In 2025, the U.S. clinical trials market is estimated to be valued at approximately $43.45 billion, with projections suggesting a surge to $80.42 billion by 2035, expanding at a compound annual growth rate of 6.35%. But what is fueling this impressive growth, and how is the industry evolving to meet the complex demands of modern medicine?
Over the last decade, the U.S. clinical trials landscape has transformed into one of the most sophisticated and well-regulated ecosystems in the world. Driven by rapid advances in biotechnology, the rising prevalence of chronic diseases, and an unprecedented focus on personalized medicine, this market has become a crucial engine for pharmaceutical innovation.
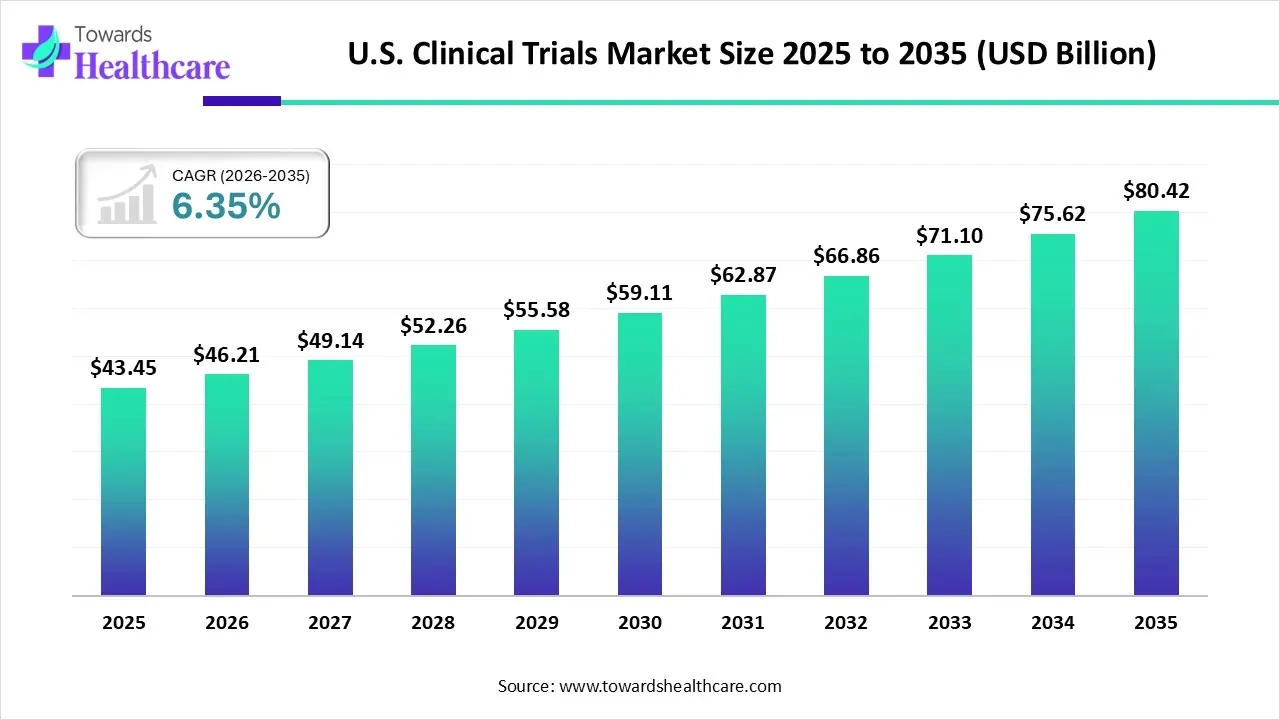
Image Source: Towards Healthcare
At the heart of this expansion is a robust infrastructure that supports everything from early-stage research to late-phase clinical trials. U.S.-based regulatory frameworks, particularly those enforced by the FDA, ensure that clinical trials meet the highest safety and efficacy standards. The market’s strength also stems from seamless collaboration between government agencies, research institutions, and pharmaceutical companies, creating a supportive ecosystem for innovation. For emerging biotech firms and established pharmaceutical companies alike, the U.S. offers an environment where science and regulation work hand-in-hand to accelerate drug development.
So, why is the U.S. such a dominant player in global clinical trials? Part of the answer lies in its significant investment in healthcare research and development. Organizations such as the National Institutes of Health (NIH) and the Department of Defense actively fund research initiatives, particularly those targeting high-burden diseases like cancer, diabetes, and cardiovascular conditions. The U.S. has a mature life sciences ecosystem that combines academic excellence with industry expertise, which encourages cutting-edge research and attracts global talent. Moreover, technological advancements in clinical trial design, patient monitoring, and data analytics have enabled faster and more precise studies.
What role do chronic diseases and personalized medicine play in this growth?
Chronic conditions like diabetes, heart disease, and cancer continue to rise in prevalence across the United States. According to recent statistics, more than 20% of American adults suffer from chronic pain, and cancer survivorship in the country exceeded 18 million as of 2022. These trends highlight a growing demand for new therapies and targeted interventions, making clinical trials a crucial component of healthcare innovation. Personalized medicine, which tailors treatment to the genetic, environmental, and lifestyle factors of individual patients, has added a new layer of complexity and opportunity. By identifying specific patient populations that will benefit most from novel therapies, clinical trials have become more efficient and more likely to deliver meaningful outcomes.
Which trial phases dominate the market, and which are poised for growth?
In 2024, phase 3 trials held the largest market share, reflecting their critical role in confirming efficacy and safety before regulatory approval. Phase 3 studies require extensive patient populations and complex infrastructure, making them the most resource-intensive part of drug development. Meanwhile, phase 2 trials are expected to experience the fastest growth between 2025 and 2034. Phase 2 trials provide crucial insights into a drug’s safety profile and effectiveness, informing decisions about progression to phase 3. Phase 1 trials, the initial human testing stage, are also expected to grow steadily, focusing on safety, dosage, pharmacokinetics, and pharmacodynamics. The emphasis on earlier trial phases highlights a broader industry trend: companies are investing in high-quality early-stage research to reduce downstream risk and accelerate the path to market.
Why is oncology at the forefront of clinical trials?
Oncology has emerged as the most active and rapidly expanding therapeutic area in the U.S. Clinical trials in cancer address a wide spectrum of diseases, from common cancers like breast and lung to rare hematologic malignancies. With over 60 oncology drug approvals in 2024 alone, the field illustrates the intensity and urgency of research efforts. Advanced therapies, such as cell and gene therapies, are further pushing trial innovation, requiring highly specialized protocols and integrated clinical and commercial strategies. The oncology focus reflects both the high unmet need and the potential for breakthrough therapies to transform patient outcomes.
How do regional dynamics affect the market? North America, led by the United States, commands a significant share of the global clinical trials market. In 2025, the North American clinical trials market is projected at $55.48 billion, expected to grow to $99.10 billion by 2035. The U.S.’s dominance stems from its well-established pharmaceutical and biotechnology industries, extensive healthcare infrastructure, and active government support. Major players in the region compete to advance novel therapeutics, attract global clinical talent, and establish best-in-class trial practices. The presence of sophisticated CROs (Contract Research Organizations) enhances operational efficiency, making the U.S. a preferred destination for both domestic and international clinical trials.
How is AI fundamentally changing clinical trial operations?
Artificial intelligence is more than a supplementary tool; it is redefining clinical research. AI algorithms accelerate patient recruitment by identifying eligible candidates faster than traditional methods, while machine learning models predict trial outcomes and optimize protocol design. Remote monitoring and biosimulation tools allow real-time tracking of patient responses, enabling faster adjustments and higher trial success rates. Beyond operational efficiencies, AI facilitates personalized medicine by helping identify subpopulations most likely to benefit from experimental therapies. The adoption of AI is no longer optional, it is becoming a requirement for companies aiming to stay competitive in this rapidly evolving landscape.
What are the current trends and sustainability drivers in the market?
Decentralized and virtual trials are gaining traction as sustainable alternatives to traditional site-based studies. By reducing the need for physical visits, these approaches lower operational costs, improve patient convenience, and expand access to underserved populations. The rise of pharmacogenetics and personalized medicine is also contributing to more sustainable and efficient trial designs, as smaller, targeted studies reduce unnecessary patient exposure and accelerate drug development. Investors, from venture capital to government agencies, are increasingly supporting innovative trial platforms that combine technology, AI, and patient-centric approaches, fueling both market growth and long-term sustainability.
How does the startup ecosystem influence clinical trials?
The U.S. clinical trials market is not just dominated by established players; a vibrant startup ecosystem is driving disruptive innovation. Startups are leveraging big data, AI, and cloud-based platforms to streamline trial operations, improve patient engagement, and create new models for decentralized and hybrid trials. Many of these companies focus on niche therapeutic areas or specialized trial designs, filling gaps left by traditional CROs. Their success demonstrates how agility and innovation can coexist with scale and regulatory compliance in one of the most complex industries in healthcare.
What do recent developments tell us about the market’s direction?
In November 2025, the American Cancer Society expanded ACS ACTS, a nationwide clinical trial matching service aimed at improving equitable access to trials. This initiative highlights a growing emphasis on patient-centric models and accessibility. Meanwhile, the U.S. Department of Defense awarded $7.3 million to support a novel metastatic triple-negative breast cancer trial, illustrating continued government investment in high-impact research. These developments underscore the market’s focus on innovation, inclusion, and collaboration, shaping a future where clinical trials are not only more efficient but also more socially responsible.
Why should the global community watch U.S. clinical trials?
The U.S. remains a bellwether for global clinical research trends. Its strong regulatory oversight, investment in technology, and ability to attract talent and capital make it a testing ground for innovations that often spread worldwide. Whether it’s the integration of AI, the rise of decentralized trials, or the focus on patient-centered oncology research, the strategies adopted in the U.S. often define global best practices. For stakeholders in pharma, biotech, and healthcare policy, understanding U.S. trends is critical to navigating the evolving landscape of clinical research.
Access our exclusive, data-rich dashboard dedicated to the Life Science Industry – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout U.S. Clinical trials Market Report Now at: https://www.towardshealthcare.com/checkout/6455
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest