access and new revenue streams for ADC developers.
Novel ADC Payloads and Targets
●Development of payloads like topoisomerase inhibitors (Camptothecin) and new targets such as CD22, Nectin-4, TROP2.
●Impact: Expands therapeutic potential for previously difficult-to-treat cancers.
4. Market Challenges – Barriers to Adoption
Costly R&D and Manufacturing
●ADC development involves high costs due to advanced technologies, biologics production, and clinical trials.
●Impact: Limits entry of smaller players and increases therapy pricing for patients.
Complex Regulatory Approvals
●Each ADC must pass rigorous preclinical and clinical trials for safety and efficacy.
●Impact: Long timelines and variable global regulations slow commercialization.
Managing Side Effects
●Even approved ADCs require careful monitoring for toxicity, which can complicate clinical adoption.
●Impact: Demands additional healthcare infrastructure and expertise, potentially restricting market growth in emerging regions.
Top Companies & Strengths

ADC Therapeutics SA
Product Focus: Oncology-focused ADCs.
Overview: Specializes in developing ADCs for various cancer types, leveraging proprietary linkers and payload technologies.
Strength: Strong expertise in precision oncology, robust R&D pipeline, and innovative linker-payload combinations.
AbbVie (Illinois, U.S.)
Product Focus: ADCs targeting SEZ6 and c-Met.
Overview: AbbVie’s ADCs focus on novel protein biomarkers overexpressed in tumors. Delivered via powerful cytotoxic payloads for targeted therapy.
Strength: Strong clinical trial program; expertise in personalized ADC therapies; global research collaborations.
Astellas Pharma, Inc.
Product Focus: Innovative ADCs for multiple cancers.
Overview: Develops ADCs with global regulatory approvals; pipeline includes therapies for solid tumors and hematologic malignancies.
Strength: Regulatory compliance, innovation in antibody design, strong international presence.
GlaxoSmithKline Plc (GSK)
Product Focus: Advanced ADCs with novel antibody technologies and linkers.
Overview: Focuses on optimizing antibody-linker-drug combinations to improve efficacy and safety.
Strength: Expertise in linker chemistry, robust biologics manufacturing capabilities.
Daiichi Sankyo
Product Focus: ADCs for pancreatic, breast, and other cancers.
Overview: Develops targeted ADCs like Enhertu (trastuzumab deruxtecan) with strong clinical data.
Strength: Strong R&D collaborations, global licensing agreements, and expanding clinical pipeline.
Gilead Sciences, Inc.
Product Focus: Trodelvy (ADC for breast and lung cancer).
Overview: Invests heavily in clinical trials, focusing on high-unmet-need cancers.
Strength: Strong financial backing for ADC trials, strategic partnerships, and commercialization expertise.
Pfizer, Inc.
Product Focus: Broad ADC portfolio; acquired Seagen for ADC expertise.
Overview: Plans to develop 8 blockbuster cancer therapies by 2030; integrates Seagen’s ADC technologies.
Strength: Global reach, manufacturing capabilities, and large-scale clinical development infrastructure.
F. Hoffmann-La Roche Ltd. (Roche)
Product Focus: HER2-targeting ADCs (Kadcyla, others).
Overview: Pioneered HER2-directed ADC therapies for breast cancer; global regulatory approvals.
Strength: Strong oncology R&D, proven clinical efficacy, and global market leadership.
AstraZeneca
Product Focus: ADCs for solid tumors; developing large-scale manufacturing.
Overview: Building a $1.5B Singapore ADC plant for end-to-end production.
Strength: Complete vertical integration from R&D to commercial production, strategic investments in global manufacturing.
Seagen, Inc.
Product Focus: Innovative ADCs, FDA-approved therapies.
Overview: Focused entirely on ADC development; acquired by Pfizer for enhanced collaboration.
Strength: Industry leader in ADC innovation, strong FDA approvals, and robust intellectual property portfolio.
Latest Announcements
AbbVie: Presented ADCs targeting SEZ6 & c-Met at ASCO 2024.
AstraZeneca: Announced $1.5B ADC production plant in Singapore, 2024.
Adcytherix SAS: €30M seed capital to develop ADCs for unmet medical needs, 2024.
Daiichi Sankyo Singapore: Expanding ADC access for oncology patients, 2024.
Pfizer: Cancer strategy update post Seagen acquisition, targeting 8 blockbuster ADCs by 2030.
Recent Developments
IPH4502 ADC by Innate Pharma SA – Targets Nectin-4; preclinical data presented at AACR 2025.
Heidelberg Pharma AG ADC for PDAC – Targets TROP2; shows promising efficacy.
Seagen & Kadcyla Clinical Trial – HER2CLIMB-02; progression-free survival endpoint.
Trastuzumab Deruxtecan (Enhertu) – FDA approved for HER2-positive NSCLC and breast cancer.
Galen Breast HER2 AI Tool – Enhances HER2 scoring for personalized therapy.
Segments Covered
By Product
Kadcyla
Market Position: Holds the largest market share in the ADC market (2023).
Targeted Cancer: Specifically targets HER2-positive breast cancer, delivering cytotoxic drugs directly to HER2-expressing cells.
Clinical Impact: Reduces harm to healthy tissue compared to conventional chemotherapy.
Global Reach: Approved in over 100 countries, including the U.S. and EU.
Efficacy Data: Recurrence rates are significantly lower—~13% with Kadcyla vs. 23% in standard treatment.
Recent Trials: HER2CLIMB-02 by Seagen showed progression-free survival improvement when combined with TUKYSA, even in patients with brain metastases.
Enhertu (Trastuzumab Deruxtecan)
Growth Rate: Fastest CAGR in the market (2024–2033).
Indications: HER2-positive breast cancer, HER2-low breast cancer, and non-small cell lung cancer (NSCLC).
FDA Approval: Approved for metastatic/unresectable HER2-positive cases after prior anti-HER2 treatment.
Mechanism: Precisely targets HER2 receptors, delivering a potent cytotoxic payload, allowing “smart chemotherapy.”
Market Trend: Rising adoption due to expanding indications and clinical efficacy.
Adcetris (Brentuximab Vedotin)
Use Case: Primarily for CD30-positive hematologic malignancies like Hodgkin lymphoma.
Technology: Employs a cleavable linker for targeted drug delivery, minimizing systemic toxicity.
Market Share: Strong presence in hematologic ADC segment.
Padcev (Enfortumab Vedotin)
Indications: Urothelial cancer and bladder cancer.
Market Trend: Emerging adoption in oncology with promising trial outcomes.
Trodelvy (Sacituzumab Govitecan)
Focus: Breast and lung cancers.
Investment: Gilead has received up to $210 million for clinical trials, emphasizing lung cancer applications.
Polivy (Polatuzumab Vedotin)
Use Case: Approved for blood cancers, especially B-cell lymphoma.
Targeted Action: Minimizes side effects compared to traditional chemotherapy.
Others:
New ADCs under clinical trials target a range of cancers including pancreatic ductal adenocarcinoma (PDAC), Nectin-4, and TROP2, indicating a robust pipeline for future growth.
By Target
HER2 (Human Epidermal Growth Factor Receptor 2)
Market Share: Largest share of the ADC market.
Clinical Importance: HER2 overexpression drives aggressive breast cancer proliferation.
Therapeutic Impact: ADCs targeting HER2 (Kadcyla, Enhertu) significantly improve overall survival with manageable side effects.
Technology Advancement: Preclinical and clinical research continually enhances antibody-linker-payload design for HER2.
AI Integration: Tools like Galen Breast HER2 improve HER2 scoring, aiding precise patient selection for therapy.
CD22
Growth: Fastest-growing target segment.
Disease Focus: B-cell cancers (like leukemia) and autoimmune diseases (e.g., Sjogren’s, lupus).
Therapeutic Potential: Multiple modalities including monoclonal antibodies, ADCs, CAR-T therapy, and bispecific antibodies.
Clinical Impact: Offers more precise treatment with potentially fewer systemic side effects.
CD30
Indications: Primarily hematologic malignancies (Hodgkin lymphoma).
ADC Example: Adcetris uses CD30 targeting with cleavable linkers.
Impact: Improves patient outcomes in difficult-to-treat lymphomas.
Others:
Emerging targets like Nectin-4 (IPH4502 ADC) and TROP2 (PDAC ADC) indicate market diversification.
Future ADCs are expected to target novel antigens for personalized therapy.
By Technology
Cleavable Linkers
Dominance: 73% of ADCs use cleavable linkers.
Mechanism: Release cytotoxic payload specifically in tumor cells, exploiting tumor microenvironment properties.
Examples: Brentuximab Vedotin, Inotuzumab Ozogamicin.
Advantage: Stability in bloodstream minimizes off-target toxicity.
Non-Cleavable Linkers
Mechanism: Payload remains attached to antibody until internalized in tumor cells.
Use Case: Provides slower, controlled release of cytotoxic drug.
Advantage: Enhanced safety profile for certain tumor types.
Linkerless ADCs
Mechanism: Direct conjugation of cytotoxic drug to antibody.
Potential: Still experimental; may reduce complexity but requires precise targeting.
Payload Technologies:
MMAE (Monomethyl Auristatin E): Highly potent microtubule inhibitor.
MMAF (Monomethyl Auristatin F): Similar to MMAE but less cell-permeable, reducing off-target effects.
DM4: Microtubule inhibitor used in several ADCs for hematologic cancers.
Camptothecin: Topoisomerase I inhibitor; emerging in novel ADCs.
Others: Constant innovation in payload chemistry drives ADC differentiation.
By Region
North America
Dominance: Largest share at 53% in 2023.
Drivers: High cancer incidence, robust biotech startups, strong FDA regulatory framework.
U.S. Cancer Data (2024): 2,001,140 new cases; 611,720 deaths.
Healthcare Spend: $208.9 billion in 2020; projected increase with aging population.
Europe
Mature Market: Strong presence of established ADC manufacturers (Roche, AstraZeneca).
Regulatory Support: EMA approvals streamline ADC adoption.
Asia Pacific
Fastest Growth: Driven by rising disease incidence, government support, and adoption of advanced technology.
China: High population → greater demand; industry-institute collaborations expanding.
India: Rising ADC adoption; government support for affordability and healthcare infrastructure.
Latin America & Middle East/Africa
Emerging Markets: Growing healthcare investment and awareness driving future ADC adoption.
Trend: Focus on cost-effective treatments and increasing access to ADCs.
Top 5 FAQs
1 What is the ADC market size and growth?
●Projected to grow from USD 13.51B in 2025 to USD 29.9B by 2034, CAGR 9.23%.
2 Which region leads ADC adoption?
●North America dominated with 53% share in 2023.
3 Which ADC products dominate the market?
●Kadcyla holds the largest share; Enhertu grows fastest.
4 How is AI impacting the ADC market?
●Enhances drug discovery, trial efficiency, patient personalization, manufacturing, and predictive analytics.
5 What are major market drivers?
●Rising cancer incidence, technological advancements, personalized medicine, AI adoption, and regulatory support.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Immediate Delivery Available | Buy This Premium Research @ https://www.towardshealthcare.com/price/5178
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Web: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest