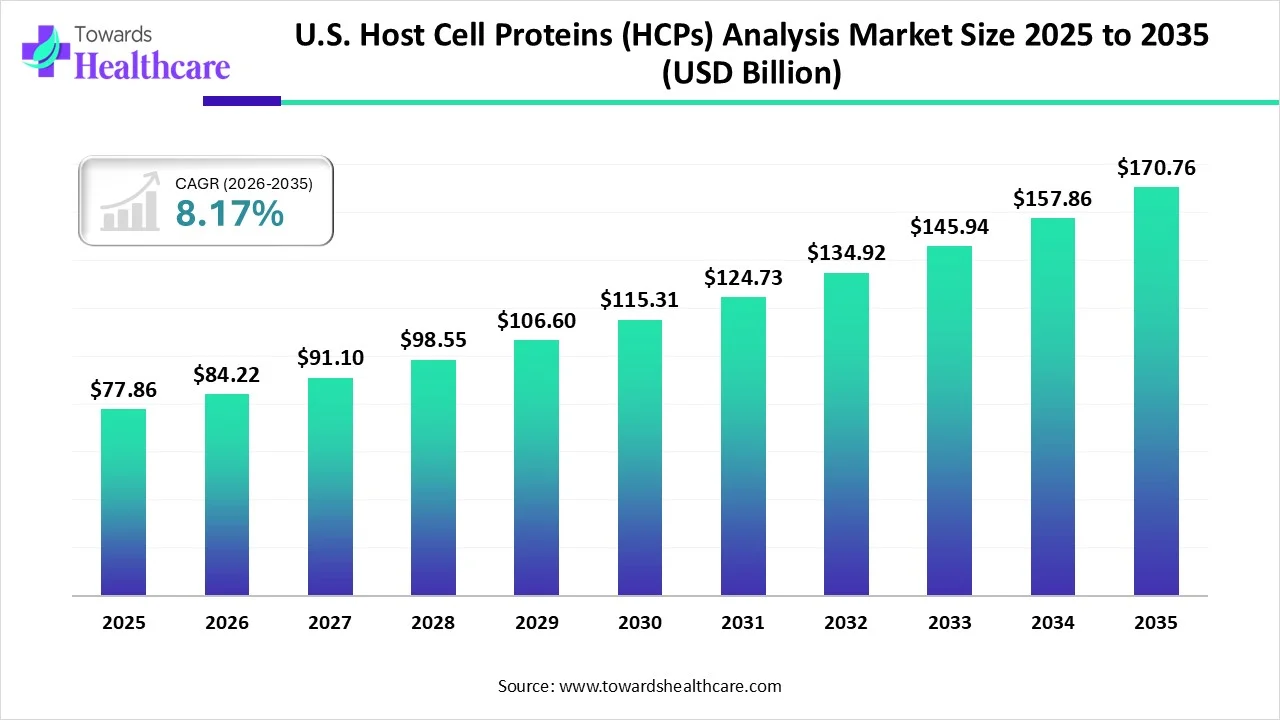
The U.S. host cell proteins (HCPs) analysis market is entering a decisive phase. With projections pointing toward USD 170.76 million by 2035, the industry is not just growing—it is evolving.
Download Free Sample: https://www.towardshealthcare.com/download-sample/6685
Biopharmaceutical innovation, stricter quality expectations, and advanced analytical tools are pushing HCP analysis into the spotlight. What was once a routine quality check is now a strategic pillar in drug development.
Why HCP Analysis Is No Longer “Just Testing”
HCPs are process-related impurities that come from host cells used in biologics production. Even trace amounts can impact drug safety and effectiveness.
Regulatory bodies are tightening expectations, and companies are responding by upgrading their testing standards. This shift is turning HCP analysis into a critical quality benchmark rather than a compliance checkbox.
As biologics and biosimilars grow in demand, the pressure to detect, quantify, and control HCPs is increasing significantly.
The U.S. Leads with Strong Scientific Momentum
The United States continues to dominate this market due to its deep-rooted biopharmaceutical ecosystem. From early-stage research to commercial manufacturing, HCP testing plays a role at every step.
High R&D investments, advanced lab infrastructure, and a strong presence of contract research organizations have created a robust environment for growth.
Another key driver is regulatory rigor. Strict guidelines are forcing companies to adopt more accurate and sensitive analytical methods.
Technology Is Redefining the Playing Field
Traditional methods like ELISA-based assays still hold the largest share, mainly because they are reliable and widely accepted.
However, newer technologies are quickly gaining ground. Mass spectrometry, especially LC-MS/MS, is transforming how companies analyze protein impurities.
Here’s how technology is shaping the market:
- ELISA continues to dominate due to its cost-effectiveness and established protocols
- Mass spectrometry offers deeper insights and higher precision in identifying complex impurities
- PCR-based assays are gaining traction for DNA-related impurity detection
This mix of traditional and advanced methods reflects a market that is balancing reliability with innovation.
AI Is Quietly Becoming a Game Changer
Artificial intelligence is beginning to reshape HCP analysis in subtle but powerful ways.
Instead of relying solely on manual interpretation, companies are now using AI to process complex datasets faster and more accurately. This reduces human error and speeds up decision-making.
AI also enables predictive impurity profiling. This means companies can anticipate potential risks before they occur, rather than reacting after detection.
In the long run, AI could redefine quality control by making it more proactive and less reactive.
Cell & Gene Therapy Is Fueling Demand
One of the biggest growth drivers in this market is the rise of cell and gene therapy. These advanced treatments require extremely high purity levels.
Even minor impurities can affect therapeutic outcomes, making HCP analysis essential.
Key application areas shaping demand include:
- Quality control, which ensures product safety and regulatory compliance
- Cell and gene therapy, which demands ultra-sensitive impurity detection
- Biopharmaceutical manufacturing, where large-scale production increases risk exposure
Among these, cell and gene therapy stands out as the fastest-growing segment, pushing companies to adopt more sophisticated testing approaches.
Biopharma Companies Take the Lead—But CROs Catch Up Fast
Biopharmaceutical companies currently dominate the market, accounting for the majority of HCP analysis activities. Their need to maintain quality across pipelines keeps demand steady.
However, contract research organizations (CROs) are expanding rapidly. Many companies are outsourcing testing to reduce costs and access specialized expertise.
This shift is creating a more collaborative ecosystem where CROs play a crucial role in innovation and scalability.
The Market Is Moving Toward Precision and Speed
The future of HCP analysis lies in precision, automation, and integration.
Companies are not just looking for detection—they want actionable insights. They want faster turnaround times without compromising accuracy.
As a result, we are seeing a shift toward integrated workflows that combine multiple technologies. This approach improves efficiency and delivers a more comprehensive understanding of impurities.
Final Thought: From Compliance to Competitive Advantage
HCP analysis is no longer a backend process. It is becoming a competitive differentiator.
Companies that invest in advanced technologies, AI-driven analytics, and robust testing strategies will gain a clear edge. They will not only meet regulatory standards but also build trust in their products.
In a market driven by innovation and precision, HCP analysis is quietly becoming one of the most important tools in the biopharmaceutical industry.
Access our exclusive, data-rich dashboard dedicated to the healthcare market – built specifically for decision-makers, strategists, and industry leaders. The dashboard features comprehensive statistical data, segment-wise market breakdowns, regional performance shares, detailed company profiles, annual updates, and much more. From market sizing to competitive intelligence, this powerful tool is one-stop solution to your gateway.
Access the Dashboard: https://www.towardshealthcare.com/access-dashboard
Checkout the Market Report Now at: https://www.towardshealthcare.com/checkout/6685
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Europe Region – +44 778 256 0738
North America Region – +1 8044 4193 44
APAC Region: +91 9356 9282 04
Visit Our Website: https://www.towardshealthcare.com
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium