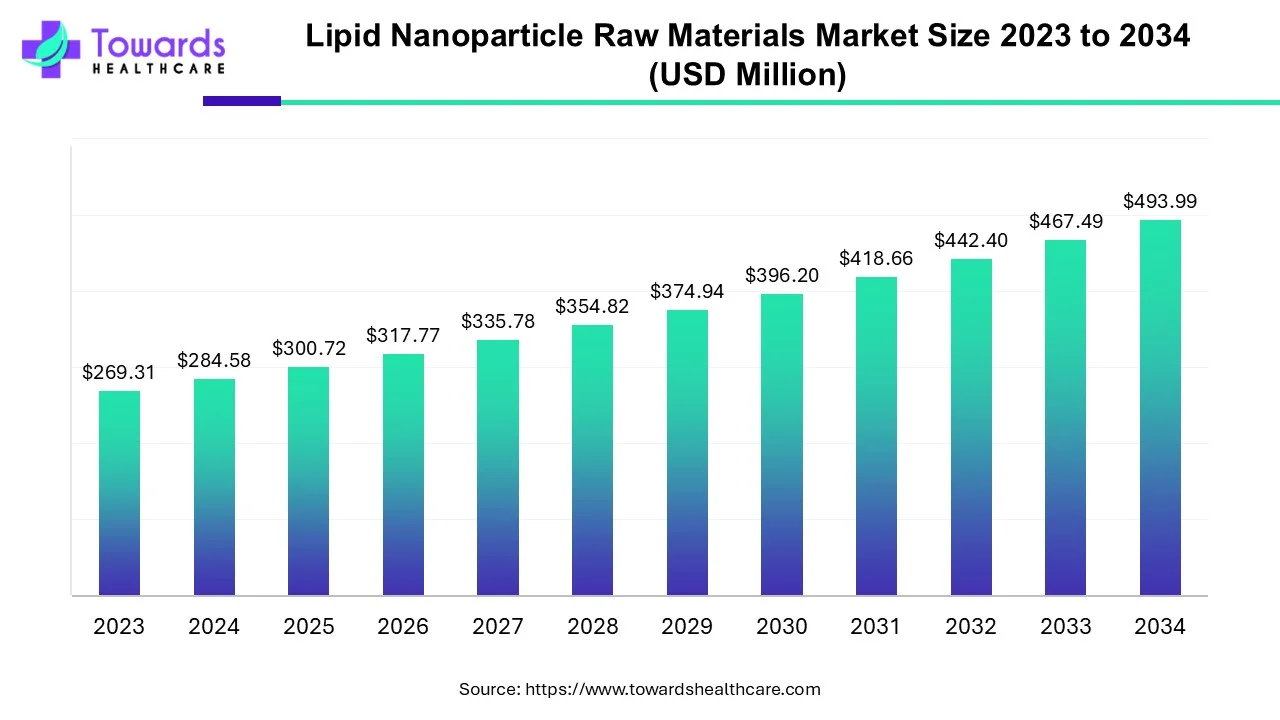
The global lipid nanoparticle (LNP) raw materials market, valued at USD 300.72 million in 2025, is projected to reach USD 493.99 million by 2034 at a 5.67% CAGR, driven by rising demand for advanced drug delivery systems, mRNA vaccines, and gene therapies.
Download Sample: https://www.towardshealthcare.com/download-sample/5168
Market Size Overview
1. Overall Market Growth
The market grows from USD 300.72 million (2025) to USD 493.99 million (2034), driven by rising global investments in biologics, mRNA-based vaccines, and gene editing therapies.
2. Long-Term Growth Stability
The CAGR of 5.67% (2025–2034) reflects consistent medium-paced growth, supported by rapidly expanding pipelines in oncology, infectious disease therapies, and genomic medicine.
3. Dominant Market Geography
North America held 41% of global revenue in 2023, driven by strong biotech presence, large mRNA vaccine production, and robust R&D ecosystems.
4. Strong Academic and Pharma Research Demand
Increasing use of LNPs in research institutions and pharmaceutical development pipelines drives multi-year procurement of lipids and kits.
5. High Clinical Trial Pipeline Contribution
100+ clinical-stage RNA, mRNA, CRISPR, and therapeutic protein programs globally rely on LNPs, increasing raw materials demand.
6. Infectious Disease Dominance
Infectious diseases contributed >47% market share (2023) due to high use of LNPs in mRNA vaccine formulation.
7. Cancer as Fastest-Growing Indication
Cancer therapies using LNPs for siRNA, mRNA, and CRISPR delivery are expected to grow at the highest CAGR during 2025–2034.
8. Growth Driven by Therapeutics Demand
Therapeutics accounted for >63% revenue share (2023) reflecting strong pipeline for LNP-based drug formulations.
9. High Adoption of Kits
Kits accounted for 55% share in 2023, used widely in early-stage research, optimization, and preclinical development.
10. Asia-Pacific as Fastest Growing Region
China, India, and Japan drive rapid growth due to investments in manufacturing infrastructure, collaborations, and raw material export capability.
Market Trends
1. Rapid Expansion of mRNA Vaccine Technologies
mRNA technologies expanded after COVID-19, with companies like Moderna and BioNTech generating major revenues.
mRNA-based RSV vaccine mRESVIA launched in 2024, contributing USD 2M in Q1 2025 sales.
2. Gene Therapy Driving Strong Demand
RNA/DNA-based therapies require precise LNP formulations.
LNPs protect nucleic acids from degradation—crucial for CRISPR, siRNA, and gene replacement therapies.
3. Rise of AI-Based LNP Design
Mana.bio introduced an AI-powered LNP design platform (2023) enabling advanced formulation design, targeting extra-hepatic tissues.
4. Increasing Use of Starter Kits
55% market share in 2023 held by kits, boosted by CordenPharma’s 2024 LNP starter kits for mRNA formulations.
5. Technological Advancements in Microfluidics
2025: Shin-Etsu Chemical + Hokkaido University developed microfluidic-based LNP production, improving scalability and uniformity.
6. Growing CRISPR Delivery Research
2025: East China Normal University used LNP-CRISPR delivery for treating primary hyperoxaluria type 1 with sustained therapeutic effect.
7. Strong Oncological Pipeline Adoption
Cancer is fastest growing indication backed by rising global cases: 20 million new cases & 9.7 million deaths in 2022.
8. Heavy R&D Investments in North America
USAID’s Global Health R&D Strategy (2023–2028) pushes adoption of novel delivery technologies like LNPs.
9. Rise in Contract Manufacturing Expansion
WuXi STA (2023–2024) expanded large-scale sterile injectable and LNP platforms up to 50L batch production capacity.
10. Regulatory Push in Europe
The European Pharmacopoeia Commission (2024) adopted new guidelines on mRNA vaccine components—boosting raw material demand.
AI Impacts & Roles in the LNP Raw Materials Market
1. AI-Driven Lipid Structure Optimization
AI designs new lipid molecules with enhanced biocompatibility, stability, and targeting efficiency.
2. Predictive Modeling for LNP Behavior
AI simulates LNP biodistribution, membrane fusion, toxicity, and endosomal escape—reducing experimental costs.
3. Automated Microfluidic LNP Production
AI controls mixing parameters, temperature, and flow rate to ensure batch consistency.
4. AI-Enabled CRISPR Delivery Optimization
Predicts optimal LNP components for delivering CRISPR/Cas cargo to specific tissues.
5. AI-Assisted Raw Material Quality Control
Detects impurities in ionizable lipids, PEG-lipids, and sterols using ML-based chemical profiling.
6. Supply Chain Forecasting
AI predicts global demand surges (e.g., pandemics) enabling better raw material availability.
7. Drug Loading Optimization
AI models maximize encapsulation efficiency (EE) for mRNA, siRNA, and proteins.
8. Toxicity Prediction
Identifies toxicity risks early by analyzing structural interactions between lipids and biological membranes.
9. Personalized LNP Formulation
AI tailors lipid ratios for individual patient needs in personalized vaccines and gene therapies.
10. AI-Driven Market Forecasting
Predicts LNP material price fluctuations, competition, emerging regions, and clinical trial success impact.
Regional Insights
1. North America (Largest Share – 41% in 2023)
Key Drivers
Strong biotech ecosystem: Moderna, Pfizer-BioNTech, Cytiva.
High investment in R&D (USAID Global Health R&D Strategy 2023–2028).
Growing cancer and infectious disease burden.
Subpoints
48 million diarrheal cases annually in U.S. stimulate infectious disease R&D.
Expected 20M new cancer cases yearly in U.S. among aging population.
2. Europe (Regulation-Driven Growth)
Drivers
Adoption of mRNA vaccine quality guidelines (EPC 2024).
Moderna and BioNTech operations fueling raw material demand.
Subpoints
Strong funding for mRNA vaccine procurement.
Increasing shift toward biologics and novel delivery platforms.
3. Asia-Pacific (Fastest Growing Region)
Drivers
Rapid investments in biomanufacturing.
Key players: WuXi Biologics, Shin-Etsu Chemical, VAV Life Sciences.
High export of lipid raw materials (India, China).
Subpoints
Microfluidic LNP production innovations (Japan).
Large-scale automated injectable lines by WuXi STA.
4. Latin America
Drivers
Growing interest in mRNA vaccines post-pandemic.
Increasing collaborations with global suppliers.
5. Middle East & Africa
Drivers
Slow but emerging rise in biologics production.
Export dependence on Asia-Pacific and Europe.
Market Dynamics
Drivers
Growth of mRNA vaccines (Moderna, BioNTech revenues).
Rising demand for targeted drug delivery systems.
Increased cancer cases globally.
Expanding gene therapy pipeline requiring LNP delivery.
North America’s heavy R&D investments.
Technological advancements (microfluidics, automated production).
Restraints
Burst release behavior limiting control over drug release.
Temperature-sensitive lipid properties.
Complex production process requiring advanced manufacturing controls.
Opportunities
CRISPR and RNA therapeutics adoption.
AI-driven LNP formulation engineering.
Large-scale mRNA vaccine production expansions.
Increasing partnerships (Genevant–Tome Biosciences, 2024).
Challenges
Maintaining stability across temperature variations.
High regulatory scrutiny for biologic delivery systems.
Need for consistent quality in lipid raw materials.
Top 10 Companies

1. Polysciences Inc.
Product: Ionizable lipids, PEG lipids.
Strength: Strong lipid synthesis expertise, high-quality research-grade materials.
2. NOF America Corporation
Product: PEGylated lipids.
Strength: Global leader in PEG chemistry, long-term supplier to major pharma.
3. Biopharma PEG Scientific
Product: PEG derivatives & PEG lipids.
Strength: Large portfolio, highly customizable PEG solutions.
4. Creative Biolabs
Product: Custom LNP formulation kits and services.
Strength: Strong R&D and customized service capability.
5. CordenPharma International
Product: LNP Starter Kits (launched Feb 2024).
Strength: Full-service CDMO with integrated lipid manufacturing.
6. Tebubio
Product: Reagents, phospholipids, sterols.
Strength: Strong distribution network in Europe.
7. Avanti Polar Lipids
Product: High-purity ionizable lipids.
Strength: Gold standard for research-grade lipids.
8. Hopewell Therapeutics
Product: Tissue-targeted LNPs (ttLNPs).
Strength: Advanced next-gen delivery for genomic medicines.
9. Echelon Biosciences
Product: Specialty lipids, sterols.
Strength: Strong innovation in biochemical lipid derivatives.
10. Merck KGaA
Product: LNP raw materials, reagents, lipids.
Strength: Global-scale manufacturing and stringent quality control.
Latest Announcements
Hopewell Therapeutics (May 2024)
Presented preclinical data for ttLNPs in oncology, pulmonary, and vaccine programs.
Focus on next-gen lipid structures for targeted delivery.
Cytiva (July 2023)
Introduced NanoAssemblr commercial formulation system.
Provides scalable, automated, GMP-compliant LNP manufacturing capability.
Recent Developments
1. Shin-Etsu Chemical + Hokkaido University (April 2025)
New microfluidic LNP production system.
Lilac Pharma designated as distributor.
2. CRISPR-LNP Delivery for PH1 Treatment (March 2025)
Achieved sustained therapeutic effects in mouse models using LNP-CRISPR platform.
3. Genevant + Tome Biosciences (Jan 2024)
Agreement valued at up to $114.3 million for gene-editing therapies.
4. Mana.bio AI LNP Platform (Oct 2023)
Raised $19.5M seed funding.
AI-first approach to lipid design and LNP formulation.
5. GenScript LNP & circRNA Services (Sept 2023)
Added LNP and circRNA formation to IVT mRNA synthesis.
6. WuXi STA Expansion (July 2023–2024)
Introduced high-potency sterile injectable line.
LNP platform scaled to 50L batch capacity.
New lines planned in Switzerland and U.S.
Segments Covered
1. Product Segment
The LNP raw materials market is categorized by the type of lipids and support materials required for LNP formulation. Each component plays a distinct functional role in enabling stability, delivery efficiency, and therapeutic performance.
a. Ionizable Lipids
Role:
Core functional component responsible for RNA encapsulation and efficient intracellular delivery.
Key Characteristics:
Exhibit neutral charge at physiological pH, reducing toxicity.
Become positively charged in acidic endosomal environments, enabling endosomal escape.
Drive gene expression efficiency in mRNA therapeutics.
Market Insights:
High demand due to the expansion of mRNA vaccines, gene therapy vectors, and CRISPR delivery systems.
Innovation focused on improving biodegradability and reducing immunogenicity.
b. PEGylated Lipids
Role:
Provide steric stabilization and extend circulation time in the bloodstream.
Key Characteristics:
Reduce aggregation and opsonization.
Enable better pharmacokinetics, promoting sustained release.
Market Insights:
Demand reinforced by long-acting RNA therapeutics.
Regulatory concerns about PEG-induced hypersensitivity drive development of alternative stealth lipids.
c. Sterol Lipids
Role:
Essential for maintaining membrane rigidity and stability of the nanoparticle.
Key Characteristics:
Typically include cholesterol or modified sterols.
Improve LNP fusion capability and cellular uptake.
Market Insights:
Increasing adoption of synthetic sterol analogs that enhance encapsulation efficiency and serum stability.
Larger volumes consumed in vaccine-scale manufacturing.
d. Neutral Phospholipids
Role:
Provide structural architecture and help mimic natural lipid bilayers.
Key Characteristics:
Improve biocompatibility.
Reduce toxicity and enhance stability during storage.
Market Insights:
Widely used in both clinical-grade and research-grade LNP formulations.
Growth driven by demand for LNPs in protein replacement and rare disease therapies.
e. Kits (55% Market Share in 2023)
Role:
Ready-to-use formulation kits for small-scale and medium-scale development.
Usage Areas:
Preclinical formulation optimization.
Screening multiple LNP compositions for gene delivery efficiency.
Academic and biotech R&D programs.
Market Insights:
Dominate due to ease of use and suitability for early-stage research.
Increasing customization based on specific RNA types (mRNA, saRNA, siRNA, microRNA).
f. Reagents
Role:
Support LNP creation, characterization, and stability testing.
Types:
Buffers, solvents, cryoprotectants.
Encapsulation assessment reagents.
Nanoparticle tracking analysis (NTA) and QC reagents.
Market Insights:
Growth tied to the rising need for analytical quality controls in GMP manufacturing.
Essential for scale-up and large-batch validation.
g. Other Raw Materials
Includes:
Specialty lipids, synthetic analogs, biodegradable lipids.
Stabilizers used during lyophilization and long-term storage.
Novel materials supporting LNP shelf-life extension.
Market Insights:
Innovation accelerating as companies try to reduce cold-chain reliance.
Growing collaboration with polymer chemistry firms to develop hybrid LNP technologies.
2. Disease Indication Segment
LNP raw materials are used across multiple therapeutic areas, particularly in genetic and RNA-based therapies.
a. Cancer
Growth Driver:
Fastest-growing segment due to rapid adoption of mRNA cancer vaccines, siRNA therapeutics, and CRISPR-based oncology programs.
Key Developments:
Personalized neoantigen vaccines.
Delivery of gene-editing tools for tumor suppression.
LNP systems designed for targeted tumor microenvironment penetration.
Market Outlook:
Increasing collaborations between oncology platforms and LNP suppliers.
Demand for next-generation ionizable lipids with tumor-specific release properties.
b. Infectious Diseases (>47% Share in 2023)
Dominance Reason:
Strong demand driven by mRNA vaccine development for COVID-19, influenza, RSV, HIV, and emerging pathogens.
Applications:
Rapid-response vaccine platforms.
Pandemic preparedness programs.
Multivalent mRNA vaccines.
Market Outlook:
Governments and health bodies investing heavily into LNP-based vaccine stockpiling.
Growth supported by global mRNA manufacturing infrastructure expansion.
c. Blood Diseases
Applications:
Gene-replacement therapies for hemophilia, beta-thalassemia, sickle cell disease.
Delivery of CRISPR components to hematopoietic stem cells.
Market Outlook:
Growing number of clinical trials using LNPs for ex vivo and in vivo gene correction.
Increasing partnerships between biotech firms and LNP raw material providers.
d. Others (Metabolic, Genetic & Rare Disorders)
Includes diseases such as:
Metabolic enzyme deficiencies.
Neuromuscular diseases.
Lysosomal storage disorders.
Rare monogenic disorders.
Market Outlook:
Expansion driven by breakthroughs in in vivo gene editing.
High unmet medical needs attracting orphan drug funding.
3. Application Segment
The LNP raw materials market is split between therapeutic use and research applications, driven by the global growth of RNA and gene-modifying technologies.
a. Therapeutics (>63% Share)
Major Uses:
mRNA therapeutics and vaccines.
siRNA drug delivery.
CRISPR/Cas gene editing.
Self-amplifying RNA (saRNA) programs.
Demand Drivers:
Increasing commercialization of RNA-based therapies.
Expansion of GMP manufacturing facilities by Moderna, BioNTech, Pfizer, and emerging RNA companies.
Shift toward LNPs as the preferred non-viral delivery method for genetic cargo.
Market Outlook:
Most lucrative segment due to rapid clinical translation.
Higher regulatory standards drive premium pricing for high-purity raw materials.
b. Research (Fastest Growing Segment)
Applications:
Preclinical development of RNA platforms.
Toxicity and pharmacokinetic studies.
Screening of lipid compositions for custom LNPs.
Demand Drivers:
Increasing academic and biotech research in nanomedicine.
Surge in government and private funding for RNA technology innovation.
Growth of contract research organizations (CROs) offering LNP-based services.
Market Outlook:
Expected to grow faster than therapeutics due to:
Strong R&D pipelines.
Ease of adoption of LNP kits and ready-mix reagents.
Expansion of early-stage gene therapy research.
Top 5 FAQs
1. What is the projected market size of the LNP raw materials market?
The market is projected to grow from USD 300.72 million (2025) to USD 493.99 million by 2034 at a 5.67% CAGR.
2. Which region dominated the market?
North America dominated with 41% market share in 2023, driven by strong R&D investments.
3. Which segment led by product category?
The kits segment led with 55% revenue share in 2023.
4. Which disease indication contributed the most in 2023?
Infectious diseases, contributing >47% market share, mainly due to mRNA vaccine production.
5. Who are the key companies involved?
Major players include Polysciences, NOF America, CordenPharma, Avanti Polar Lipids, Merck KGaA, Cytiva, Hopewell Therapeutics, WuXi STA, and others.
Access our exclusive, data-rich dashboard dedicated to the biotechnology sector – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout Lipid Nanoparticle Raw Materials Market Report Now at: https://www.towardshealthcare.com/checkout/5168
About Us
Towards Healthcare is a leading global provider of technological solutions, clinical research services, and advanced analytics, with a strong emphasis on life science research. Dedicated to advancing innovation in the life sciences sector, we build strategic partnerships that generate actionable insights and transformative breakthroughs. As a global strategy consulting firm, we empower life science leaders to gain a competitive edge, drive research excellence, and accelerate sustainable growth.
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest