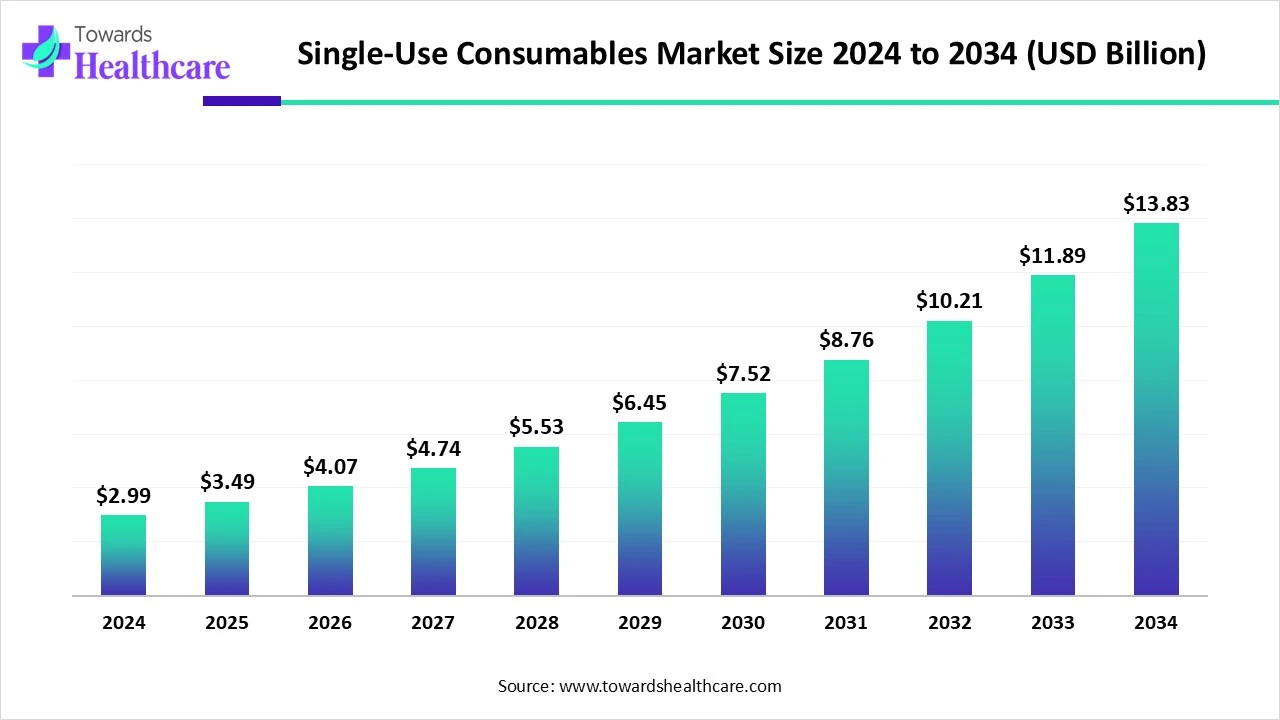
The global single-use consumables market, valued at USD 2.99 billion in 2024, is projected to grow to USD 13.83 billion by 2034 at a robust CAGR of 16.56%, driven by infection control needs, biopharma expansion, and demand for sterile, disposable medical products.
Download the free sample and get the complete insights and forecasts report on this market @ https://www.towardshealthcare.com/download-sample/6070
Market Size
◉2024 → USD 2.99 billion (baseline year).
◉2025 → USD 3.49 billion, early acceleration.
◉2030 (Mid-Term Projection) → ~USD 8.09 billion (strong growth as biopharma capacity expands).
◉2034 (Forecast Horizon) → USD 13.83 billion, over 4.6x expansion from 2024.
◉CAGR → 16.56% (2025–2034), one of the fastest growth rates in the medtech consumables sector.
Revenue Contribution by Region (2024):
◉North America → 36–40%
◉Europe → ~25–28%
◉Asia-Pacific → ~22–25% (fastest-growing)
◉Latin America & MEA → ~10–15% combined.
High-Growth Product Segments (2034 projection):
◉Catheters & vascular access → fastest CAGR.
◉Infection control & PPE → largest share in 2024, steady growth.
Material Share (2024):
◉Plastics & polymers → 55% (dominant).
◉Advanced dressings → fastest CAGR.
Market trends
Infection prevention institutionalized
◉PPE & infection-control products held 30% share in 2024—now embedded into routine clinical ops beyond pandemic spikes, reinforced by strict regulatory standards and HAI mitigation mandates.
Biologics & vaccines reshape throughput
◉The biopharma build-out (vaccines, mAbs, cell & gene therapies) favors single-use sterility and fast changeovers (e.g., tubing sets, bags, filters, preassembled kits), lifting recurring consumable demand.
Material innovation—pragmatic sustainability
◉Plastics & polymers remain dominant (55% in 2024) for sterility, cost, and manufacturability.
◉Biodegradable/compostable and low-allergen innovations (e.g., accelerator-free nitrile) gain traction as waste and skin-safety pressures rise.
Provider purchasing is unbundling
◉Direct & GPO contracts (55%) still anchor volume in 2024, but online/B2B marketplaces are the fastest-growing, enabling price transparency, long-tail SKUs, and JIT replenishment.
Pricing models modernize
◉Per-unit purchasing dominates today; subscription & managed inventory models accelerate to reduce stockouts, smooth cashflow, and tie replenishment to actual procedure volumes.
Network scale matters
◉Regional DC expansion (e.g., Cardinal Health’s 2024 Ohio distribution center intent) underscores the premium on fill-rate reliability, last-mile speed, and GxP-compliant traceability.
North America leads; APAC scales fastest
◉NA’s 36–40% share stems from advanced infrastructure and regulatory rigor; APAC outpaces on growth via capex in biomanufacturing and an expanding CMO/CDMO base.
AI impact/role
Demand & procedure forecasting
◉ML models forecast SKU-level demand (gloves, masks, syringes, catheters) from surgical schedules, census data, seasonality, and outbreak signals—reducing stockouts and costly overstock.
Smart inventory & PAR optimization
◉AI tunes PAR levels by unit/ward, maps ABC/XYZ item criticality, and triggers auto-replenishment; aligns consignment and trunk-stock to real utilization.
Computer vision quality control
◉Real-time vision models flag micro-defects (microtears, lumen occlusion, tip deformities, contamination) in catheters, IV sets, and gloves, tightening AQL and sterility assurance.
Lot genealogy & traceability
◉AI reconciles batch/lot/device identifiers across ERP + WMS + MES, improving recall readiness, expiry control, and UDI compliance.
Supply-chain risk sensing
◉Models detect upstream resin/film/tubing risks, DC bottlenecks, and carrier disruptions; autonomously re-routes POs, shifts demand to approved alternates, and rebalances safety stock.
Dynamic pricing & contracting
◉AI simulates per-unit vs. subscription cost curves by historical burn rates; proposes tiered bundles (e.g., procedure kits) to lower total cost per case while meeting compliance.
Sustainability analytics
◉Lifecycle and waste analytics pinpoint swap-in SKUs (e.g., biodegradable drapes in non-critical settings), quantify waste-disposal savings, and model CO₂e reductions.
Regulatory document automation
◉NLP agents classify sterility/biocompatibility dossiers, map them to FDA/EMA/ISO sections, and flag gaps for faster audit readiness.
E-commerce personalization
◉Recommenders build facility-specific catalogs, auto-substitute equivalent SKUs during backorders, and pre-build procedure packs tuned to case-mix.
Workforce enablement
◉Copilots surface IFUs, donning/doffing checklists, and lot/expiry alerts at POC (point of care), improving compliance and reducing errors.
Regional insights
North America (Leading, 36–40% in 2024)
Why it leads
◉Advanced hospital networks, high surgical volumes, mature GPO ecosystems, and strict infection-control enforcement.
Market energy
◉Strong CMO/CDMO presence and biologics pipelines → high pull for sterile, single-use components.
Distribution muscle
◉Ongoing DC expansions to tighten lead times and inventory reliability; direct & GPO contracts dominate today.
Europe
Compliance-driven steadiness
◉Stringent HAI standards and procurement oversight sustain demand for PPE, sterile drapes, vascular access, and safety syringes.
Material shift
◉Elevated interest in sustainable textiles and polymers in non-critical settings; plastic-reduction initiatives shape SKU choices.
Asia-Pacific (Fastest growth)
Industrial build-out
◉Accelerating biopharma manufacturing (China, India, South Korea, Japan) drives sterile single-use adoption across upstream/downstream steps.
Domestic champions
◉Growth of local manufacturing (e.g., gloves, dressings, tubing) shortens supply lines and lowers landed cost.
Care delivery
◉Rapid expansion of ASCs/outpatient care boosts demand for procedure-specific kits and non-sterile patient-care consumables.
Latin America & Middle East/Africa
LatAm
◉Brazil/Mexico lead on hospital capacity growth; uptake paced by budget cycles and import dynamics.
MEA
◉KSA/UAE invest in advanced care (oncology, CV), lifting sterile consumable usage; South Africa anchors regional adoption.
Market dynamics
Drivers
◉CMO/CDMO expansion → multiproduct, small-batch agility needs sterile, fast-changeover single-use (no cleaning validation burden).
◉Example: Akums Drugs & Pharmaceuticals (2025): new sterile lyophilized/injectables facility in India → high reliance on single-use to ensure sterility and adaptability.
◉Vaccine & biologics pipelines → mRNA, viral-vector, protein vaccines scale faster with single-use assemblies.
◉Example: Pfizer (2025) mRNA expansion (influenza, RSV), leveraging single-use intensive bioprocessing.
◉Infection control engrained in policy & practice → PPE, sterile drapes, safety syringes remain structural demand.
Restraints
◉Environmental impact & disposal costs → regulatory and financial pressure to curb plastic waste; push for greener materials.
◉Medical waste management capacity → incineration and segregation constraints elevate total cost of ownership for disposables.
Opportunities
◉Sustainable polymers & advanced dressings → growth runway for biodegradable/compostable options and bioactive/antimicrobial dressings.
◉Digitized procurement → fastest growth via online/B2B marketplaces and integrated inventory services.
◉Commercial innovation → managed inventory / subscription bundles reduce stockouts and smooth budgeting; high appeal to ASCs/OPDs.
Top companies

3M
◉Products: Infection-control/PPE (masks, respirators), surgical drapes & barrier materials.
◉Overview: Global materials & medical solutions leader.
◉Strengths: Filtration/barrier science, large-scale manufacturing, brand trust in infection control.
Medline Industries
◉Products: Broad single-use portfolio—PPE, drapes, trays, patient-care disposables.
◉Overview: Vertically integrated manufacturer/distributor to providers.
◉Strengths: Depth of catalog, private-label scale, logistics reach in U.S. hospitals.
Cardinal Health
◉Products: PPE, surgical consumables; large medical distribution.
◉Overview: Major U.S. med-surg distributor with owned brands.
◉Strengths: GPO relationships, DC network efficiency; 2024 plan for Walton Hills, OH distribution expansion underscores fulfillment muscle.
Becton, Dickinson & Co. (BD)
◉Products: Syringes, needles, safety devices; vascular access.
◉Overview: Leader in medication delivery & safety.
◉Strengths: Safety IP, manufacturing scale, clinical workflow integration.
Baxter International
◉Products: IV sets, infusion consumables, sterile solutions.
◉Overview: Hospital therapy & infusion specialist.
◉Strengths: Installed base in acute care, therapy-set standardization.
B. Braun Melsungen
◉Products: Catheters, infusion, vascular access systems.
◉Overview: Procedural & infusion therapy focus.
◉Strengths: Vascular expertise, clinician trust, EU footprint.
Kimberly-Clark Health / Kimtech
◉Products: Gloves, masks, gowns, lab apparel.
◉Overview: PPE & hygiene specialist.
◉Strengths: Non-woven know-how, comfort/fit innovation, sustainability initiatives.
Ansell
◉Products: Medical & surgical gloves.
◉Overview: Hand-protection leader.
◉Strengths: Material science, allergy-reduction focus, breadth of glove portfolio.
Terumo Corporation
◉Products: IV/infusion sets, catheters, access devices.
◉Overview: Vascular & interventional specialist.
◉Strengths: Precision manufacturing, cardiovascular credibility.
Smiths Medical
◉Products: Catheters, infusion disposables, airway management items.
◉Overview: Acute care & critical-care disposables.
◉Strengths: Clinical breadth, integration with monitoring/therapy workflows.
ConvaTec / Coloplast / Mölnlycke Health Care
◉Products: Advanced wound dressings, ostomy & continence care; surgical drapes/gowns.
◉Strengths: Wound-healing science, skin integrity, OR barrier systems.
Owens & Minor / Henry Schein
◉Products: Wide med-surg disposables; dental/clinic channels (H. Schein).
◉Strengths: Distribution depth, practice-level relationships.
Fresenius Kabi / Teleflex / Cook Medical / Nipro Medical
◉Products: IV therapy sets, vascular access, specialty catheters and kits.
◉Strengths: Therapy specialization, kit customization, global reach.
Investment & Funding Insights In Single-Use Consumables Market
Market funding thesis — why investors care
◉Large, recurring demand: Consumables are repeat-purchase products with predictable recurring revenue (P/RR), driven by routine procedures, PPE use, and steady hospital throughput.
◉High gross margins on specialty items: Advanced dressings, procedure kits, and specialty catheters often command higher margins than commodity plastics.
◉Resilience to economic cycles: Clinical necessity (infection control, surgeries, chronic-care consumables) makes cashflows relatively inelastic.
◉Scalability via CMOs/CDMOs & contract manufacturing: Manufacturers can scale capacity quickly via contract partners, reducing time-to-market.
◉Regulatory and quality moat: Proven sterility, biocompatibility testing and approvals (GMP, ISO, CE, FDA) create barriers to entry.
◉Innovation pathways: Sustainable polymers, accelerator-free gloves, single-use endoscopes, and systemized procedure kits create product differentiation and premium positioning.
◉Tech-enabled operational uplift: AI for demand forecasting, QA, and inventory management reduces working capital and shrinkage — attractive to investors seeking operational leverage.
Who invests and typical instruments
Strategic corporate investors (corporates & PE-backed strategics)
◉Motive: market share, proprietary SKUs, supply-chain control, fill capacity (e.g., 3M, Cardinal Health initiatives).
◉Instrument: M&A, bolt-on acquisitions, JV, equity stakes.
Private Equity (buyout / growth)
◉Motive: consolidate fragmented regional manufacturers/distributors, optimize operations, roll-up strategies, scale distribution (GPO access).
◉Instrument: Majority buyouts, growth equity, earn-outs.
Venture Capital / Growth Equity
◉Motive: early-stage material innovation (biodegradable polymers), advanced dressings, single-use endoscope startups, AI-enabled supply platforms.
◉Instrument: Series A–C equity, convertible notes, SAFE (early-stage).
Debt / Project Finance
◉Motive: capex for high-speed manufacturing lines, plant expansions (e.g., mask lines).
◉Instrument: Term loans, vendor financing, asset-backed loans, export credit for cross-border capex.
Public markets / PIPEs
◉Motive: Established manufacturers may access public equity for large capex or M&A. PIPEs used for growth-stage raises.
Government grants & concessional finance
◉Motive: local manufacturing incentives (vaccine/biopharma capacity building), supply-chain resilience funds.
◉Instrument: Grants, subsidized loans, capex rebates (relevant in NA/APAC policy pushes).
Typical capital requirements / use of proceeds (by stage)
Early-stage (R&D / material tech / prototypes)
◉Use: lab R&D, polymer formulation, biocompatibility testing, first ISO/FDA compliance tests.
◉Typical raise: seed to Series A (small relative to manufacturing).
Scale-up (commercialization / market entry)
◉Use: pilot production, clinical evaluations, sales & limited distribution, regulatory submissions.
◉Typical raise: Series A–B or growth equity.
Manufacturing & distribution expansion (capex heavy)
◉Use: high-speed lines, cleanrooms, packaging automation, DCs. Example: USMGC mask lines; Akums sterile facility.
◉Typical raise: large growth equity ± debt, project finance.
Consolidation / M&A
◉Use: roll-up, integrating distribution networks, systems harmonization. Typically PE-led.
Valuation drivers & value levers investors watch
◉Recurring revenue share & renewal rates (per-unit purchases vs subscriptions) — subscription/managed inventory increases enterprise value multiples.
◉Gross margin by SKU — advanced dressings, specialty catheters > commodity gloves.
◉Customer concentration (GPO & direct contracts) — long-term GPO deals reduce revenue risk and support higher valuation.
◉Regulatory clearances & quality systems — FDA/CE/ISO create defensibility.
◉Manufacturing footprint & capacity utilization — higher utilization materially boosts cashflow.
◉Proprietary IP / differentiated materials — patents on polymers/coatings or exclusive supplier contracts.
◉Geographic revenue diversification — NA + APAC presence reduces single-market risk.
◉Supply-chain resilience — dual sourcing, local DCs, vertical integration (reduces input risk).
◉Data & tech stack — AI-enabled forecasting, lot traceability and e-commerce storefronts improve margins and reduce WC.
Key financial metrics and KPIs investors track
◉Annual Recurring Revenue (ARR) for subscription models.
◉Revenue retention: gross & net (cohort analysis for hospital/ASC customers).
◉SKU churn & SKU rationalization benefit (concentration of top 20 SKUs).
◉Gross margin % by product family (PPE vs advanced dressings vs catheters).
◉Contribution margin per procedure/kit.
◉Working capital days (inventory days, payables days).
◉Capacity utilization (% of installed lines in operation).
Order fill rate & on-time delivery %.
◉Cost per sterile unit (manufacturing overhead allocation).
◉R&D / regulatory spend as % revenue (for innovators).
◉Customer concentration (top 5 customers % of revenue).
◉EBITDA margin and free cash flow (for PE/strategic buyers).
Risk factors investors must quantify
◉Regulatory risk: delayed approvals or product non-compliance (FDA/EMA/PMDA) → revenue impact.
◉Environmental/regulatory pushback: plastic bans or costly disposal rules may compress margins or require capital to switch materials.
◉Commodity input volatility: resin/resin derivatives price swings materially affect low-margin plastics SKUs.
◉Customer concentration & GPO dependence — contract loss is high-impact.
◉Supplier single-sourcing & geo-political risk (e.g., import dependence).
◉Capex intensiveness: overbuilding capacity before demand can drive underutilization.
◉Reprocessing/reusable competitor cost-parity: if reuse models or sterilization tech materially reduce TCO for hospitals, unit demand may decline.
◉Reputational/product safety: contamination or failure incidents can lead to recalls and large liabilities.
◉Region-specific funding considerations & signals
North America
◉Strong private capital availability (PE/strategic). Expect larger multipliers for companies with GPO contracts and robust QA. Government incentives for reshoring (supply-chain resilience) can co-finance capex.
Asia-Pacific
◉Growth capital focused on local manufacturing scale-up and export capacity. National biotech & vaccine funding accelerates demand for single-use upstream consumables (bags, tubing sets). Investors should expect faster volume ramp potential but also price competition.
Europe
◉Funding appetite for sustainability-focused innovations is high (grants & green loans). Compliance with EU environmental directives influences capex for greener lines.
LatAm / MEA
◉Risk-adjusted returns higher; investors prefer distributors or asset-light plays rather than heavy manufacturing until demand and reimbursement structures stabilize.
Latest announcement
May 2025 — U.S. Medical Glove Company (USMGC)
◉What: Full-scale opening of a U.S. face-mask manufacturing facility with six high-speed lines for 3-ply disposable masks.
Why it matters:
◉Strengthens domestic PPE self-reliance, diversifying away from import shocks.
◉Adds redundant capacity for surge scenarios; shortens replenishment lead times.
◉Enhances supply-chain resiliency for U.S. healthcare networks and GPOs.
Recent developments
April 2025 — INTCO Medical
◉Launch: Syntex Synthetic Disposable Latex Gloves (patented).
◉Compliance: Passed EN455 / EN374; aligned to FDA and EU CE requirements.
Implications:
◉Raises quality/safety bar for latex alternatives; pushes performance parity with natural latex.
◉Broad cross-industry relevance (healthcare, food processing, industrial)—expands addressable demand.
May 2025 — Wadi Surgicals (India)
◉Launch: Enliva accelerator-free nitrile gloves (India’s first).
◉Clinical value: Minimizes accelerator-induced dermatitis/allergies; improves clinician comfort for prolonged use.
◉Strategic angle: Positions India as a specialty glove innovation hub; supports Asia’s fastest growth profile.
Segments covered
A) By Product Category
Infection Control & PPE (Gloves, Masks, Gowns)
◉Largest 2024 share (30%). Essential for HAI reduction and perioperative safety.
IV & Infusion Consumables (sets, tubing, connectors, filters)
◉Core for medication delivery; high volume, standardized SKUs.
Syringes, Needles & Safety Devices
◉Safety-engineered designs reduce sharps injuries; crucial for vaccines/biologics.
Catheters & Vascular Access (Peripheral & Central)
◉Fastest-growing product set; incidence of chronic disease and ICU use drives demand.
Surgical Disposables & Drapes
◉Maintains sterile field; customization via procedure packs.
Diagnostic & Sample Collection Kits
◉Swabs, tubes, POC consumables; standard in labs and outreach testing.
Wound Care Single-Use Dressings & NPWT Interfaces
◉Advanced dressings improve moisture management and healing trajectories.
Single-Use Endoscopes & Accessories
◉Reduces cross-contamination concerns; procedure-specific economics.
Urinary & Ostomy Consumables
◉Chronic care consumables with stable recurring use.
Other (feeding sets, nebulizer kits, sterilization pouches, trays)
◉Long-tail, essential support SKUs across wards.
B) By Material / Technology
Single-Use Plastics & Polymers (55% in 2024)
◉Dominant for cost/sterility/formability; compatible with multiple sterilization modes.
Non-woven Textiles
◉Foundation for gowns/drapes; comfort and barrier balance.
Advanced Dressings (hydrogel, alginate, foams)
◉Fastest-growing for wound complexity and aging populations.
Prefilled Glass/Plastic Syringes & Molded Components
◉Dose accuracy and aseptic handling benefits.
Biodegradable/Compostable Polymers
◉Emerging—addresses disposal cost and environmental pressure.
C) By Sterility / Use Case
Sterile Surgical & Procedural Consumables (45% in 2024)
◉OR/ICU critical supplies; stringent barrier and packaging needs.
Non-Sterile Patient Care Consumables (fastest-growing)
◉High-throughput, convenience-led, expanding in home care and general wards.
Single-Use Preloaded/Prefilled Systems
◉Reduces prep time, error risk; improves workflow standardization.
D) By End User / Care Setting
Hospitals & Acute Care (60% in 2024)
◉Largest concentration of procedures and critical care.
Ambulatory Surgery Centers & Outpatient Clinics (fastest-growing)
◉Minimally invasive & same-day care expand SKU turnover.
Long-Term Care & Nursing Homes / Home Healthcare / Diagnostic Labs & Reference Centers
◉Stable demand for non-sterile patient-care items and collection kits.
E) By Distribution Channel
Direct Sales & GPO Contracts (55% in 2024)
◉Pricing leverage, compliance, fill-rate SLAs.
Medical Distributors & Wholesalers
◉Breadth and regional reach.
Online / E-commerce & B2B Marketplaces (fastest-growing)
◉JIT ordering, SKU discoverability, analytics.
Retail Pharmacies
◉Outpatient & home-care consumables access point.
F) By Commercial Model / Pricing
Per-Unit Purchase / Consumable Sales (dominant)
◉Budget transparency; pay-as-used.
Managed Inventory / Subscription Bundles (fastest-growing)
◉Automated replenishment; lower stockouts and carrying cost.
Private-Label & Contract Manufacturing
◉Cost control and formulary standardization for large networks.
G) By Region
◉North America (leading share), Europe (steady), Asia-Pacific (fastest growth), Latin America & MEA (developing but rising)
◉Align product/price/channel to each region’s regulatory rigor, care-setting mix, and local manufacturing maturity.
Top-5 FAQs
1 What’s the market size now and by 2034?
US$ 2.99B (2024) → US$ 3.49B (2025) → US$ 13.83B (2034), CAGR 16.56% (2025–2034).
2 Which region leads and which grows fastest?
North America leads with 36–40% (2024); Asia-Pacific is fastest-growing.
3 Which product categories are largest vs. fastest?
Largest (2024): Infection-control & PPE (30%)
Fastest: Catheters & vascular access.
4 Which channels and models dominate?
Direct & GPO (55%) dominate; online/B2B grows fastest. Per-unit purchase dominates; managed inventory/subscriptions grow fastest.
5 What are the main headwinds?
Environmental impact & high disposal costs (waste handling/regulatory pressure), balanced by innovation in sustainable materials and AI-enabled efficiency.
Access our exclusive, data-rich dashboard dedicated to the Life Science Sector built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Immediate Delivery Available | Buy This Premium Research @ https://www.towardshealthcare.com/price/6070
Become a valued research partner with us – https://www.towardshealthcare.com/schedule-meeting
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Powering Healthcare Leaders with Real-Time Insights: https://www.towardshealthcare.com/healthcare-intelligence-platform
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
Web: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest