Medical Devices Testing Market Growth
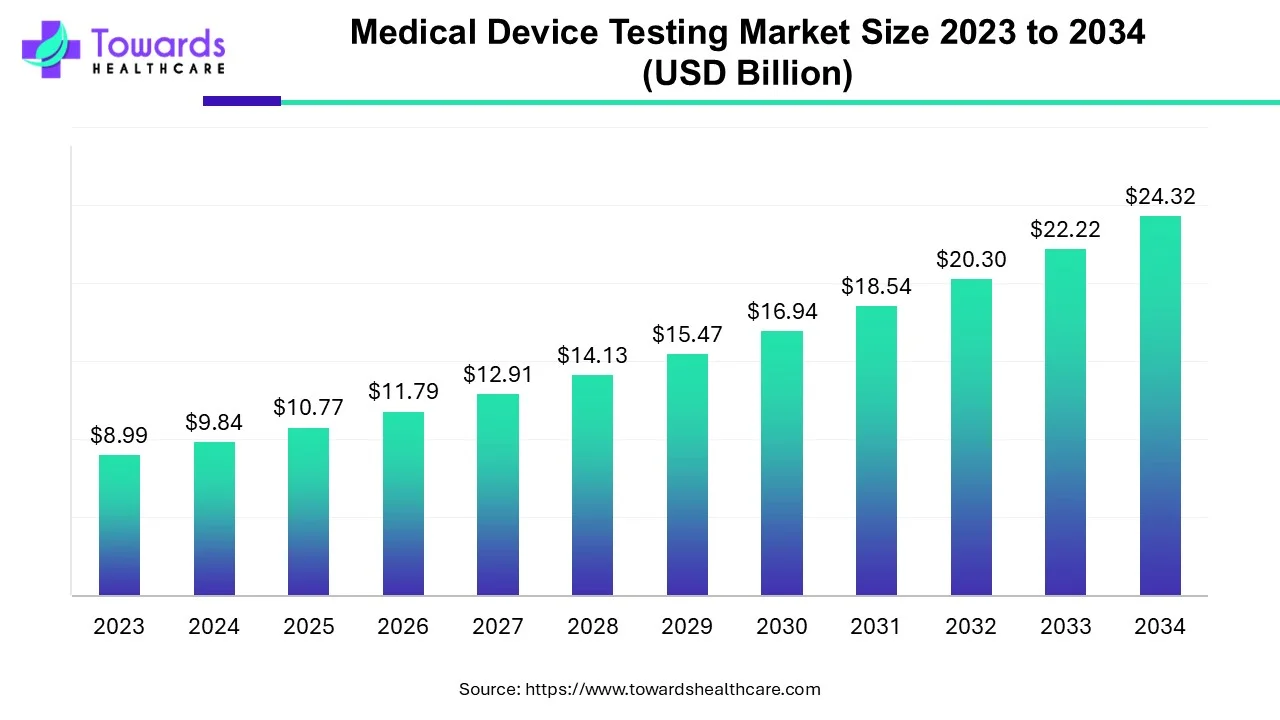
The global medical device testing market was valued at USD 10.77 billion in 2025 and is projected to reach USD 24.32 billion by 2034, growing at a CAGR of 9.47%, driven by strict regulatory standards, rising healthcare spending, and rapid medical innovation.
Download Free Sample of Medical Devices Testing Market Now and Get the Complete Report Easily at: https://www.towardshealthcare.com/download-sample/5457
Medical Device Testing Introduction
The medical device testing market is undergoing unprecedented growth as healthcare systems worldwide demand safer, high-quality, and technologically advanced medical devices. Rising chronic diseases, increasing global healthcare spending, expanding medical device manufacturing hubs (especially in Asia-Pacific and North America), and stricter regulatory compliance norms are pushing manufacturers to invest heavily in comprehensive testing solutions. The market is further strengthened by government initiatives such as India’s ₹500 crore scheme to boost domestic device manufacturing, Europe’s stringent MDR & IVDR regulations, and the US FDA’s growing focus on personalized medicine and cybersecurity. As the number of clinical trials increases and innovation accelerates, spanning respiratory, orthopedic, cardiovascular, and smart surgical devicesthe need for precise, reliable, and cost-effective testing becomes indispensable.
What is Medical Device Testing?
Medical device testing refers to the systematic process of evaluating medical devices to ensure their safety, performance, biocompatibility, chemical stability, sterility, functionality, and regulatory compliance before they enter the market or are used on patients. It includes preclinical and clinical evaluations, biocompatibility assessments, chemistry tests, microbiology tests, package validation, and performance verification. The objective is to ensure that every medical devicefrom implants to diagnostics, surgical tools to wearablesmeets global regulatory standards such as FDA, CDSCO, MDR, and NMPA, guaranteeing patient safety and product effectiveness.
Medical Device Testing Market Size
Global Market Value Growth
USD 10.77 billion (2025) → USD 24.32 billion (2034)
CAGR: 9.47%, indicating strong long-term demand for testing services due to regulatory pressures and device complexity.
Long-Term Market Outlook (2023–2034)
Market growth is supported by expanding device innovations, rising clinical trial volumes (1,053 active + 4,101 recruiting trials in 2024), and healthcare infrastructure upgrades.
Investment Boost Through Government Schemes
India’s ₹500 crore medical device strengthening scheme (2024) accelerates testing capabilities and domestic manufacturing.
Canada allocated US$ 801 million for drugs & device testing improvement.
Healthcare Spending Influence
U.S. healthcare expenditure projected at USD 6.2 trillion by 2028, directly increasing demand for device testing.
Expansion of Global Medical Device Market
Medical devices market size:
USD 586.20B (2025) → USD 1083.96B (2035)
CAGR: 6.34%, creating a proportional rise in testing demand.
Increased Device Launches (2025 Pipeline)
Higher global R&D results in more testing needs:
Magcath female incontinence device
MediBuddy x ELECOM IoT devices
PsiFGuard smart drilling system
Medical Device Testing Market Trends
Government Programs Boosting Testing Demand
India’s ₹500 crore scheme (2024) strengthens testing labs, clinical studies & R&D infrastructure.
Supports self-reliance in device production → increases testing needs domestically.
Surge in Pediatric and Condition-Specific Devices
TidalSense (UK) introduced a child asthma diagnostic tool in 2024 → increases pediatric testing demand.
High Clinical Trial Volumes
5,154+ device-related trials in 2024 → huge testing pipeline.
Rising Global Healthcare Spend
Improved incomes & demand for quality healthcare lead to increased device production & testing.
Cybersecurity & Software Testing Demand
As devices become IoT-enabled (Ex: MediBuddy-ELECOM collaboration), testing for cybersecurity grows.
Shift Toward AI-Driven Testing & Automation
AI being used for anomaly detection, predictive maintenance, and compliance documentation.
Growing Emphasis on Biocompatibility
Biocompatibility test segment led in 2024 due to rise in implants, dermal fillers, ophthalmic devices.
Chemistry Testing Surge
Chemical characterization, toxicology & hazard compliance needs growing rapidly.
Clinical Trial-Linked Testing Demand
Clinical segment dominated 2024 due to regulatory evolution and complex device categories.
Rapid Asia-Pacific Expansion
China & India leading due to affordability, workforce, government support, and manufacturing base.
AI Impacts / Roles in the Medical Device Testing Market
Automated Defect Detection
AI algorithms scan huge datasets from device sensors & test runs, catching defects humans may miss.
Predictive Maintenance Modeling
ML predicts device failure points, reducing recall risks.
Real-Time Device Monitoring
AI monitors devices during testing for instantaneous alerts on performance deviations.
Testing Process Automation
AI speeds up chemical, biological & mechanical testing cycles by automating repetition tasks.
Regulatory Documentation Automation
AI generates, organizes & audits compliance documents for FDA, MDR, CDSCO, etc.
Simulation of Patient Conditions
ML models simulate real-world patient scenarios for cardiovascular, orthopedic, and neural devices.
Improved Cybersecurity Testing
AI identifies possible cyber vulnerabilities in smart & IoT-ready medical devices.
Enhanced Clinical Trial Analysis
AI improves safety monitoring, trial selection & anomaly detection in the clinical testing phase.
Anomaly Pattern Recognition
AI identifies unseen error patterns in device behavior during long-duration testing.
Post-Market Surveillance Intelligence
AI continuously scans performance data → helps identify potential recalls faster.
Regional Insights
Asia-Pacific (Dominant Region – 2024)
Why dominant?
Rapid healthcare infrastructure growth.
Government investments (ex: India’s ₹500 crore scheme).
Large domestic manufacturing hubs.
High demand for cost-effective testing services.
China Highlights:
NMPA’s 2023 regulations upgraded testing standards.
“Made in China 2025” emphasizes AI-driven & automated testing systems.
Large-scale biocompatibility and chemistry testing expansion.
India Highlights:
39 CDSCO-approved MTDLs (2023).
Strong startup ecosystem with 800+ medical device manufacturers.
Push for self-reliance in manufacturing & testing.
North America (Fastest Growth)
Drivers:
Stringent FDA regulations → increasing testing requirements.
Large R&D hubs & high technology adoption (AI, sensors, robotics).
OEM–EMS collaborations driving electronic device testing demand.
USA:
Leading in AI-driven testing (ex: Medtronic advanced AI testing for implants).
High investment in personalized medicine & cybersecurity testing.
Canada:
US$ 801M allocated for drug & device oversight improvements.
Focus on device safety under Health Canada’s Medical Device Action Plan.
Europe (Strong Growth)
Key Drivers:
MDR & IVDR creating demand for extensive testing.
Strong R&D investments and advanced testing labs.
Rising elderly population → more device usage → more testing demand.
Germany:
Strict CE compliance, MPG law, and MDR testing rules fuel market growth.
UK:
£10M government investment to accelerate device development & testing.
Target areas: Alzheimer’s, COPD, diagnostics.
Middle East & Africa
Growth Reasons:
Digitization of healthcare.
Government support for medical device development.
Increasing adoption of advanced diagnostic & surgical devices.
UAE:
MOHAP regulates device approvals.
Strong domestic production and export to Iran.
Companies like OMC Medical, SGS SA offering local testing facilities.
Market Dynamics
Driver – Global Healthcare Growth
Rising incomes, population expansion, and health awareness drive demand for advanced devices.
More device production → more mandatory testing.
Reliability and safety expectations higher than ever.
Driver – Increased Healthcare Spending
Example: U.S. spending to reach $6.2 trillion by 2028 → huge need for quality testing.
Governments globally increasing investments: India (INR 98,461 crore), Canada (US$ 801M).
Restraint – Risks & Failures
Component failures, design defects, software bugs, mishandling → testing delays & costs.
WHO: 30% of adverse events linked to device misuse or poor evaluation.
Cybersecurity threats increasing.
Opportunity – Policy & Innovation Push
Global startups and R&D investment expanding testing demand.
China, India, Europe, USA all supporting device manufacturing → testing market grows automatically.
Which are the Top 10 Companies in the Medical Device Testing Market

SGS SA
Product/Services: Biocompatibility, microbiology, chemistry, package validation.
Strength: Global testing dominance; strong regulatory expertise.
LabCorp (Laboratory Corporation of America)
Product: Clinical & preclinical device testing.
Strength: Large lab network + strong clinical research capability.
Nelson Laboratories
Product: Sterility, bioburden, endotoxin, antimicrobial testing.
Strength: Specialization in microbiology & ISO 17025 accredited labs.
TÜV SÜD
Product: CE marking, regulatory approval testing.
Strength: Trusted certification body in Europe.
Charles River Laboratories
Product: Preclinical testing, animal research, toxicology.
Strength: Strong in GLP preclinical studies.
Element Minnetonka
Product: Physical and environmental device testing.
Strength: High-tech laboratory setups.
NAMSA
Product: Biocompatibility & clinical trial solutions.
Strength: Pioneers in customized biocompatibility research.
Eurofins Scientific
Product: Chemical characterization, E&L testing.
Strength: Largest analytical chemistry network globally.
Pace Analytical Services
Product: Microbiology & chemistry testing.
Strength: Deep U.S. market presence.
Intertek Group
Product: Functional, electrical, and performance testing.
Strength: Global testing & certification leader.
Latest Announcements
CEO Statement MedTech Solutions (John Doe)
Highlights increasing importance of reliable testing to support device innovation.
Emphasizes focus on faster time-to-market + compliance.
Reinforces industry-wide push for cost-effective and safe device approval.
Recent Developments
Intertek x Emitech Collaboration (2023)
Combines engineering and testing expertise.
Expands capabilities in electronics, performance & environmental testing.
IIT Madras – Mobile Device Calibration Unit (2024)
India’s first mobile unit for calibration testing.
Improves access to high-quality device testing → boosts diagnostic accuracy.
Segments Covered
12.1 By Service
A. Biocompatibility Tests
Biocompatibility testing evaluates how well a medical device material interacts with human tissues, ensuring it does not cause toxicity, irritation, inflammation, or long-term adverse responses. This is one of the most regulated and mandatory service segments under standards like ISO 10993 and FDA guidelines.
Cardiovascular Devices
Includes stents, heart valves, catheters, vascular grafts.
Must undergo hemocompatibility tests (thrombosis, hemolysis) because they contact blood directly.
Long-term implant studies are done to assess tissue overgrowth, calcification, and foreign-body reactions.
Orthopedic Implants
Metal and polymer-based devices such as joint replacements, screws, plates, rods.
Tested for chronic toxicity, wear debris impact, and compatibility with bone and muscle tissues.
Mechanical stress during use requires additional fatigue and degradation evaluations.
Dental Devices
Fillings, crowns, orthodontic brackets, implants.
Saliva exposure leads to unique corrosion and material-leaching tests.
Tests include mucosal irritation, cytotoxicity, and long-term stability under chewing forces.
Dermal Devices
Skin-contact products like patches, adhesives, wound dressings.
Assessed for irritation, sensitization, and allergenicity.
Must remain safe during continuous or repeated skin exposure.
Neurosurgical Devices
Neural implants, shunts, electrodes, spinal fixation products.
Require high-stringency testing for neurotoxicity and inflammation due to direct brain/spinal interaction.
Long-term implant performance under cerebrospinal fluid conditions is evaluated.
Ophthalmic Devices
Contact lenses, intraocular lenses, surgical tools.
Eye tissue is extremely sensitive; tests for ocular irritation, compatibility, and optical clarity are mandatory.
Even low-level chemical leaching must be eliminated.
General Implants
All other long-term implants (breast implants, mesh, pacemaker casings).
Assessed for systemic toxicity, implant degradation, and host-tissue interaction across multiple organs.
Biocompatibility ensures the device’s materials will not trigger harmful biological reactions when placed inside or in contact with the human body.
Chemistry Tests
These tests evaluate the chemical composition, leachables, and extractables from device materials to ensure patient safety.
Extractables & Leachables (E&L) Characterization
Extractables: Chemicals released from materials under harsh conditions.
Leachables: Chemicals that migrate into the body under real-use conditions.
These tests detect carcinogens, endocrine disruptors, toxic metals, or plasticizers.
Analytical Method Development
Customized assays to quantify specific chemicals.
Includes mass spectrometry, chromatography, and spectroscopy techniques.
Ensures regulatory bodies have validated, reproducible data.
Toxicological Assessments
Toxicologists analyze chemical risk levels based on exposure, dose, and duration.
Generates toxicological risk assessments (TRAs) required by regulators for clearance.
Identifies unsafe chemicals and recommends formulation changes.
Chemistry tests ensure devices do not release harmful chemicals into the human body throughout their life cycle.
Microbiology & Sterility Testing
This segment ensures that devices are free from harmful microorganisms, and that sterilization processes are effective and compliant with global standards.
Bioburden Testing
Measures the microbial load present on the device before sterilization.
Helps determine if the sterilization process is sufficient.
Important for single-use disposable devices.
Endotoxin Testing
Detects bacterial endotoxins that can cause fever, shock, or inflammation.
Especially critical for devices contacting blood or cerebrospinal fluid.
Sterility Tests
Confirms that the device packaging and sterilization method eliminate all viable microorganisms.
ISO/FDA-mandated for surgical tools, implants, and diagnostics.
Antimicrobial Resistance (AMR) Evaluation
For devices with antimicrobial coatings.
Ensures coatings don’t unintentionally promote resistant strains.
Evaluates long-term antimicrobial efficiency.
Microbiology ensures devices are microbially safe, sterile, and do not trigger infections after clinical use.
Package Validation
Packaging maintains sterility and protects devices during storage, transport, and shelf life.
Integrity Testing
Detects leaks, seal defects, or micro-holes.
Ensures no microbial entry during shipping.
Transportation Simulation
Vibrations, drops, compression tests mimic real-world logistics conditions.
Confirms packaging durability.
Accelerated Aging Studies
Determines shelf life by simulating years of storage.
Ensures sterility and mechanical safety throughout the expiration period.
Material Compatibility
Ensures packaging materials do not react with the device or compromise sterilization.
Important for radiation-sterilized products.
Package validation prevents contamination, damage, and failure before the device reaches the patient or hospital.
By Phase
Preclinical Phase
Preclinical testing evaluates safety, functionality, and biological response before human use. This phase is mandatory for all moderate- and high-risk devices.
Large Animal Studies (Implantable & Advanced Devices)
Used for heart valves, stents, orthopedic implants, neuro devices.
Animals like pigs, sheep, or dogs replicate human physiology.
Studies include:
Implant stability
Tissue response & inflammation
Hemodynamic performance
Mechanical wear
Essential for FDA/CE regulatory submissions.
Small Animal Studies (Early Toxicology & Performance)
Mice, rats, rabbits used for initial safety evaluations.
Focus on:
Toxicity
Irritation & sensitization
Systemic responses
Helps refine device prototypes before large-animal or human trials.
Preclinical testing acts as the safety and performance foundation before human studies begin.
Clinical Phase
Clinical testing involves real-patient evaluation under strict regulatory environments.
Human-Use Evaluation Under Controlled Trials
Devices are tested on real patients under clinical supervision.
Evaluates:
Clinical performance
Safety outcomes
Adverse events reporting
Long-term effectiveness
Dominated 2024 due to Regulatory Evolution
Regulatory bodies (FDA, EMA, CDSCO) increased requirements for higher-risk devices.
Rising demand for:
Post-market surveillance
Real-world evidence
Stronger clinical validation
Clinical phase became the largest and fastest-growing segment because companies must generate more robust data for approvals.
Clinical trials validate real-world device performance, ensuring safety and compliance before commercial launch.
Top 5 FAQs
1. What is the market size of the medical device testing industry?
The market is USD 10.77 billion in 2025 and expected to reach USD 24.32 billion by 2034 at 9.47% CAGR.
2. Which region dominated the market in 2024?
Asia-Pacific due to strong manufacturing bases, low-cost testing, and supportive government programs.
3. Which service segment led the market?
Biocompatibility testing, driven by high implant usage and strict regulatory standards.
4. How is AI contributing to medical device testing?
AI enhances defect detection, predictive maintenance, automated documentation, cybersecurity checks, real-time monitoring, and patient-condition simulations.
5. Which country is expected to grow the fastest?
North America, especially the U.S., due to strict FDA regulations, advanced testing technology, and cybersecurity demands.
Access our exclusive, data-rich dashboard dedicated to the medical devices industry – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout Medical Devices Testing Market Report Now at: https://www.towardshealthcare.com/checkout/5457
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest