Drug Safety Gold Rush? Pharmacovigilance Market Set to Redefine Global Healthcare by 2035!
The global healthcare ecosystem is undergoing a silent but powerful transformation. Behind every newly approved drug, every vaccine rollout, and every biologic breakthrough lies an intricate system designed to protect patients from harm. That system is pharmacovigilance.
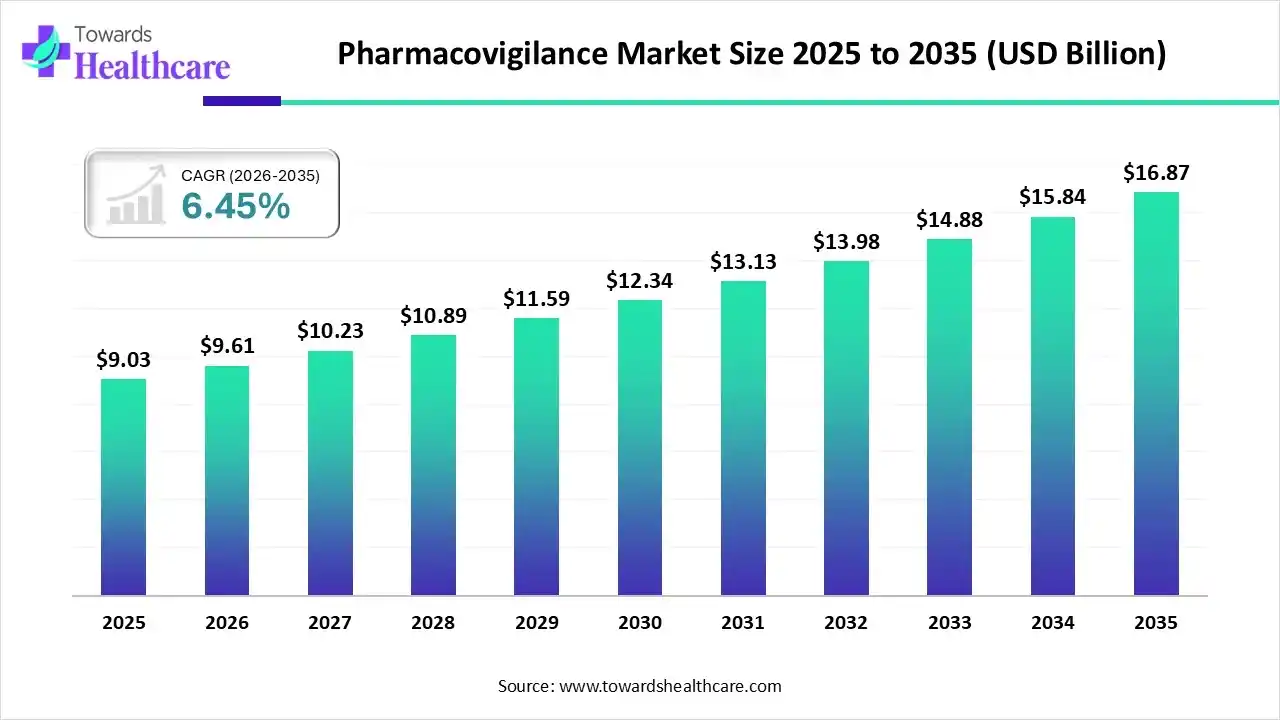
The global pharmacovigilance market was valued at USD 9.03 billion in 2025. It is projected to grow from USD 9.61 billion in 2026 to approximately USD 16.87 billion by 2035, expanding at a steady CAGR of 6.45% between 2026 and 2035. This growth reflects more than financial momentum; it signals a structural shift in how the world manages drug safety, compliance, and real-world risk monitoring.
Download Free Sample and Send Customize Request as per Your Business Goals: https://www.towardshealthcare.com/download-sample/6504
Pharmacovigilance is no longer a regulatory obligation operating quietly in the background. It is becoming one of the most strategic investment frontiers in healthcare analytics.
Table of Contents
ToggleWhy Pharmacovigilance Now Commands Strategic Attention
Pharmacovigilance refers to the science and activities involved in detecting, assessing, understanding, and preventing adverse effects or drug-related problems. In simpler terms, it ensures medicines remain safe after they reach patients.
The market is expanding for several interconnected reasons:
-
Rising cases of adverse drug reactions (ADRs)
-
Increasing complexity of biologics and advanced therapies
-
Stricter regulatory frameworks worldwide
-
Growing clinical trial volumes
-
Demand for real-time safety monitoring systems
Every newly approved therapy increases the burden of safety monitoring. In 2024 alone, the U.S. regulatory system approved 50 novel drugs. Across innovators and biosimilars, total approvals reached 127. Each approval translates into thousands of safety reports, regulatory submissions, signal detection analyses, and post-marketing surveillance activities.
More drugs mean more data. More data demands better systems. And better systems require structured pharmacovigilance.
AI Is Rewriting the Rules of Drug Safety
Artificial intelligence is no longer an experimental add-on in pharmacovigilance. It is actively reshaping operations.
AI-driven systems now:
-
Automate adverse event case intake and processing
-
Extract insights from unstructured clinical narratives using natural language processing
-
Detect safety signals earlier through machine learning
-
Analyze real-world data from electronic health records and patient registries
-
Support predictive risk modeling during clinical trials
Traditional pharmacovigilance relied heavily on manual review and spontaneous reporting. Today, AI accelerates detection, reduces human error, and improves regulatory turnaround times.
For global pharmaceutical companies managing thousands of safety cases daily, automation is no longer optional—it is operationally critical.
North America Leads; but Asia Pacific Accelerates Fastest
In 2025, North America dominated the pharmacovigilance market with a 42.5% share. The region benefits from:
-
Strong regulatory oversight led by the U.S. Food and Drug Administration (FDA)
-
Advanced digital infrastructure
-
High R&D spending
-
Widespread adoption of AI-enabled safety systems
The U.S. continues to lead due to its stringent reporting standards and high volume of clinical research activity. The regulatory environment enforces continuous safety evaluation, which sustains demand for advanced pharmacovigilance services.
However, Asia Pacific is emerging as the fastest-growing region, expanding at a CAGR of 7.8%. Countries such as India and China are strengthening regulatory reforms, increasing clinical trial participation, and attracting outsourced pharmacovigilance contracts.
Cost-efficient skilled labor, digital adoption, and expanding pharmaceutical manufacturing capacity position Asia Pacific as a long-term growth engine for the industry.
Case Processing: The Engine Room of Pharmacovigilance
Among service types, case processing dominated the market in 2025 with a 31.3% revenue share.
Case processing involves:
-
Collecting adverse event reports
-
Validating safety data
-
Assessing seriousness and causality
-
Preparing regulatory submissions
This function forms the operational backbone of pharmacovigilance. As drug consumption increases globally, safety report volumes multiply. Automation improves efficiency, but structured workflows remain essential.
Signal detection, however, is the fastest-growing segment, projected to expand at a CAGR of 6.6%. Regulators increasingly demand proactive identification of safety risks rather than reactive reporting. Companies now invest heavily in analytics platforms that identify patterns across massive datasets.
Early signal detection saves not only regulatory costs—but potentially lives.
Outsourcing Takes Center Stage
The outsourced pharmacovigilance model dominated the market in 2025 with a 72.4% share and is expected to maintain the fastest growth rate of 6.5%.
Pharmaceutical companies increasingly partner with Contract Research Organizations (CROs) and technology firms to manage:
-
Case processing
-
Literature monitoring
-
Risk management planning
-
Regulatory compliance
-
Safety database management
Outsourcing offers scalability, cost efficiency, and access to global expertise. It also enables companies to integrate AI tools without building infrastructure internally.
At the same time, in-house pharmacovigilance operations are growing steadily as companies seek greater control over sensitive safety data and strategic decision-making. The future likely belongs to hybrid models—blending internal oversight with external execution.
Phase IV Surveillance: The Long Game of Drug Safety
Post-marketing surveillance (Phase IV) led the process flow segment with a 34.5% share in 2025.
Clinical trials cannot capture every adverse effect. Rare reactions, long-term complications, and population-specific risks often emerge only after a drug reaches the broader public.
Phase IV surveillance ensures:
-
Continuous safety evaluation
-
Regulatory compliance
-
Real-world effectiveness monitoring
-
Public trust preservation
Meanwhile, Phase III is the fastest-growing process stage, with a CAGR of 6.7%. Large patient pools in late-stage trials generate extensive safety data requiring structured pharmacovigilance systems.
As trial designs become more complex and multinational, real-time safety monitoring becomes indispensable.
Biologics and Advanced Therapies Change the Equation
The rise of biologics, gene therapies, and advanced immunotherapies introduces new pharmacovigilance challenges.
Unlike small molecules, biologics often:
-
Trigger immune-mediated responses
-
Require cold-chain logistics
-
Exhibit variable patient-specific reactions
Biotechnology companies are therefore the fastest-growing end-user segment, expanding at 6.7% CAGR.
As biologics account for a growing percentage of global approvals, pharmacovigilance systems must evolve beyond traditional models. They must integrate immunogenicity monitoring, vaccine safety tracking, and gene therapy risk management frameworks.
Government Initiatives Strengthen Global Monitoring
Governments are actively strengthening drug safety frameworks.
In India, the Central Drugs Standard Control Organisation (CDSCO) mandated QR codes and toll-free ADR reporting systems across pharmacies to connect consumers directly with national reporting platforms. This initiative aims to simplify adverse drug reaction reporting under the Pharmacovigilance Programme of India (PvPI).
Similarly, global regulators are upgrading legislation, revising compliance requirements, and emphasizing digital safety reporting platforms.
These measures increase reporting rates and demand more structured pharmacovigilance infrastructure.
Drug safety is becoming a public health priority—not just a compliance obligation.
Cloud Platforms Transform Global Collaboration
Cloud-based pharmacovigilance systems are reshaping operational scalability.
Cloud platforms enable:
-
Real-time data access across regions
-
Secure safety database hosting
-
Global regulatory submission management
-
Faster signal detection workflows
For multinational pharmaceutical companies running simultaneous trials across continents, cloud systems eliminate fragmentation.
They also reduce infrastructure costs while enabling seamless compliance tracking.
The transition from legacy systems to cloud-based PV platforms represents a structural industry shift.
Real-World Evidence Becomes a Strategic Asset
Real-world evidence (RWE) now plays a pivotal role in pharmacovigilance.
Safety data flows from:
-
Electronic health records
-
Patient registries
-
Wearable devices
-
Insurance claims databases
-
Social media platforms
RWE strengthens signal detection, supports regulatory decisions, and enhances long-term safety evaluations.
As regulators increasingly accept RWE for safety assessment, pharmacovigilance teams must integrate structured analytics tools capable of processing massive, heterogeneous datasets.
The Competitive Landscape: Technology Meets Compliance
Leading players in the pharmacovigilance market include:
-
IQVIA Holdings Inc.
-
Parexel International Corporation
-
Accenture PLC
-
ICON plc
-
Cognizant Technology Solutions
-
ArisGlobal LLC
-
Thermo Fisher Scientific
-
Genpact Limited
-
Capgemini Services SAS
These companies combine clinical research expertise, AI integration, digital transformation services, and regulatory consulting capabilities.
Competition increasingly revolves around automation capabilities, AI-enabled signal detection, and scalable global delivery models.
Pharmacovigilance is no longer just about reporting—it is about predictive analytics, intelligent workflows, and digital compliance ecosystems.
Strengths, Risks, and the Road Ahead
Strengths
-
Strong global regulatory frameworks
-
High awareness of drug safety
-
AI-driven efficiency gains
-
Expanding clinical trial ecosystem
Weaknesses
-
High operational costs
-
Skilled workforce shortages
-
Data fragmentation
-
Manual processes in developing regions
Opportunities
-
Outsourced PV expansion
-
Cloud-based platforms
-
Growth in emerging markets
-
Increasing biologics pipeline
Threats
-
Cybersecurity risks
-
Regulatory volatility
-
Price competition
-
Reporting delays in fragmented systems
Despite operational challenges, structural growth drivers remain intact.
The Bigger Picture: Trust as the Ultimate Currency
Healthcare runs on trust. Patients trust physicians. Physicians trust clinical evidence. Regulators trust safety data.
Pharmacovigilance protects that trust.
As global drug pipelines expand and therapeutic complexity increases, pharmacovigilance transitions from a compliance function to a strategic pillar of healthcare governance.
By 2035, the market is expected to reach USD 16.87 billion. But its real value cannot be measured in revenue alone.
It lies in:
-
Prevented adverse reactions
-
Improved therapeutic outcomes
-
Faster regulatory responses
-
Data-driven decision-making
-
Strengthened public confidence
The future of pharmacovigilance will integrate AI, cloud infrastructure, real-world evidence, and cross-border regulatory harmonization.
The question is no longer whether pharmacovigilance will grow.
The real question is: Who will lead the next phase of intelligent drug safety?
As the pharmaceutical industry accelerates innovation, pharmacovigilance stands ready—not as a silent observer, but as an active guardian of global health.
Access our exclusive, data-rich dashboard dedicated to the pharmaceuticals industry – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout Pharmacovigilance Market Report Now at: https://www.towardshealthcare.com/checkout/6504
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest




