Image Credit: News-Medical
The Seasonal Vaccines Market added hundreds of millions of dollars in revenue (2025–2034) on top of a mature base, led by North America (40% share, 2024), influenza vaccines (60%), intramuscular delivery (70%), and rapid uptake of mRNA platforms across high-need geographies.
Download this Free Sample Now and Get the Complete Report and Insights of this Market Easily @ https://www.towardshealthcare.com/download-sample/6347
Market Size
➤2024 market mix (by major levers):
➤Region: North America 40%; Asia Pacific fastest growth through 2034.
➤Vaccine Type: Influenza 60%; RSV = fastest-growing.
➤Platform: Inactivated/split virion 35% (2024 lead); mRNA = fastest-growing.
➤Route: Intramuscular 70% (2024 lead); Intranasal = fastest-growing.
➤End-user: Public health agencies 45% (2024 lead); Pharmacies/retail = fastest-growing.
Revenue trajectory (2025–2034):
➤Incremental addition: “hundreds of millions” over 2025–2034, driven by RSV launches, annualized COVID-19 boosters, and broader retail vaccination.
➤Volume drivers: Larger eligible cohorts (aging, risk-comorbidities), expanded pediatric/older-adult indications, and combo-vaccine pipelines.
Mix shift implications:
➤Platform tilt to mRNA (speed, strain agility) gradually dilutes inactivated share from 35% as multivalent and combo mRNA programs scale.
➤Channel shift: Retail pharmacies capture a rising share from public programs, compressing administration costs and improving convenience.
Seasonality math checks (demand signals):
➤U.S. 2024–2025 flu testing: 3,978,954 specimens; 489,579 positives (Influenza A: 434,985; B: 54,594), underscoring recurring vaccination need.
Recent commercial catalysts (2025):
➤Zydus VaxiFlu™ trivalent launch (India) aligned with WHO recommendations.
➤Pfizer PCV20 adult launch (India) expanding pneumococcal protection.
Market Trends
➤Annual influenza backbone: Persistent 60% share supported by quadrivalent coverage, strain refresh, and employer/retail campaigns.
➤RSV breakout: Fastest growth on older-adult and maternal strategies; increased disease awareness post-pandemic.
➤mRNA acceleration: Fastest-growing platform for strain-agile updates, combo candidates (e.g., flu+COVID+RSV), and rapid scale-up.
➤Intranasal momentum: Fastest-growing route for pediatric/needle-phobic groups; mucosal immunity appeal.
➤Sustainability pivot: Shift away from egg-based processes; biodegradable/recyclable packaging and greener supply chains.
➤Retailization of vaccines: Pharmacies/retail centers = fastest-growing end-user, widening access and offloading clinics.
➤AI-assisted R&D: Antigen/adjuvant optimization; epitope prediction; synthetic biology + single-cell integration for precision design.
➤Policy tailwinds: Government-funded campaigns, faster seasonal approvals when manufacturing consistency is maintained.
➤Quality & real-world evidence (RWE): Season-specific effectiveness readouts (e.g., Germany interim VE 31% primary care / 69% secondary) guide updates.
➤Emerging-market scaling: Asia Pacific outpaces on population scale, program funding, and local manufacturing expansions.
10 Deep AI Roles & Impacts
➤Strain Forecasting & Antigen Selection: Models predict dominant strains earlier (e.g., VaxSeer concept), reducing mismatch risk and boosting VE.
➤Epitope & Adjuvant Discovery: Protein-structure + omics models prioritize B/T-cell epitopes and adjuvant pairings to raise immunogenicity.
➤mRNA Sequence Optimization: AI tunes coding regions, UTRs, codons for expression, stability, and manufacturability—shortening design-to-clinic cycles.
➤Formulation Design Space Search: Multivariate AI explores stabilizers/buffers to enhance cold-chain robustness and shelf life.
➤In-silico Toxicology & Safety Flags: Predictive safety screens shrink attrition, refining dose ranges and schedule before FIH trials.
➤Trial Site & Cohort Selection: ML targets hot-spots (season timing, positivity rates) to accelerate enrollment and capture attack rates efficiently.
➤Adaptive Trial Ops: Bayesian/AI engines re-weight arms mid-season, compressing time to interim efficacy decisions.
➤Manufacturing Yield Prediction: Predictive maintenance + process analytics stabilize fill-finish and antigen yields, cutting batch failures.
➤Real-time Pharmacovigilance: NLP on EHR/AE streams detects rare signals sooner; dynamic benefit-risk dashboards for regulators.
➤Programmatic Targeting for Uptake: Propensity models route reminders via pharmacies/retail to boost coverage in high-risk cohorts.
Regional Insights
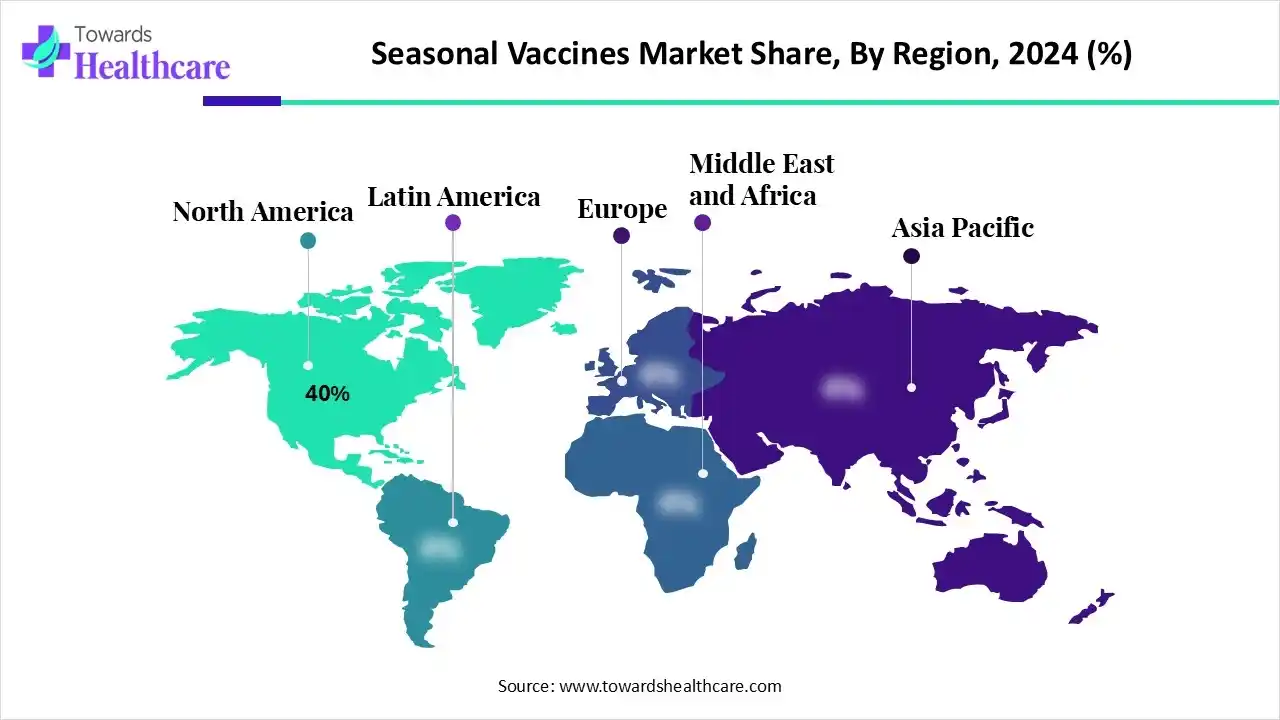
North America – 40% share (2024)
➤Access & Funding: Strong payer coverage and public stockpiles sustain high uptake; retail chains widen reach.
➤Innovation Hub: Early adoption of mRNA and RSV adult/maternal programs; combo vaccine trials active.
➤Epidemiology signals: High testing volume (4.0M specimens; 490k positives) validates recurring seasonal demand.
United States
➤Campaigns & Logistics: National flu/RSV/COVID pushes; faster approvals for strain updates maintain timely supply.
➤Channel shift: Pharmacy-led vaccinations reduce clinic burden; analytics target seniors and chronic-disease clusters.
Asia Pacific – Fastest growth
➤Scale economics: Large populations in China/India/Japan drive dose volumes; expanding domestic capacity lowers costs.
➤Program expansion: Government-backed immunization and local launches (e.g., VaxiFlu™, PCV20) accelerate adoption.
➤India signal: NCDC reports 2,400 influenza cases / 13 deaths by June 2025, prompting awareness and pediatric focus.
Europe – Robust healthcare, steady growth
➤Coverage & Policy: Insurance and public programs cover geriatric/HCW/chronic cohorts; strong cold-chain and clinic networks.
➤Effectiveness feedback: Germany 2024/25 co-circulation of A & B with 31% VE (primary care) / 69% (secondary) guides season updates.
Latin America & MEA – Emerging access
➤LATAM: Brazil/Mexico scale up public campaigns; local finishing improves availability pre-season.
➤MEA: Saudi/UAE fund adult and Hajj-related respiratory programs; South Africa strengthens lab surveillance for timing.
Market Dynamics
Drivers
➤High disease burden & aging: Recurrent influenza and rising geriatric share expand annual eligible populations.
➤Awareness & public programs: Public health agencies (45% share, 2024) fund high-risk cohorts; campaigns lift coverage.
➤Platform agility: mRNA = fastest-growing enables rapid strain refresh and combo development (flu/RSV/COVID).
Restraints
➤Seasonal mismatch risk: Antigenic drift can undercut VE despite strong coverage (e.g., Germany interim VE split).
➤Cold-chain & last-mile: Intramuscular 70% reliance demands reliable logistics; intranasal options still scaling.
➤Hesitancy pockets: Misinformation depresses uptake; uneven retail legalization limits pharmacy growth in parts of APAC/MEA.
Opportunities
➤RSV runway: Fastest-growing vaccine type (older adults, maternal/pediatric strategies).
➤Retail vaccination scale: Pharmacies/retail = fastest-growing end-user—shorter queues, evening/weekend access.
➤Sustainable manufacturing: Move off egg-based; adopt recyclable packaging; lower footprint attracts tenders.
Challenges
➤Manufacturing switchover cadence: Scaling mRNA while maintaining inactivated (35%) supply parity.
➤Regulatory timing: Tight strain-decision → release windows each year.
➤Equity & affordability: Ensuring Asia Pacific and LATAM/MEA access during peak seasons.
Recent Developments
➤Zydus Lifesciences (Sep 2025): VaxiFlu™ TIV launched in India, aligned to WHO seasonal guidance.
➤Pfizer (Aug 2025): PCV20 launch for adults in India broadens pneumococcal protection.
➤AI milestone (Aug 2025): VaxSeer (MIT CSAIL + Jameel Clinic) showcases AI-driven protective candidate and flu-strain prediction.
Top Leading Companies 2025
1. AstraZeneca plc
Strengths:
Strong R&D pipeline in viral vector and intranasal vaccines (e.g., AZD1222 platform).
Global distribution network across >100 countries.
Strategic collaborations with Oxford University and emerging-market manufacturers.
Weaknesses:
Limited presence in mRNA platforms compared to Pfizer and Moderna.
Past supply and safety perception issues during early COVID-19 vaccine rollouts.
Over-reliance on contract manufacturing partnerships.
Opportunities:
Expansion into RSV and next-gen flu boosters.
Potential to combine adenoviral and protein-based technologies.
Demand in low- and middle-income countries for affordable vaccines.
Threats:
Competition from mRNA-based developers.
Regulatory scrutiny over viral vector technologies.
Supply chain dependencies on third-party manufacturers.
2. CSL Seqirus
Strengths:
One of the world’s largest influenza vaccine producers.
Advanced cell-based and adjuvanted vaccine technologies (e.g., Flucelvax, Fluad).
Strong presence in U.S., Europe, and Asia-Pacific seasonal vaccine markets.
Weaknesses:
Product line mainly focused on influenza, limited diversification.
Dependence on seasonal demand cycles.
High manufacturing cost of cell-based vaccines.
Opportunities:
Growing adoption of cell-based production for improved strain matching.
Expansion into mRNA and combination respiratory vaccines.
Partnerships with AI developers for strain selection optimization.
Threats:
Fierce competition from Sanofi, GSK, and Pfizer.
Price pressures from public health procurement programs.
Supply fluctuations due to egg shortages or pandemic disruption.
3. Novavax Inc.
Strengths:
Proprietary Matrix-M adjuvant technology enhances immunogenicity.
Expertise in recombinant nanoparticle vaccines.
Strong R&D progress in COVID-19 and RSV combination candidates.
Weaknesses:
Limited global manufacturing network compared to larger peers.
Financial instability and high dependency on government funding.
Delayed regulatory approvals for certain markets.
Opportunities:
Potential breakthrough with flu-RSV-COVID combination vaccines.
Strategic licensing or partnerships with large pharma firms.
Expanding footprint in low-income countries through technology transfer.
Threats:
Competition from mRNA players with faster platform adaptability.
Market consolidation reducing standalone opportunities.
Investor skepticism affecting long-term R&D funding.
4. Johnson & Johnson (Janssen Pharmaceuticals)
Strengths:
Established adenovirus vector vaccine platform (Ad26).
Broad global manufacturing footprint and logistics.
Robust financial stability and diversified pharma portfolio.
Weaknesses:
Limited ongoing innovation in influenza and RSV vaccines.
Declining public confidence post-COVID-19 vaccine phase-out.
Smaller vaccine business compared to pharma segment.
Opportunities:
Expansion into combination respiratory vaccines.
Potential application of Ad26 platform for future seasonal variants.
Strategic collaborations with AI partners for vaccine design.
Threats:
Intense competition in respiratory vaccine segment.
Shifting regulatory priorities toward mRNA-based products.
Reduced revenue visibility post-pandemic.
5. Bharat Biotech
Strengths:
Strong presence in emerging markets with cost-effective vaccines.
Proven manufacturing capabilities (e.g., Covaxin, influenza formulations).
Government collaborations for public immunization programs in India.
Weaknesses:
Limited presence in Western markets due to slower regulatory approvals.
R&D resources lower than multinational competitors.
Relatively smaller marketing and distribution network.
Opportunities:
Expansion into global seasonal influenza and pediatric RSV vaccines.
Export potential through WHO prequalification.
Investment in nasal and intradermal vaccine innovations.
Threats:
Competition from Serum Institute of India and multinational players.
Volatile raw material pricing and cold-chain challenges.
Regulatory and compliance hurdles in developed markets.
6. Serum Institute of India Pvt. Ltd.
Strengths:
World’s largest vaccine manufacturer by volume.
High production capacity for influenza, pneumococcal, and COVID-19 vaccines.
Strategic partnerships with AstraZeneca, Novavax, and WHO.
Weaknesses:
Dependence on licensing and technology transfer from global innovators.
Limited original R&D pipeline.
Heavy reliance on low-margin public sector contracts.
Opportunities:
Expansion into mRNA and next-gen RSV vaccines.
Growth in Africa and Southeast Asia through GAVI and UNICEF programs.
Vertical integration in cold-chain logistics and packaging.
Threats:
Increasing competition from Chinese and Korean manufacturers.
Regulatory delays in developed market entries.
Supply chain volatility affecting exports.
7. Sinovac Biotech Ltd.
Strengths:
Proven expertise in inactivated vaccine technology.
Strong domestic market share in China.
Rapid scalability and cost-effective production.
Weaknesses:
Limited innovation in advanced platforms like mRNA or recombinant proteins.
Geopolitical and trade-related barriers in Western markets.
Transparency and data credibility concerns.
Opportunities:
Expansion into developing markets in Asia, Latin America, and Africa.
Development of inactivated RSV and influenza boosters.
Collaboration with biotech firms for new delivery systems.
Threats:
Competition from Sinopharm and international vaccine leaders.
Decreasing demand for inactivated COVID-19 vaccines.
Regulatory scrutiny in foreign markets.
8. Sinopharm Group Co. Ltd.
Strengths:
Backed by Chinese government; robust funding and infrastructure.
Broad vaccine portfolio and advanced R&D network.
High-volume production of COVID-19 and influenza vaccines.
Weaknesses:
Dependence on domestic market for revenue.
Limited adoption in Western countries.
Slower innovation compared to mRNA competitors.
Opportunities:
Expansion through Belt and Road vaccine diplomacy.
Entry into Africa and Latin America via supply partnerships.
Investment in next-gen technologies (mRNA and recombinant platforms).
Threats:
Political tensions restricting global access.
Global competition in low-cost vaccine supply.
Rising domestic competition from emerging Chinese biotech firms.
9. Daiichi Sankyo Company, Limited
Strengths:
Strong research capabilities in adjuvanted and recombinant vaccines.
Strategic presence in Japan and international collaborations.
Long-term investment in mRNA technology platforms.
Weaknesses:
Smaller vaccine division compared to its pharmaceutical arm.
High production cost structure.
Limited exports beyond Asia.
Opportunities:
Development of universal flu and mRNA-based RSV vaccines.
Joint ventures with Western partners to globalize portfolio.
Growth from government immunization funding in Japan.
Threats:
Competitive pressure from GSK, Sanofi, and CSL in Asia-Pacific.
Regulatory hurdles for novel vaccine approvals.
Market saturation in Japan’s domestic vaccine sector.
10. Mitsubishi Tanabe Pharma Corporation
Strengths:
Strong presence in Japanese immunization programs.
Expertise in biotechnology and cell culture-based vaccine production.
Strategic alliances with research universities and biotech startups.
Weaknesses:
Limited global market footprint.
Narrow vaccine product portfolio.
Lower production capacity compared to global peers.
Opportunities:
Expansion into Asia-Pacific with influenza and COVID-19 boosters.
Collaborations in AI-driven vaccine optimization.
Entry into pediatric and geriatric vaccine segments.
Threats:
Domestic competition from Daiichi Sankyo and foreign entrants.
Pricing pressure in Japan’s government-controlled health system.
Rapid innovation in mRNA space threatening traditional platforms.
11. Bavarian Nordic
Strengths:
Specialization in viral vector and recombinant vaccines.
Strong reputation in smallpox and monkeypox immunization.
Expanding portfolio into influenza and RSV using proprietary MVA-BN platform.
Weaknesses:
Relatively small-scale production capacity.
High manufacturing costs for vector-based vaccines.
Limited penetration outside Europe and North America.
Opportunities:
Development of multivalent respiratory vaccines.
EU funding support for pandemic preparedness.
Partnerships for large-scale seasonal vaccine rollout.
Threats:
Strong competition from mRNA and recombinant platforms.
High R&D expenses with uncertain commercial returns.
Complex regulatory landscape for novel viral vector technologies.
12. Valneva SE
Strengths:
Diverse vaccine portfolio including Lyme disease, chikungunya, and flu candidates.
Proven expertise in inactivated and adjuvanted vaccines.
Strong R&D collaborations with Pfizer and CEPI.
Weaknesses:
Limited commercial infrastructure outside Europe.
Financial dependence on licensing and grants.
Smaller pipeline compared to top-tier peers.
Opportunities:
Entry into seasonal influenza and travel-related vaccines.
Potential EU and U.S. approvals for new candidates.
Co-development opportunities with pharma giants.
Threats:
Competitive intensity in emerging vaccine fields.
Regulatory delays impacting revenue timelines.
Manufacturing scale-up risks.
13. BioNTech SE
Strengths:
Pioneer in mRNA technology; co-developed Comirnaty with Pfizer.
Strong AI and bioinformatics capabilities for vaccine optimization.
Expanding pipeline in flu, RSV, malaria, and shingles.
Weaknesses:
Heavy dependence on Pfizer collaboration for manufacturing and distribution.
Limited independent global infrastructure.
High R&D expenditure with short-term profit volatility.
Opportunities:
Development of multi-pathogen mRNA vaccines (flu + RSV + COVID).
Penetration into oncology and infectious disease cross-platform synergies.
Partnerships with developing nations for localized production.
Threats:
Competitive innovation race with Moderna and GSK.
Market saturation in post-pandemic COVID-19 boosters.
Regulatory complexities in multi-pathogen vaccine trials.
14. Vaxine Pty Ltd
Strengths:
Australian biotech specializing in recombinant and adjuvanted vaccines.
Proprietary Advax adjuvant platform enhances immune response.
Recognized for rapid-response vaccine development capacity.
Weaknesses:
Small-scale manufacturer with limited commercialization channels.
Low brand recognition globally.
Funding constraints limiting expansion.
Opportunities:
Licensing of Advax adjuvant to global partners.
Entry into influenza and RSV vaccine collaborations.
Government and CEPI grants for pandemic preparedness.
Threats:
Dependence on grant-based funding.
Competition from large biotech firms with mRNA platforms.
Regulatory and logistical challenges in scaling production.
15. SK Bioscience Co., Ltd.
Strengths:
Strong South Korean manufacturer with advanced cell culture and recombinant platforms.
Partnerships with AstraZeneca, GSK, and Novavax.
Robust government support and domestic vaccination infrastructure.
Weaknesses:
Limited brand visibility outside Asia.
Reliance on contract manufacturing for global partners.
Still expanding independent R&D capabilities.
Opportunities:
Development of self-branded influenza and RSV vaccines.
Expansion into ASEAN and Middle East markets.
Collaboration with AI firms for vaccine design optimization.
Threats:
Pricing competition with Indian and Chinese suppliers.
Volatile global demand post-pandemic.
Dependency on licensing agreements for key revenue streams.
Segments Covered in the Seasonal Vaccines Market (In-depth)
1. By Vaccine Type
Influenza Vaccines (60% share, 2024):
➤Dominant segment driven by annual flu prevention programs and WHO-recommended strain updates.
➤Includes Trivalent (TIV) and Quadrivalent (QIV) formulations for broader strain coverage.
➤Rising production capacity and public awareness boost seasonal uptake.
RSV (Respiratory Syncytial Virus) Vaccines (Fastest-growing):
➤Strong demand among older adults, infants, and maternal health programs.
➤Boosted by newly approved vaccines and ongoing global rollout campaigns.
Pneumococcal Vaccines:
➤Seasonal/annual boosters for adults; protection against pneumonia and invasive disease.
➤Driven by rising geriatric immunization coverage and newer 20-valent (PCV20) formulations.
COVID-19 Booster/Annualized Vaccines:
➤Integrated into seasonal vaccination schedules for high-risk groups.
➤mRNA-based combinations with flu and RSV are under development.
Others (e.g., Meningococcal, Tick-borne Encephalitis):
➤Address niche or regional pathogens with cyclical infection patterns.
2. By Technology Platform
Inactivated/Split Virion Vaccines (35% share, 2024):
➤Most widely used for influenza; safe for geriatric, pediatric, and pregnant populations.
➤Stable storage, long shelf life, and established manufacturing infrastructure.
Live Attenuated Vaccines:
➤Nasal formulations gaining popularity for children and needle-averse adults.
➤Stimulate mucosal immunity and strong antibody response.
Recombinant Protein Vaccines:
➤Egg-free production and improved antigenic match increase acceptance.
➤Lower allergenic risks and faster scalability.
mRNA-based Vaccines (Fastest-growing):
➤Enable rapid strain updates, multi-pathogen combinations, and high efficacy.
➤Central to next-generation influenza, RSV, and COVID-19 programs.
DNA-based Vaccines:
➤Early-stage innovations offering easy manufacturing and thermostability.
Vector-based Vaccines:
➤Use viral vectors for durable immunity; under trial for RSV and COVID boosters.
Others:
➤Include nanoparticle, peptide, and self-amplifying RNA (saRNA) technologies in development.
3. By Route of Administration
Intramuscular (IM) – 70% share, 2024:
➤Gold-standard method offering consistent absorption and strong systemic immunity.
➤Widely used across hospitals, clinics, and retail pharmacies.
Subcutaneous:
➤Used for select live vaccines; preferred in pediatrics due to lower injection pain.
Intranasal (Fastest-growing):
➤Needle-free delivery improving compliance; targets mucosal immunity in respiratory infections.
Oral:
➤Under development for next-gen polio and rotavirus formulations; convenient but stability-sensitive.
Others (e.g., Transdermal Patches):
➤Innovative, self-administered systems improving accessibility and logistics in remote areas.
4. By End User
Public Health Agencies (45% share, 2024):
➤Bulk procurement and subsidized programs for mass immunization campaigns.
➤Focus on high-risk populations, including elderly, infants, and healthcare workers.
Hospitals & Immunization Clinics:
➤Core administration centers for influenza, pneumococcal, and RSV vaccines.
➤Equipped for cold-chain management and large-volume storage.
Pharmacies & Retail Vaccination Centers (Fastest-growing):
➤Expansion driven by convenience, extended hours, and walk-in services.
➤Increasingly integrated into national immunization systems.
Ambulatory Care Centers:
➤Growing role in outpatient vaccination; connected with digital scheduling and reminders.
Others (Corporate/Institutional Programs):
➤Workplace vaccination drives enhancing employee safety and compliance.
5. By Region
North America (40% share, 2024):
➤Mature infrastructure, high awareness, and strong retail distribution network.
➤U.S. leads global vaccine R&D, manufacturing, and adoption.
Europe:
➤Advanced public health programs, strong insurance support, and high vaccination rates.
➤Notable focus on elderly and healthcare worker immunization.
Asia Pacific (Fastest-growing):
➤Expanding population base, government immunization programs, and affordable vaccine launches.
➤India and China lead regional volume growth.
Latin America:
➤Emerging adoption, supported by WHO and PAHO immunization initiatives.
➤Brazil and Mexico focus on universal flu and COVID-19 boosters.
Middle East & Africa (MEA):
➤Strengthening through adult immunization campaigns and regional disease surveillance.
➤Increasing investments in cold-chain infrastructure and vaccine manufacturing.
Top 5 Searched FAQs
1) What is driving the Seasonal Vaccines Market growth (2025–2034)?
●Rising influenza and RSV incidence.
●Expansion of public vaccination programs.
●Growing use of mRNA technology for faster updates.
●Increased retail and pharmacy vaccination access.
●Aging population and broader immunization coverage.
2) Which segments led the market in 2024?
●Region: North America – 40% share.
●Vaccine Type: Influenza – 60% share.
●Platform: Inactivated/Split Virion – 35% share.
●Route: Intramuscular – 70% share.
●End User: Public Health Agencies – 45% share.
3) Which segments are growing fastest?
●Vaccine Type: RSV vaccines.
●Platform: mRNA-based vaccines.
●Route: Intranasal delivery.
●End User: Pharmacies & Retail Centers.
●Region: Asia Pacific.
4) How is AI transforming the Seasonal Vaccines Market?
●Predicts dominant flu strains.
●Optimizes antigen and adjuvant design.
●Enhances mRNA sequence and formulation.
●Improves trial design and manufacturing efficiency.
●Enables real-time safety monitoring and coverage targeting.
5) What are the key recent developments in 2025?
●Zydus Lifesciences: Launched VaxiFlu™ trivalent flu vaccine in India.
●Pfizer: Released PCV20 for adult pneumococcal protection.
●MIT & Jameel Clinic: Introduced AI tool VaxSeer for flu strain prediction.
Access our exclusive, data-rich dashboard dedicated to the pharmaceuticals sector – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Immediate Delivery Available | Checkout this Premium Research Report @ https://www.towardshealthcare.com/checkout/6347
About Us
Towards Healthcare is a leading global provider of technological solutions, clinical research services, and advanced analytics, with a strong emphasis on life science research. Dedicated to advancing innovation in the life sciences sector, we build strategic partnerships that generate actionable insights and transformative breakthroughs. As a global strategy consulting firm, we empower life science leaders to gain a competitive edge, drive research excellence, and accelerate sustainable growth.
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Web: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest